1. The respiratory system is a highly ordered structure composed of over 40 cell types involved in a multitude of functions. Development of the lungs spans from embryogenesis to adult life, passing through several distinct stages of growth.
2. Oxidant gases, airborne particles and environmental tobacco smoke are common air pollutants that could have a significant impact on the lungs during both pre- and post-natal periods of life. Although the specific target cells for exposure to these pollutants are not clearly identified, these cells are likely to affect critical signals or mediators expressed during distinct stages of lung development.
3. Neonatal susceptibility to environmental pollutants may be due to either direct or indirect hits on a number of cell types to influence cell differentiation, proliferation and/or maturation. Air pollutants may also alter the normal developmental pattern for metabolic, immune and neurological functions which are constantly changing during in utero and postnatal growth.
4. The sensitivity of neonatal cells to environmental insults is likely to be completely different to these same cell types found in the adult. Delivery of an environmental toxicant to the respiratory system is also dramatically different during the fetal period compared to the postnatal period. Passage and interaction of environmental factors through other organ systems and the vasculature, as well as maternal influences must be taken into consideration when evaluating the impact of an environmental toxicant during early life.
5. To understand the heath outcomes of exposure to a variety of environmental factors in the respiratory system of children requires careful consideration that lung development is a multi-step process and cannot be based on studies in adults.
Exposure to a variety of toxicants and/or conditions during lung development has the potential to significantly affect the overall growth and function of the respiratory system in children. The target of a toxic insult to the lungs during development is likely to involve the disruption and/or alteration of a specific molecular signal or transcription factor, but to date, little information is available as to the precise effect of such exposures. Timing of exposure during development appears to be critical in the subsequent effects observed. For example, maternal malnutrition during gestation may significantly retard fetal growth and the development of the lungs leading to compromised lung function throughout life. In contrast, exposure to environmental toxicants such as second-hand cigarette smoke may actually accelerate the maturation of specific cell types in the fetal lung,1 but the effects of such a change on overall lung function in the newborn to the adult are unknown. In general, very little is known regarding the precise effects of maternal exposure of airborne particles on the fetus.
Cellular differentiation, branching morphogenesis and overall lung growth encompass several different phases of lung development. A growing body of evidence suggests that these processes can be affected by exposure to chemicals and particles. However, the effects of exposure are likely to be different during each phase of development. For example, during embryogenesis and progressive stages of fetal development, cell number, cell type and cell function of the airways and alveoli continue to change, and may be differentially affected based on the timing of the exposure. Since cells continue to differentiate and divide during the postnatal period, chemical exposure during the postnatal period is also likely to impact on the respiratory system in a different fashion, based on further changes in the cellular differentiation and anatomical growth.2 Since growth is essentially complete by the end of adolescence, exposure to chemicals and other factors are further likely to have completely different consequences in the adult compared to that found in children.2-4 Table 1 summarizes a number of differences in ventilatory parameters between children and adults. These differences are further
Table 1. Differences in ventilatory characteristics between infants and adults.
| Infants | Adults | |
| Tidal volume (ml/kg body weight) | 10 | 10 |
| Alveolar surface area (m2) | 3 | 75 |
| Respiration rate (breaths/min) | 40 | 15 |
| Minute ventilation (ml/kg body weight/m2 lung surface area/min) | 133 | 2 |
Exposure to substances during specific periods or windows of development may have profound effects that would not be seen if the same exposure were to occur in the adult. Since lung development occurs over the entire perinatal period, exposure effects can have significant consequences whether they occur during the pre- or postnatal period of life. Although our understanding of these changes at this time is extremely limited, it makes sense that abnormal developmental changes which occur in the perinatal period due to exposure to a variety of toxic chemicals, such as particulate matter, may have long-term effects persisting into adult life.
Development of the human respiratory system involves the formation of a highly ordered airway branching system with 25,000 distinct terminations giving rise to more than 300 million alveoli as well as the differentiation and proliferation of over 40 different cell types. The transition of the lungs from a simple protruding bud of tissue from the embryonic foregut into a highly organized, integrated, complex structure that is innervated, ventilated and vascularized is a multi-step process.
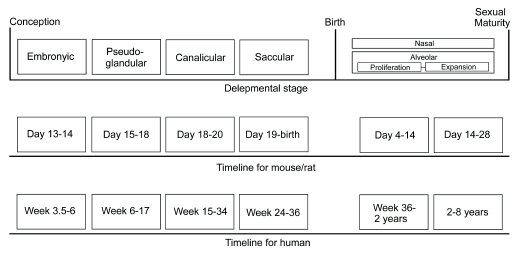
The development of the lungs begins in the embryo with the evagination of an avascular epithelial bud and subsequent growth into surrounding mesenchymal tissues. Following embryogenesis, the fetal lungs in all mammalian species undergo three anatomically distinct stages of growth termed pseudoglandular, canalicular and saccular5 (Figure 1). Although the lungs have developed sufficiently to sustain life at birth, growth is far from complete. Approximately 80 percent of alveoli in the human adult lung arise following birth. In essence, lung development is a continuum from embryogenesis to early adolescence.

Figure 1. Timeline for lung development in the mouse/rat and human respiratory system. Although all pass through identical phases of lung development, the timing for each phase is markedly different between species.
The stages of lung development are controlled by a variety of factors that modulate the timing and pattern of cellular proliferation and differentiation as well as branching morphogenesis. Recent reviews by Hackett and Gitlin (1997)6, Shannon and Deterding (1997)7 and Ramon (1998)1 stress the importance of a number of transcription factors, molecular signals and soluble factors in orchestrating the developmental process of the respiratory tract. These molecular signals are expressed in both a temporal and spatial pattern to facilitate normal lung development and to regulate epithelial-mesenchymal interactions, cellular proliferation, extracellular matrix deposition and composition, growth factor and receptor expression, as well as cell-to-cell interactions.
Many of the enzymes that play a critical role in lung metabolism are not fully developed at birth. A number of these enzymes are responsible for both the activation and detoxification of foreign or xenobiotic compounds. Among the most important enzyme systems to develop during the mid- to late gestational periods through to early childhood are the glutathione-S-transferases8 and epoxide hydrolases.9 Antioxidant enzymes including superoxide dismutase, catalase and glutathione peroxidase also appear during this same period.10-12
A highly critical family of isozymes involved in the bioactivation and detoxification of xenobiotic compounds is the cytochrome P450 monooxygenase system. The developmental profile of the cytochrome P450 monooxygenase system closely coincides with changes in subcellular composition of Clara cells and endothelial cells during the late gestational and early postnatal period of development. Although this family of isozymes may first appear near the end of gestation, they are more likely to develop during the postnatal period. A number of studies have shown that early postnatal exposure to environmental toxicants can alter the developmental profile for these isozymes.13
Administration of corticosteroids during lung development can also alter the normal process of lung maturation.14,15 The most likely target cell is the epithelial cell that may be altered by an accelerated maturation and a reduction in the rate of proliferation. Our knowledge of the precise effects of chemicals on lung development is still extremely limited. In addition to toxicants, it is also important to keep in mind that nutritional deficiency can significantly alter lung development.16
The effects of many toxicants on the respiratory system have been well characterized in the adult. Although less is known about the effects of toxicants in the developing lung, a number of toxicants are known to affect the developing lungs. These include environmental tobacco smoke,13,17-19 bioactivated compounds,2-4 and oxidant gases.20,21 The target for a number of these compounds is largely airway epithelial cells undergoing maturation and/or rapid proliferation. However, the precise mechanism leading to greater sensitivity of these cells in the neonate compared to the adult is still unknown.22 The impact that particles may have on lung, cardiovascular, immune development is expected to vary from that of adults, although the actual effects are not known.
The lungs are extremely sensitive to a large number of inhaled toxicants.23 Environmental tobacco smoke (ETS) is an excellent example of an airborne pollutant that contains many of these toxicants. ETS is defined as a combination of exhaled mainstream smoke and sidestream smoke given off from the smoldering end of a cigarette. A strong relationship has been demonstrated between respiratory illness in young children and ETS exposure.17 Exposure to ETS is also associated with significant risks in the development and/or exacerbation of asthma, airway hyperresponsiveness, and other respiratory symptoms such as cough, wheeze, and mucus production (Table 2). Epidemiological studies suggest that exposure to smoke during the perinatal period may have adverse effects on lung function that can persist into adulthood. However, the mechanisms leading to this process are unknown.
Table 2.
| Children raised in homes of smokers have: |
| Increased respiratory symptoms |
| Airway obstruction |
| Airway hyperactivity |
| Higher incidence of severity of clinical asthma |
Studies from our laboratory have used various exposure regimens to aged and diluted sidestream cigarette smoke (SS) as a surrogate to environmental tobacco smoke to determine the existence of critical windows of exposure during the perinatal period of development. Timed pregnant rats were exposed to environmentally relevant concentrations of SS using a concentration of 1 mg/m3 of total suspended particulates (TSP). Timed pregnant dams were examined under the following four exposure regimens: (1) exposure to SS only during the in utero period, (2) exposure to SS only during the postnatal period, (3) exposure to SS during both in utero and postnatal periods and (4) exposure to filtered air during both in utero and postnatal periods. All rat offspring were examined at 7 to 10 weeks of age.18 Animals exposed to SS during both the in utero and postnatal periods exhibited marked alterations in airway sensitivity (Figure 2) and pulmonary neuroendocrine cell (PNEC) frequency (Figure 3) in contrast to animals which had been exposed to SS only during the prenatal period or only to SS during the postnatal period, but not both. We hypothesize that these changes are due to SS exposure during critical windows of lung growth and development that include both fetal and early postnatal periods of life. Such exposure conditions are not uncommon in humans and appear to be essential for airway hyperresponsiveness to develop.
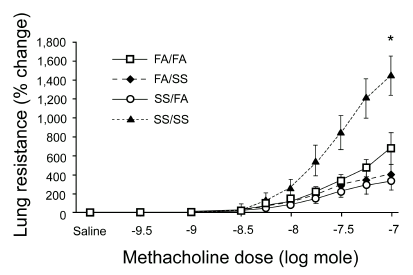
Figure 2. Methacholine-induced changes in lung resistance after differential timing of exposure to ETS during the perinatal period of life. Abbreviations: FA, filtered air; SS, sidestream cigarette smoke. Rats were exposed to FA and/or aged and diluted SS using four different timing regiments: a) exposure to FA during both in utero and postnatal periods (FA/FA), b) exposure to SS only during the postnatal period (FA/SS), c) exposure to SS only during the in utero period (SS/FA), and d) exposure to SS during both in utero and postnatal periods (SS/SS). Reproduced from Joad et al. 1995, with permission of Academic Press, Inc.
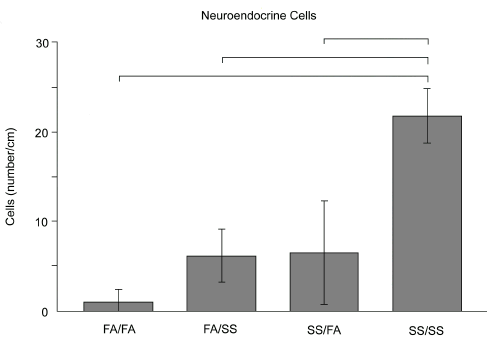
Figure 3. Pulmonary neuroendocrine cell number per centimeter basal lamina length in the airways of rats after differential exposure to ETS. Abbreviations: FA, filtered air; SS, sidestream cigarette smoke. Neuroendocrine cells were identified by neuron-specific enolase staining (Joad et al., 1995). Labeling of each exposure group is identical to that found in Figure 2.
Since differences noted in this study could also be attributed to the duration of exposure rather than the timing of exposure, we tested the hypothesis that early exposure to environmental tobacco smoke during the perinatal period is sufficient to produce a lasting response into adulthood. Maternal exposure to SS was begun on gestational day 5, followed by continued exposure to SS during the first 3 weeks of life. At 8 weeks of age, the airway reactivity of these animals was markedly increased compared to the lungs of rats exposed only to filtered air, despite the absence of SS exposure from 3 to 8 weeks of age.19 This model appears to be highly analogous to the development of pulmonary function decrements in children exposed to tobacco smoke during the perinatal period.24,25 Therefore, this animal model affords the opportunity to examine more precisely critical periods during lung development when exposure to SS leads to increased airway responsiveness and whether changes in PNEC number and/or function might be responsible for these changes. Critical windows of exposure during the perinatal period which affect airway reactivity are clearly defined in these experimental studies, but the long-term effects of such perinatal exposures into adulthood are not known.
To better understand the potential effects of critical windows of exposure in children on the respiratory system, it is important to consider the following factors that characterize the process of lung development. First, lung development is a multi-event process that is not restricted to prenatal life. Although the lungs undergo dramatic changes during the embryonic, pseudoglandular, canalicular and saccular stages, the majority of changes to the lungs continue postnatally during the process of alveolarization. Second, only a limited number of maturational events must be finished at birth for successful survival of the organism. Third, cellular differentiation, branching morphogenesis and overall growth define lung development during both the pre- and postnatal periods. Finally, all these developmental events occur in the presence of an increasing mass of total cells.
This work was funded in part by the National Institutes of Health (ES011634, ES05707, ES00628 and RR00169), the National Institute for Occupational Health Safety (OH07550), the United States Environmental Protection Agency (R829215) and the California Tobacco-Related Disease Research Program (7RT-0118 and 13RT-0004).
1. Ramon J. Cellular mechanisms of lung development. In: Fishman’s Pulmonary Diseases and Disorders, Third Edition, (AP Fishman, JA Elias, JA Fishman, MA Grippi, LR Kaiser, RM Senior, eds). 1998; McGraw-Hill, 73-89.
2. Smiley-Jewell S, Nishio SJ, Weir AJ, Plopper CG. Neonatal Clara cell toxicity by 4-ipomeanol alters bronciolar organization in adult rabbits. Am. J. Physiol., Lung Cell Mol. Physiol. 1998; 274 (4 part 1):L485-498.
3. Plopper CG, Weir AJ, Nishio SJ, Chang A, Voit M, Philpot RM, Buckpitt AR. Elevated susceptibility to 4-ipomeanol cytotoxicity in immature Clara cells of neonatal rabbits. J. Pharmacol. Exp. Ther. 1994; 269(2):867-80.
4. Fanucchi MV, Buckpitt AR, Murphy ME, Plopper CG. Naphthalene cytotoxicity of differentiating Clara cells in neonatal mice. Toxicol. Appl. Pharmacol. 1997; 144(1):96-10.
5. Burri PH. Lung development and pulmonary angiogenesis. In: Lung Development (C. Gaultier, JR Bourbon, M. Post, Eds). New York:Oxford University Press. 1999; 122.
6. Hackett BP, Gitlin JD. Role of transcription factors in the development of the pulmonary epithelium. In: Lung Growth and Development, Vol 100. New York: Marcel Dekker Inc. 1997; 55-80.
7. Shannon JM, Deterding RR. Epithelial-mesenchymal interactions in lung development. In: Lung Growth and Development, (ed. JA McDonald). Vol 100, New York: Marcel Dekker Inc. 1997; 81-118.
8. Fryer AA, Hume R, Strange RC. The development of glutathione-S-transferase and glutathione peroxidase activities in human lung. Biochim. Biophys. Acta. 1986; 883(3):448-453.
9. Omiecinski C, Aicher L, Swenson L. Developmental expression of human microsomal expoxide hydrolase. J. Pharmacol. Exp. Ther. 1994; 269(1):417-423.
10. Tanswell AK, Freeman BA. Pulmonary antioxidant enzyme maturation in the fetal and neonatal rat. I. Developmental profiles. Pediatr. Res. 1984; 18(7):584-587.
11. Hayashibe H, Asayama K, Dobashi K, Kato K. Prenatal development of antioxidant enzymes in rat lung, kidney, and heart: Marked increase in immunoreactive superoxide dismutases, glutathione peroxidase, and catalase in the kidney. Pediatr. Res. 1980; 27(5):472-475.
12. Rickett GM, Kelly FJ. Developmental expression of antioxidant enzymes in guinea pig lung and liver. Development. 1990; 108(2):331-336.
13. Gebremichael A, Chang AM, Buckpitt AR, Plopper CG, Pinkerton KE. Postnatal development of cytochrome P450 1A1 and 2B1 in rat lung and liver: Effect of aged and diluted sidestream cigarette smoke. Toxicol. Appl. Pharmacol. 1995; 135(2):246-253.
14. Massaro GD, Massaro D. Formation of alveoli in rats: postnatal effect of prenatal dexamethasone. Am. J. Physiol. 1992; Jul, 263(1 Pt 1):L37-41.
15. Pinkerton KE, Willet KE, Peake JL, Sly PD, Jobe AH, Ikegami M. Prenatal glucocorticoid and T4 effects on lung morphology in preterm lambs. Am. J. Respir. Crit. Care Med. 1997; 156:624-630.
16. Lechner AJ. Perinatal age determines the severity of retarded lung development induced by starvation. Am. Rev. Resp. Dis. 1985; 131:638-643.
17. U.S. Environmental Protection Agency. Respiratory health effects of passive smoking: lung cancer and other disorders. 1992; EPA/600/6-90/006F.
18. Joad JP, Ji C, Kott KS, Bric JM, Pinkerton KE. In utero and postnatal effects of sidestream cigarette smoke exposure on lung function, hyperresponsiveness, and neuroendocrine cells in rats. Toxicol. Appl. Pharmacol. 1995; 132(1):63-71.
19. Joad JP, Bric JM, Peake JL, Pinkerton KE. Perinatal exposure to aged and diluted sidestream cigarette smoke produces airway hyperresponsiveness in older rats. Toxicol. Appl. Pharmacol. 1999; 155(3):253-260.
20. Randell SH, Mercer RR, Young SL. Postnatal growth of pulmonary acini and alveoli in normal and oxygen-exposed rats studied by serial section reconstructions. Am. J. Anat. 1989; 186(1):55-68.
21. Gunnison AF, Weideman PA, Sobo M, Koenig KL, Chen LC. Age-dependence of responses to acute ozone exposure in rats. Fund. Appl. Toxicol. 1992; 18(3):360-9.
22. Pinkerton KE, Zhou Y-M, Teague SV, Peake JL, Walther RC, Kennedy IM, Leppert VJ, Aust AE. Reduced lung cell proliferation following short-term exposure to ultrafine soot and iron particles in neonatal rats: Key to impaired lung growth? Inhalation Toxicol. 2004; 16(suppl. 1):73-81.
23. Yost G, Buckpitt A, Roth R, McLemore R. Mechanisms of lung injury by systemically administered chemicals. Toxicol. Appl. Pharmacol. 1989; 101(2):179-195.
24. Hanrahan JP, Tager IB, Segal MR, Tosteson TD, Castile RG, van Vunakis H, Weiss ST, Speizer FE. The effect of maternal smoking during pregnancy on early infant lung function. Am. Rev. Respir. Dis. 1992; 145:1129-1135.
25. Cunningham J, Dockery DW, Speizer FE. Maternal smoking during pregnancy as a predictor of lung function in children. Am. J. Epidemiol. 1994; 139:1139-1152.