1. In muscle, intracellular calcium concentration, hence skeletal muscle force and cardiac output, is regulated by uptake and release of calcium from the sarcoplasmic reticulum. The ryanodine receptor (RyR) forms the calcium release channel in the sarcoplasmic reticulum.
2. Calcium release through RyRs is modulated by a wide variety of endogenous molecules including small diffusible ligands such as ATP, Ca2+ and Mg2+. The regulation of RyR channels by ATP, Ca2+ and Mg2+ is a complex interplay of several regulatory mechanisms which are still being unravelled. Consequently it is not clearly known how RyRs are regulated in resting muscle and during contraction. Reviewed here are:
3. Factors controlling the activity of RyRs in skeletal and cardiac muscle with an emphasis on mechanistic insights derived from single channel recording methods.
4. The nature of dihydropyridine receptor (DHPR) control of RyRs in skeletal muscle derived from experiments with peptide fragments of the DHPR II-III loop.
5. Recent experiments on coupled RyRs in lipid bilayers and their potential for resolving the elusive mechanisms controlling calcium release during cardiac contraction.
“Almost everything we do is controlled by calcium”.1 The vast array of processes governed by intracellular calcium signals relies on their precise spatial and temporal control.2 To achieve this cells have evolved intracellular stores that provide a stable, reliably controlled release of calcium into the cytoplasm. Two types of calcium ion channels are known to provide the calcium release pathway from intracellular stores, namely ryanodine receptors (RyRs) and inositol 1,4,5-trisphosphate receptors.
In striated muscle (skeletal and cardiac), the sarcoplasmic reticulum (SR) is the calcium store from which calcium release through RyRs is the key determinate of muscle force. The RyR has several isoforms found in different animal species. In mammals three isoforms have been cloned and sequenced: RyR1 in skeletal muscle; RyR2 in cardiac and smooth muscle; and RyR3 in many cell types. The two isoforms found in amphibian, fish and avian muscle (RyRα and RyRβ) are similar to RyR1 and RyR3 respectively.3 Invertebrates carry a single RyR isoform which is similar to RyR2.4
During excitation contraction coupling (EC coupling) in muscle, depolarisation of the surface membrane and transverse (t)-tubules causes release of Ca2+ from the sarcoplasmic reticulum (SR). This leads to an increase in cytoplasmic [Ca2+], which in turn is the signal for contraction. Membrane depolarisation activates dihydropyridine receptors (DHPR, L-type calcium channels) which trigger the opening of RyRs. The EC coupling mechanisms in skeletal and cardiac muscle differ. In the heart, t-tubule depolarization triggers an influx of Ca2+ through the DHPR which activates RyRs and calcium release.5-8 In skeletal muscle the influx of Ca2+ is not required9 and the trigger for RyR activation depends on a physical link between the DHPR and the RyR.10 Discussed below are the ways in which cardiac and skeletal RyRs (mammalian isoforms RyR1 and RyR2) are differently regulated by components of the cytoplasm and lumen and how this underlies the differences in skeletal and cardiac EC coupling.
In the absence of Mg2+, the activity of RyR1s has a bell-shaped dependence on cytoplasmic [Ca2+]. RyR1s are activated by ∼1 μM cytoplasmic [Ca2+] and exposure of their cytoplasmic face to >1 mM [Ca2+] will inhibit them.11 In resting muscle, cytoplasmic [Ca2+] is 100-150 nM12,13 whereas during muscle contraction the cytoplasmic [Ca2+] rises to ∼20 μM.14
ATP is a RyR agonist which can activate RyR1s in the virtual absence of cytoplasmic Ca2+, and in conjunction with Ca2+, can cause almost full activation. Activation of RyR1 by ATP and its non-hydrolysable analogue AMP-PCP has been reported to have a Ka in the range 0.2 to 1 mM.15-18 In muscle [ATP] ∼8 mM,19 facilitating near maximal ATP activation of RyRs. Therefore, in the absence of Mg2+ (see below), ATP alone can trigger SR Ca2+ release in resting skeletal muscle. The subsequent rise in cytoplasmic Ca2+ will reinforce RyR activation, a process called Ca2+ induced Ca2+ release (CICR).
In resting skeletal muscle the RyRs are inactive because of the inhibitory effects of cytoplasmic Mg2+. The total Mg2+ concentration in the cytoplasm is ∼9 mM,19 much of which is bound to ATP so that the free Mg2+ is ∼1 mM.20,21 Skinned fibre experiments by Lamb and Stephenson22,23 have shown that a reduction of cytoplasmic free Mg2+ from physiological levels to ≤0.2 mM causes Ca2+ release from the SR. Mg2+ is believed to inhibit RyRs by two mechanisms (the dual-inhibition hypothesis17,24). Mg2+ can inhibit RyRs by competing with Ca2+ for the activation sites (A-sites) or Mg2+ can close RyRs by binding to low affinity, non-selective divalent cation inhibition sites that also mediate Ca2+-inhibition (I-sites). The dual inhibition model predicts that competition between Ca2+ and Mg2+ at the A-sites will only produce significant Mg2+-inhibition when cytoplasmic Ca2+ is less than 1 μM (i.e. when the muscle is at rest). On the other hand, I-sites produce Mg2+-inhibition with Ki ∼200 μM over the entire physiological Ca2+ range.24,25
The properties of RyR1s measured in isolation are consistent with the calcium permeability of the SR in resting muscle. However, during EC coupling the regulation of RyRs by cytoplasmic ligands is substantially altered. At rest, Mg2+ is the primary inhibitor of Ca2+ release from the SR. During t-tubule depolarization and activation of the RyR by the DHPR, the sensitivity of the RyRs to inhibition by Mg2+ is reduced by more than 10-fold.23,26,27 This is thought to be due to a reduction in the Mg2+ affinity of the I-sites and A-sites.24,27,28 This would relieve the Mg2+-inhibition of the RyR, permitting RyR activation by ATP and its reinforcement by CICR (the Mg2+ de-repression hypothesis26). Upon repolarisation of the surface membrane Mg2+-inhibition is reinstated. Since the Mg2+ affinity of the I-sites is relatively insensitive to Ca2+, the I-sites would again be able to inactivate the RyRs even though the cytoplasmic [Ca2+] is elevated during muscle excitation. The Mg2+ de-repression hypothesis is further supported by the observation that depolarization induced SR calcium release can be substantially inhibited by reduced [ATP] (0.5 mM) or by ATP antagonists such as adenosine.18,29,30 This indicates that the DHPR stimulation of RyRs is not sufficient on its own for Ca2+ release and that stimulation of RyRs by intracellular ligands is necessary for EC coupling.18
Several regions of the DHPR are involved in its physical interaction with the RyR1 in skeletal muscle.31 Expression of cDNAs encoding chimeric constructs of skeletal and cardiac DHPR α1 subunits in myotubes have demonstrated that the cytoplasmic loop between repeats II and III of the skeletal DHPR α1 subunit (666Glu-Leu791),32 and more specifically residues 725Phe-Pro742,33 are crucial to skeletal EC coupling. Application of peptide corresponding to the II-III loop to isolated RyRs resulted in activation of skeletal but not cardiac RyRs in some studies but not in others.34,35 Shorter sequences have been synthesized to determine the specific regions of the II-III loop involved in RyR regulation.36 Two peptides, AS (671Thr-Leu690) and CS (724Glu-Pro760), were found to interact with isolated RyRs.36-42
Of particular interest is the CS peptide which is the region of the II-III loop which is thought to be a determinant of skeletal EC coupling. Application of this peptide to isolated skeletal RyRs resulted in a decrease in their Mg2+-inhibition which was mediated by a 2-fold increase in the Ki of the I-sites.42 This result shows the striking parallel between the actions of the peptide and changes in RyR activity during EC coupling. The 2-fold change in RyR affinity for Mg2+ caused by CS is much smaller than the 10-100 fold reduction that occurs during EC coupling in skinned muscle fibres23,27 and in triad preparations.43 However, it would be surprising if the mere presence of the CS peptide mimicked EC coupling because the peptide fragment itself does not possess the DHPR sensor for membrane depolarisation and the bilayer experiments are incapable of inducing conformational changes in the peptides that mimic depolarisation. The effect of CS does indicate an interaction between the 724Glu-Pro760 region of the DHPR and the part of the RyR which affects the I-sites. During EC coupling this interaction could indeed produce large changes in Mg2+-inhibition by transmitting depolarisation induced conformational changes from the DHPR to the RyR.
The precise location of the A- and I- Mg2+/Ca2+ sites in the RyR protein are not yet known but it was clear at an early stage that these sites are located in very different parts of the RyR protein.44 Electron microscope image reconstruction shows RyRs to have a large cytoplasmic domain (the foot region) and a relatively small trans-membrane region that forms the Ca2+ pore.45 The RyR is a homotetramer of ∼560 kDa subunits containing ∼5035 amino acids (aa). The trans-membrane pore is comprised of the ∼1000 C-terminal amino acids (aa 4000-5000) and the remaining amino acids form the foot region. Expression of the RyR C-terminus produces Ca2+ channels with similar conductance and Ca2+-activation properties as full RyRs46 but they lack regulatory sites for Ca2+-inhibition. Thus it appears that the A-sites are located in the C-terminal region whereas the I-sites are located in the foot region. RyR aa 4032 has been proposed to form part of the A-site because the E4032A substitution decreases RyR1 sensitivity to Ca2+-activation by four orders of magnitude.47 However, this is not settled yet because others have proposed that aa 4032 is a crucial part of the signal transduction between the A-site and channel gating.48
A likely candidate for the I-site is the very distinctive sequence of 30 consecutive negative amino acids (with only one intervening positive residue) at positions 1873-1903 in RyR1. Such a string of negative charges could form a low affinity, low specificity cation binding site. Significantly, the homologous sequence in the cardiac RyR2, where the divalent cation affinity is 10-fold lower (see below), has a far lower net charge. Moreover, deletion of aa 1641-2437 from RyR1 appeared to reduce the I-site affinity by 10-fold without effecting the A-site.49 Another candidate for the I-site is in the amino acids near position 615. The R615C mutation in pig RyRs (leading to malignant hyperthermia) causes a 3-fold reduction in the I-site affinity for Ca2+ and Mg2+.25 It is quite possible that both these regions could form the I-sites via inter-domain interactions between these widely separated sections of the amino acid sequence.
The part of the RyR I-sites that encompass the Arg615 residue may also be important for EC coupling. Several lines of evidence indicate that the DHPR II-III loop interacts with RyRs somewhere between aa 450-1500. 1) The peptide CS has recently been shown to bind to the region between aa 450 and 1400 on RyR1.50 2) RyR peptides from within this region (922-1112 and 1303-1406) bind to both II-III and III-IV loops of the DHPR.51, 52 3) Aa 1303-140653 and 1272-1455 of RyR154 have been identified as critical for EC coupling. 4) The Arg615Cys mutation in pig skeletal RyRs not only reduces I-site affinity but also reduces the activating effect of CS.55
Thus, an overall picture is emerging in which depolarization induced conformation changes in the DHPRs are transmitted to the RyR1 region (aa 450-1500) via the DHPR II-III loop. The 450-1500 region of the RyR also contains the I-site for Mg2+-inhibition which undergoes a marked decrease in Mg2+ affinity in response to DHPR depolarisation.23 In conjunction with this the DHPRs reduce the Mg2+ affinity of the A-sites. It has been proposed that DHPRs might do this by commandeering the mechanism that decreases the A-site affinity in response to elevated [Ca2+] in the SR.28 The subsequent alleviation of Mg2+-inhibition permits RyR activation by ATP and its reinforcement by CICR.
Regulation of cardiac RyRs by Ca2+ and Mg2+ and ATP operate by similar mechanisms as they do in skeletal RyR1s except that the ligand sensitivities vary between the two isoforms. Cardiac RyR2s are not appreciably activated by ATP in the absence of Ca2+ but ATP augments their activation by Ca2+.29,56 In RyR2 the I- sites have a 10-fold lower affinity for divalent ions than RyR1.24,44 The dual-inhibition model predicts marked differences in the way Mg2+-inhibition affects RyR1 and RyR2. At resting [Ca2+], RyR1 and RyR2 are similarly inhibited by Mg2+ thus it can be regarded as the brake on cardiac Ca2+ release at rest. However, at elevated cytoplasmic Ca2+ (∼10 μM) which occurs during muscle contraction, the I-sites produce relatively little inhibition of RyR2 compared to RyR1. Thus in cardiac muscle raised cytoplasmic Ca2+ is sufficient to alleviate the Mg2+-inhibition of RyRs.
Our present understanding of the regulation mechanisms in RyR2 fails to explain the stability of calcium release in cardiac muscle. Depolarisation of the surface membrane initiates release by providing a small increase in cytoplasmic calcium concentration which relieves Mg2+-inhibition and ‘kick starts’ RyR opening. The subsequent release of calcium from the SR further increases the cytoplasmic calcium concentration and strongly reinforces RyR activation by CICR. The inherent amplification of CICR underlies nearly all of the calcium released from the SR.5 CICR should provide an explosive, positive feedback in calcium release that will completely empty the SR by any stimulus from the surface membrane. Yet in spite of this, the quantity of calcium released from the cardiac SR has a graded and stable dependence on the magnitude of the calcium inflow through the surface membrane.57 This is the “paradox of control” of cardiac EC coupling. To demonstrate a negative feedback mechanism that breaks the CICR cycle is the main challenge to understanding control of calcium release.58
Calcium sparks once provided a ray of hope for understanding cardiac EC coupling. It is now understood that the graded response of Ca2+ release in cells is due to recruitment of different numbers of regenerative Ca2+ release events that are localised at the triad junctions. These release events are called sparks because they appear as localised bursts of light (∼2 μm diameter) in the presence of fluorescent Ca2+ indicators. Their fluorescence time-course has an exponential rising phase lasting 10-20 ms which corresponds to the time that the RyRs are open and releasing Ca2+. When the RyRs close and Ca2+ release is terminated, the fluorescence declines to baseline within ∼50-100 ms. Termination of Ca2+ release has been shown to be an inactivation mechanism with a refractory period of ∼30 ms.59 The spark intensities appear to be quantised, indicating that Ca2+ sparks involve the coordinated activation of 2-10 RyRs.60 The discovery of sparks appeared to resolve the “paradox of control” at a cellular level only to have the same paradox reappear in our understanding of spark termination.58 Ca2+ release in a spark is self reinforcing, yet the mechanisms underlying the termination of Ca2+ release remains unknown. Therefore the “paradox of control” reduces to the question of how RyRs can rapidly inactivate after they have been triggered by Ca2+ influx through the DHPRs.
One of the most powerful techniques used to understand the basic mechanisms of Ca2+ release, has been to extract the Ca2+ release channels from muscle and study them in isolation, in artificial bilayers, thus obviating the complexities of cellular function.61 The intransigence of the cardiac EC coupling problem lies in a failure of bilayer measurements to yield a plausible mechanism for termination of Ca2+ release.58 This may be because Ca2+ sparks result from cooperative activation of RyRs in close packed arrays while isolated RyRs in bilayers have been thought to be too widely separated to mimic these interactions.

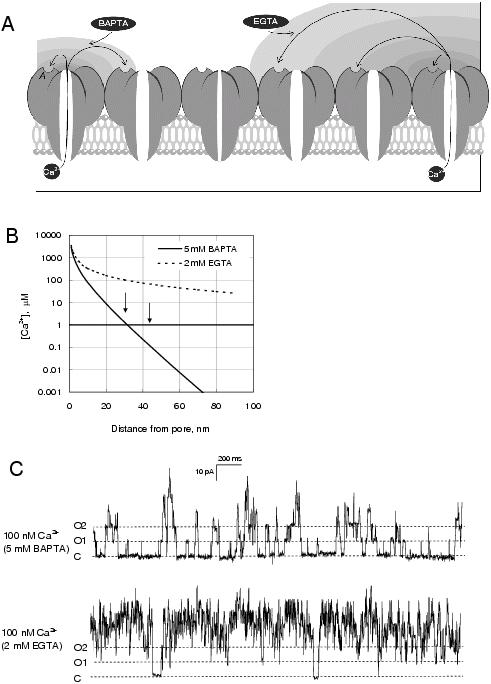
Figure 1. (A) A schematic illustrating the Ca2+ diffusion mechanism of RyR coupling in lipid bilayers. Ca2+ flowing from the SR lumen through the channel can activate neighbouring channels via their cytoplasmic Ca2+ activation sites (A-sites- labelled A on the left-most channel). The SR lumen is on the lower side of the membrane. The shading on the higher side of the membrane is a schematic of the free [Ca2+] profile due to its efflux from the SR lumen. The average distance from the pore that free Ca2+ can diffuse depends on the rate at which Ca2+ can bind to chelating agents in the bath. (B) The predicted free [Ca2+] profiles near the cytoplasmic side of the pore during Ca2+ efflux driven by 40 mV and 1 mM luminal Ca2+.67 The arrows indicate the inter-pore separations of RyRs (nearest and second nearest neighbours) in their triadic arrays determined from electron microscopy.68 (C) Recordings from an experiment with 7 cardiac RyRs in the bilayer showing coupled gating at 20 mV. The dashed lines indicate the current baselines (C) and openings of the first two channels (O1 and O2). The baths contained symmetric 250 mM Cs+ solutions with 2 mM ATP, cytoplasmic [Ca2+]c = 100 nM and luminal [Ca2+]l =0.1 mM. The channel activity depended strongly on the Ca2+ buffer. Free [Ca2+] was buffered to 100 nM by 5 mM BAPTA (top trace) and 2 mM EGTA (bottom trace, same bilayer). RyR activity was considerably greater in the presence of EGTA.
An inroad into this problem has been uncovered in recent years. A few studies have reported that when several RyRs are present, the opening of some RyRs depend on the opening of others (i.e. the gating of these RyRs are coupled) indicating that RyRs can form relatively stable, closely packed aggregates in the bilayer. Coupling of RyRs was first identified in CHAPS-purified RyR162 and RyR2.63 Those studies found that protein fractions enriched in RyR multimers could produce synchronously gated channels. This phenomenon required FK506 Binding Proteins (FKBPs) and was independent of luminal [Ca2+], suggesting that coupling arose from a direct physical link between RyRs. Another distinct phenomenon was reported where multiple RyR1 or RyR2 channels incorporated into bilayers from SR vesicles were clearly coupled but did not open in synchrony.28,64-66 These studies found that coupled gating required the flow of Ca2+ from lumen to cytoplasm and it was proposed that Ca2+ flow from one channel raised the local [Ca2+] sufficiently to activate neighbouring RyRs in the bilayer (Figure 1A).
The concentration profile of free Ca2+ near the cytoplasmic pore mouth depends on the Ca2+ flux through the pore and on the Ca2+ buffering in the cytoplasmic bath67 (Figure 1A&B). For example, the Ca2+ binding rate of EGTA is ∼1000-fold slower than BAPTA which means that Ca2+ emanating from the pore will diffuse a greater distance before it binds to EGTA than to BAPTA. It can be seen in Figure 1B that in the presence of BAPTA the free [Ca2+] falls to sub-activating levels (<1 μM) within 35 nm of the pore whereas in the presence of EGTA free [Ca2+] is still at activating levels beyond 100 nm.
Figure 1C shows recordings of coupled cardiac RyRs in a lipid bilayer. In the top trace, the free [Ca2+] is buffered to 100 nM by 5 mM BAPTA. The opening of a single RyR, shown by current transitions from the baseline (C) to the first level (O1), occurs every 200 ms on average. Once a single RyR has opened the other channels open relatively quickly (<20 ms) producing current transitions to high levels (O2 and above). In this record the opening of a single RyR increased the opening rate of its neighbours ∼10-fold. The importance of the Ca2+ buffering is apparent by comparing the top and bottom traces. In the bottom trace BAPTA was replaced by 2 mM EGTA which substantially enlarges the domain of elevated Ca2+ near the pore mouth. The opening rate of the first channel was not changed. However, activity of other channels in response to the first opening was greatly increased. This is most likely because in the presence of EGTA a single RyR can recruit more of its neighbours (Figure 1A) and because the local free [Ca2+] is higher.
Interestingly, coupling persisted even when the BAPTA confined elevated cytoplasmic [Ca2+] to within 35 nm of the pore.28,66 From these observations the separation of RyRs in the bilayer was estimated to be ∼30 nm. This separation (see arrows in Figure 1B) is consistent with the dimensions of RyR spacing in triad junctions, indicating that during isolation and reconstitution, some of the RyR arrays in muscle remain in tact and that rafts containing 3-10 RyRs remain stable in lipid bilayers. The mechanism linking the RyRs in these arrays is not known but this link is not in itself the mechanism for coupling.
Experiments on coupled channels will allow detailed investigations of the mechanisms underlying the termination of Ca2+ release that would not be possible with single channels. When the activation of RyRs is synchronised then inactivation processes operating within RyRs become apparent. To illustrate the way in which coupled RyRs could be used to resolve these mechanisms we consider the effect of cytoplasmic Mg2+ on the coupled gating of skeletal RyRs (Figure 2A). Like the coupled gating seen in Figure 1, the first channel opening in the bilayer triggers the opening of other channels (see arrow marked ‘a’). Under the conditions in Figure 2A the channel openings are such that closure of all the channels in the membrane occurs on average once every 50-100 ms (arrows ‘a’ and ‘b’). The bursts of channel activity demarcated by these closures are too long to account for the duration of Ca2+ release occurring during a Ca2+ spark. However, Mg2+-inhibition is known to decrease the mean open time of RyRs in single channel recordings. When added to coupled channels, Mg2+ causes a reduction in open duration that reduces the burst duration to < 10 ms (Figure 2B & C, arrows ‘c’ and ‘d’). The activity of Mg2+-inhibited RyR clusters in the bilayer parallels the time course of Ca2+ release during a calcium spark. However, it is not yet clear if intracellular Mg2+ has any role in terminating calcium release in cardiac muscle.
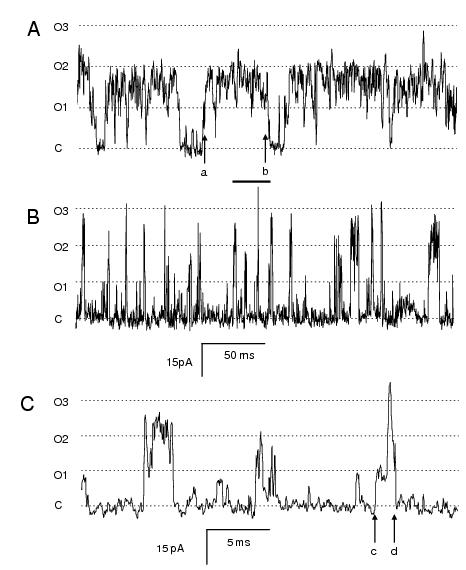
Figure 2. (A) Recordings from an experiment with 3 skeletal RyRs in a bilayer showing coupled gating. The baths contained symmetric 250 mM Cs+ solutions with 2 mM ATP, [Ca2+]c = 100 nM (with 5 mM BAPTA) and [Ca2+]l =1 mM. The channel activity was recorded at -40 mV. (B & C) The same experiment after addition of 0.5 mM Mg2+ to the cytoplasmic bath. (C) An expanded section of (B) indicated by the horizontal bar. The features labelled ‘a’-‘d’ are described in the text.
Even though skeletal and cardiac RyRs have the same basic regulation mechanisms, differences in the ligand sensitivities of these mechanisms significantly alter the way in which they are controlled by the cell. In skeletal muscle, the RyR I-sites have sufficiently high Mg2+ affinity to inhibit Ca2+ release. During EC coupling, Ca2+ release is triggered via a DHPR induced reduction in the A- and I-site affinity for Mg2+. Control of RyR1 by the I-site gives DHPRs the ability to terminate Ca2+ release upon repolarisation, thus allowing tight control of Ca2+ release by the surface membrane. In cardiac muscle, the I-sites have a 10-fold lower affinity for Mg2+ and are not utilised by the DHPR to regulate Ca2+ release. Instead, Ca2+ release is triggered by the influx of Ca2+ through the surface membrane. How this release process is controlled by the surface membrane is not yet understood. However, recent developments in recording of RyR arrays should pave the way for a much improved understanding of cardiac EC coupling.
Thanks to Katherine Bradley for assisting with the figures. This work was supported by the National Health & Medical Research Council of Australia (grant number 234420) and by an infrastructure grant from NSW Health through Hunter Medical Research Institute.
1. Berridge, MJ, Bootman, MD, Lipp, P. Calcium--a life and death signal. Nature 1998; 395: 645-648.
2. Berridge, MJ, Lipp, P, Bootman, MD. The versatility and universality of calcium signalling. Nat. Rev. Mol. Cell Biol. 2000; 1: 11-21.
3. Sutko, JL, Airey, JA. Ryanodine receptor Ca2+ release channels: does diversity in form equal diversity in function? Physiol. Rev. 1996; 76: 1027-1071.
4. Maryon, EB, Coronado, R, Anderson, P. unc-68 encodes a ryanodine receptor involved in regulating C. elegans body-wall muscle contraction. J. Cell Biol. 1996; 134: 885-93.
5. Fabiato, A. Simulated calcium current can both cause calcium loading in and trigger calcium release from the sarcoplasmic reticulum of a skinned canine cardiac Purkinje cell. J. Gen. Physiol. 1985; 85: 291-320.
6. Nabauer, M, Callewaert, G, Cleemann, L, Morad, M. Regulation of calcium release is gated by calcium current, not gating charge, in cardiac myocytes. Science 1989; 244: 800-803.
7. Nabauer, M, Ellis-Davies, GC, Kaplan, JH, Morad, M. Modulation of Ca2+ channel selectivity and cardiac contraction by photorelease of Ca2+. Am. J. Physiol. 1989; 256: H916-H920.
8. Beuckelmann, DJ, Wier, WG. Mechanism of release of calcium from sarcoplasmic reticulum of guinea-pig cardiac cells. J. Physiol. (Lond.) 1988; 405: 233-255.
9. Armstrong, CM, Bezanilla, FM, Horowicz, P. Twitches in the presence of ethylene glycol bis( -aminoethyl ether)-N,N'-tetracetic acid. Biochim. Biophys. Acta. 1972; 267: 605-608.
10. Schneider, MF, Chandler, WK. Voltage dependent charge movement of skeletal muscle: a possible step in excitation-contraction coupling. Nature 1973; 242: 244-246.
11. Meissner, G. Ryanodine receptor/Ca2+ release channels and their regulation by endogenous effectors. Ann. Rev. Physiol. 1994; 56: 485-508.
12. Perez, CF, Lopez, JR, Allen, PD. Expression levels of RyR1 and RyR3 control resting free Ca2+ in skeletal muscle. Am. J. Physiol. 2005; 288: C640-C649.
13. Lopez, JR, Contreras, J, Linares, N, Allen, PD. Hypersensitivity of malignant hyperthermia-susceptible swine skeletal muscle to caffeine is mediated by high resting myoplasmic [Ca2+]. Anesthesiology 2000; 92: 1799-1806.
14. Hollingworth, S, Zhao, M, Baylor, SM. The amplitude and time course of the myoplasmic free [Ca2+] transient in fast-twitch fibers of mouse muscle. J. Gen. Physiol. 1996; 108: 455-469.
15. Jona, I, Szegedi, C, Sarkozi, S, et al. Altered inhibition of the rat skeletal ryanodine receptor/calcium release channel by magnesium in the presence of ATP. Pflugers Arch. 2001; 441: 729-738.
16. Meissner, G. Adenine nucleotide stimulation of Ca2+-induced Ca2+ release in sarcoplasmic reticulum. J. Biol. Chem. 1984; 259: 2365-2374.
17. Meissner, G, Darling, E, Eveleth, J. Kinetics of rapid Ca2+ release by sarcoplasmic reticulum. Effects of Ca2+, Mg2+, and adenine nucleotides. Biochemistry 1986; 25: 236-244.
18. Laver, DR, Lenz, GK, Lamb, GD. Regulation of the calcium release channel from rabbit skeletal muscle by the nucleotides ATP, AMP, IMP and adenosine. J. Physiol. (Lond.) 2001; 537: 763-778.
19. Godt, RE, Maughan, DW. On the composition of the cytosol of relaxed skeletal muscle of the frog. Am. J. Physiol. 1988; 254: C591-C604.
20. Buri, A, McGuigan, JA. Intracellular free magnesium and its regulation, studied in isolated ferret ventricular muscle with ion-selective microelectrodes. Exp. Physiol. 1990; 75: 751-761.
21. Konishi, M. Cytoplasmic free concentrations of Ca2+ and Mg2+ in skeletal muscle fibers at rest and during contraction. Jpn. J. Physiol. 1998; 48: 421-438.
22. Owen, VJ, Taske, NL, Lamb, GD. Reduced inhibitory effect of Mg2+ on Ca2+ release in porcine muscle fibers with ryanodine receptor mutation for malignant hyperthermia. Am. J. Physiol. 1997; 272: C203-C211.
23. Lamb, GD, Stephenson, DG. Effects of intracellular pH and [Mg2+] on excitation-contraction coupling in skeletal muscle fibres of the rat. J. Physiol. (Lond.) 1994; 478: 331-339.
24. Laver, DR, Baynes, TM, Dulhunty, AF. Magnesium inhibition of ryanodine-receptor calcium channels: evidence for two independent mechanisms. J. Memb. Biol. 1997; 156: 213-229.
25. Laver, DR, Owen, VJ, Junankar, PR, et al. Reduced inhibitory effect of Mg2+ on ryanodine receptor-Ca2+ release channels in malignant hyperthermia. Biophys. J. 1997; 73: 1913-1924.
26. Lamb, GD, Stephenson, DG. Importance of Mg2+ in excitation-contraction coupling in skeletal muscle. NIPS 1992; 7: 270-274.
27. Lamb, GD, Stephenson, DG. Effect of Mg2+ on the control of Ca2+ release in skeletal muscle fibres of the toad. J. Physiol. (Lond.) 1991; 434: 507-528.
28. Laver, DR, O'Neill, ER, Lamb, GD. Luminal Ca2+ regulated Mg2+-inhibition of skeletal RyRs reconstituted as isolated channels or coupled clusters. J. Gen. Physiol. 2004; 124: 741-758.
29. Kermode, H, Williams, AJ, Sitsapesan, R. The interactions of ATP, ADP, and inorganic phosphate with the sheep cardiac ryanodine receptor. Biophys. J. 1998; 74: 1296-1304.
30. Dutka, TL, Lamb, GD. Effect of low cytoplasmic [ATP] on excitation-contraction coupling in fast-twitch muscle fibres of the rat. J. Physiol. (Lond) 2004; 560: 451-468.
31. Dulhunty, AF, Haarmann, CS, Green, D, et al. Interactions between dihydropyridine receptors and ryanodine receptors in striated muscle. Prog. Biophys. Mol. Biol. 2002; 79: 45-75.
32. Tanabe, T, Beam, KG, Adams, BA, Niidome, T, Numa, S. Regions of the skeletal muscle dihydropyridine receptor critical for excitation-contraction coupling. Nature 1990; 346: 567-569.
33. Nakai, J, Tanabe, T, Konno, T, Adams, B, Beam, KG. Localization in the II-III loop of the dihydropyridine receptor of a sequence critical for excitation-contraction coupling. J. Biol. Chem. 1998; 273: 24983-24986.
34. Lu, X, Xu, L, Meissner, G. Activation of the skeletal muscle calcium release channel by a cytoplasmic loop of the dihydropyridine receptor. J. Biol. Chem. 1994; 269: 6511-6516.
35. Dulhunty, AF, Karunasekara, Y, Curtis, SM, et al. The recombinant dihydropyridine receptor II-III loop and partly structured "C" region peptides modify cardiac ryanodine receptor activity. Biochem. J. 2005; 385: 803-813.
36. El-Hayek, R, Antoniu, B, Wang, J, Hamilton, SL, Ikemoto, N. Identification of calcium release-triggering and blocking regions of the II-III loop of the skeletal muscle dihydropyridine receptor. J. Biol. Chem. 1995; 270: 22116-22118.
37. Saiki, Y, Ikemoto, N. Coordination between Ca2+ release and subsequent re-uptake in the sarcoplasmic reticulum. Biochemistry 1999; 38: 3112-3119.
38. Dulhunty, AF, Laver, DR, Gallant, EM, et al. Activation and inhibition of skeletal RyR channels by a part of the skeletal DHPR II-III loop: effects of DHPR Ser687 and FKBP12. Biophys. J. 1999; 77: 189-203.
39. Lamb, GD, El-Hayek, R, Ikemoto, N, Stephenson, DG. Effects of dihydropyridine receptor II-III loop peptides on Ca2+ release in skinned skeletal muscle fibers. Am. J. Physiol. 2000; 279: C891-C905.
40. Stange, M, Tripathy, A, Meissner, G. Two domains in dihydropyridine receptor activate the skeletal muscle Ca2+ release channel. Biophys. J. 2001; 81: 1419-1429.
41. Haarmann, CS, Green, D, Casarotto, MG, Laver, DR, Dulhunty, AF. The random-coil 'C' fragment of the dihydropyridine receptor II-III loop can activate or inhibit native skeletal ryanodine receptors. Biochem. J. 2003; 372: 305-316.
42. Haarmann, CS, Dulhunty, AF, Laver, DR. Regulation of skeletal ryanodine receptors by dihydropyridine receptor II-III loop C-region peptides: Relief of Mg2+-inhibition. Biochem. J. 2005; 387: 429-436.
43. Ritucci, NA, Corbett, AM. Effect of Mg2+ and ATP on depolarization-induced Ca2+ release in isolated triads. Am. J. Physiol. 1995; 269: C85-C95.
44. Laver, DR, Roden, LD, Ahern, GP, et al. Cytoplasmic Ca2+ inhibits the ryanodine receptor from cardiac muscle. J. Memb. Biol. 1995; 147: 7-22.
45. Orlova, EV, Serysheva, II, Van Heel, M, Hamilton, SL, Chiu, W. Two structural configurations of the skeletal muscle calcium release channel. Nature Structural Biology 1996; 3: 547-552.
46. Bhat, MB, Zhao, J, Takeshima, H, Ma, J. Functional calcium release channel formed by the carboxyl-terminal portion of ryanodine receptor. Biophys. J. 1997; 73: 1329-1336.
47. Chen, SR, Ebisawa, K, Li, X, Zhang, L. Molecular identification of the ryanodine receptor Ca2+ sensor. J.Biol.Chem. 1998; 273: 14675-14678.
48. Fessenden, JD, Chen, L, Wang, Y, et al. Ryanodine receptor point mutant E4032A reveals an allosteric interaction with ryanodine. Proc. Natl. Acad. Sci. U.S.A. 2001; 98: 2865-2870.
49. Bhat, MB, Zhao, J, Hayek, S, et al. Deletion of amino acids 1641-2437 from the foot region of skeletal muscle ryanodine receptor alters the conduction properties of the Ca2+ release channel. Biophys. J. 1997; 73: 1320-1328.
50. Bannister, ML, Ikemoto, N. Effects of peptide C corresponding to the Glu724-Pro760 region of the II-III loop of the DHP (dihydropyridine) receptor alpha1 subunit on the domain- switch-mediated activation of RyR1 (ryanodine receptor 1) Ca2+ channels. Biochem. J 2006; 394: 145-152.
51. Leong, P, MacLennan, DH. A 37-amino acid sequence in the skeletal muscle ryanodine receptor interacts with the cytoplasmic loop between domains II and III in the skeletal muscle dihydropyridine receptor. J. Biol. Chem. 1998; 273: 7791-7794.
52. Leong, P, MacLennan, DH. The cytoplasmic loops between domains II and III and domains III and IV in the skeletal muscle dihydropyridine receptor bind to a contiguous site in the skeletal muscle ryanodine receptor. J. Biol. Chem. 1998; 273: 29958-29964.
53. Yamazawa, T, Takeshima, H, Shimuta, M, Iino, M. A region of the ryanodine receptor critical for excitation-contraction coupling in skeletal muscle. J. Biol. Chem. 1997; 272: 8161-8164.
54. Perez, CF, Mukherjee, S, Allen, PD. Amino acids 1-1,680 of ryanodine receptor type 1 hold critical determinants of skeletal type for excitation-contraction coupling. Role of divergence domain D2. J. Biol. Chem. 2003; 278: 39644-39652.
55. Gallant, EM, Hart, J, Eager, K, Curtis, S, Dulhunty, AF. Caffeine sensitivity of native RyR channels from normal and malignant hyperthermic pigs: effects of a DHPR II-III loop peptide. Am. J. Physiol. 2004; 286: C821-C830.
56. Meissner, G, Rousseau, E, Lai, FA, Liu, QY, Anderson, KA. Biochemical characterization of the Ca2+ release channel of skeletal and cardiac sarcoplasmic reticulum. Mol. Cell. Biochem. 1988; 82: 59-65.
57. Fabiato, A. Time and calcium dependence of activation and inactivation of calcium-induced release of calcium from the sarcoplasmic reticulum of a skinned canine cardiac Purkinje cell. J. Gen. Physiol. 1985; 85: 247-289.
58. Stern, MD, Cheng, H. Putting out the fire: what terminates calcium-induced calcium release in cardiac muscle? Cell Calcium 2004; 35: 591-601.
59. Sham, JS, Song, LS, Chen, Y, et al. Termination of Ca2+ release by a local inactivation of ryanodine receptors in cardiac myocytes. Proc. Natl. Acad. Sci. U.S.A. 1998; 95: 15096-15101.
60. Wang, SQ, Stern, MD, Rios, E, Cheng, H. The quantal nature of Ca2+ sparks and in situ operation of the ryanodine receptor array in cardiac cells. Proc. Natl. Acad. Sci. U.S.A. 2004; 101: 3979-3984.
61. Laver, D. The power of single channel recording and analysis: its application to ryanodine receptors in lipid bilayers. Clin. Exp. Pharmacol. Physiol. 2001; 28: 675-686.
62. Marx, SO, Ondrias, K, Marks, AR. Coupled gating between individual skeletal muscle Ca2+ release channels (ryanodine receptors). Science 1998; 281: 818-821.
63. Marx, SO, Gaburjakova, J, Gaburjakova, M, et al. Coupled gating between cardiac calcium release channels (ryanodine receptors). Circ. Res. 2001; 88: 1151-1158.
64. Copello, JA, Porta, M, Diaz-Silvester, P, et al. Coordinated gating of multiple ryanodine receptor channels (RyRs). Biophys. J. 2003; 84: 17a.
65. Porta, M, Diaz-Sylvester, PN, Nani, A, et al. Modulation of coordinated gating of ryanodine receptor (RyR) channels in planar lipid bilayers. Biophys. J. 2004; 86: 241a.
66. Laver, DR. Coupled calcium release channels and their regulation by luminal and cytosolic ions. Eur. Biophys. J. 2005; 34: 359-368.
67. Stern, MD. Buffering of calcium in the vicinity of a channel pore. Cell Calcium 1992; 13: 183-192.
68. Protasi, F, Franzini-Armstrong, C, Flucher, BE. Coordinated incorporation of skeletal muscle dihydropyridine receptors and ryanodine receptors in peripheral couplings of BC3H1 cells. J. Cell. Biol. 1997; 137: 859-870.