1. The Na+/H+ exchanger NHE3 associates with the actin cytoskeleton by binding ezrin both directly and indirectly. Both types of interaction are necessary for acute regulation of NHE3. Most NHE3 acute regulation is by changes in trafficking by effects on exocytosis and for endocytosis. However, NHE3 activity also can be regulated without changing the surface expression of NHE3 (change in turnover number).
2. Direct ezrin binding to NHE3: A positive amino acid cluster in the α-helical juxtamembrane region in the COOH-terminus of NHE3 (aa K516, R520, R527) is necessary for binding to the FERM domain III of ezrin. The direct ezrin binding of NHE3 is necessary for many aspects of basal trafficking, including basal exocytosis, delivery from the synthetic pathway and movement of NHE3 in BB, which probably contributes to endocytosis over a prolonged period.
3. Indirect ezrin binding to NHE3: PDZ domain containing proteins NHERF1 or NHERF2 as intermediates in linking NHE3 to ezrin are necessary for many aspects of NHE3 regulation. NHERF/ERM binding to NHE3 occurs in the cytosolic domain of NHE3 between aa 475-689. This NHERF binding is involved in NHE3 complex formation and restricts NHE3 mobility in the BB. However, it is dynamic, for instance changing in some cases of signaling. Also NHERF binding is necessary for LPA stimulation of NHE3 and inhibition of NHE3 by Ca2+, cAMP and cGMP.
The ERM (ezrin/radixin/moesin) family of proteins serve as regulated cross-linking proteins between the actin cytoskeleton and plasma membrane proteins, and they also participate in regulation of signal transduction pathways.1,2 ERM proteins concentrate in cell-surface structures which are rich in actin cytoskeleton, such as microvilli and filopodia. Their impaired expression or inactivation severely alters cell surface morphology, motility, and adhesion.3
Ezrin has been implicated in several types of diseases. Ezrin is required for bacterial invasion into epithelial or endothelial cells.4,5 Diarrheal diseases caused by enterotoxin-stimulated cAMP require a multiprotein signaling complex containing the Na+/H+ exchanger (NHE3), the Na+/H+ exchanger regulatory factor (NHERF) proteins 1 or 2, ezrin, protein kinase A (PKA), and actin.6,7 Recent studies show that ezrin is upregulated in multiple types of metastatic cancers.8,9
Ezrin is the only ERM protein detected in epithelial cells of the developing intestine and provides an essential role in configuring the mouse intestinal epithelium. Ezrin controls the localization and/or function of certain apical membrane proteins.10
Many multimembrane spanning domain containing proteins associate with the actin cytoskeleton and ezrin using proteins with protein-protein interacting PSD-95/Dlg/ZO-1 (PDZ) domains as intermediates,6,11,12,13,14,15 while single transmembrane proteins, like the cell surface hyaluronate receptor protein (CD43), the cell surface sialoglycoprotein sialophorin (CD44), and intercellular adhesion molecules (ICAM-1, 2, 3), directly interact with ERM proteins. Another model relevant to ezrin binding is NHE1, which is a 12 transmembrane domain protein that associates with the actin cytoskeleton by directly binding to ERM proteins using two positively charged amino acid-rich areas in the juxtamembrane region of its COOH-terminus. This association has structural and functional consequences for the fibroblast cytoskeleton, with NHE1 acting as an anchor in the lamellipodia for actin by binding to ERM proteins.16,17 Disruption of NHE1/ERM binding is associated with abnormal stress fibres and focal adhesions and results in lack of polar migration by fibroblasts in wound healing.16,17,18
The epithelial brush-border (BB) NHE isoform, NHE3, also associates with the actin cytoskeleton by binding with ezrin. We previously showed that NHE3 associates with the actin cytoskeleton with indirect interaction via ezrin by binding to the PDZ domain containing proteins, NHERF1 (EBP50) or NHERF2 (E3KARP). The NHE3 COOH-terminus binds NHERFs between aa 585-689.6,19 The NHERFs simultaneously bind NHE3 and link it to the actin cytoskeleton by also binding to ezrin. NHERF1 and NHERF2 are scaffolding proteins that form homo- and heterodimers, and assemble multiprotein signaling complexes through two PDZ domains and a COOH-terminal ERM binding domain.20,21 NHE3 also can associate with the actin cytoskeleton by binding directly with ezrin through an α-helical juxtamembrane region of the COOH-terminus, which has a positively charged amino acid cluster.22 This binding of a single protein to two molecules of ezrin also occurs with a single transmembrane domain protein, podocalyxin, the major sialoprotein of glomerular epithelial cells.
We review here how NHE3 associates with the actin cytoskeleton, describe the ezrin binding motif in NHE3 and review the roles of direct and indirect interactions between ezrin and NHE3, especially in trafficking and movement in the microvillus.
Specific binding of ERM proteins to juxtamembrane positively charged amino acid clusters of CD44, CD43, and ICAM-2 was confirmed by immunoprecipitation and site-directed mutagenesis. From these findings, it was concluded that ezrin binding domains are characterized as usually having two characteristics: positive amino acid clusters and a high isoelectric point in their juxtamembrane cytoplasmic domain.23
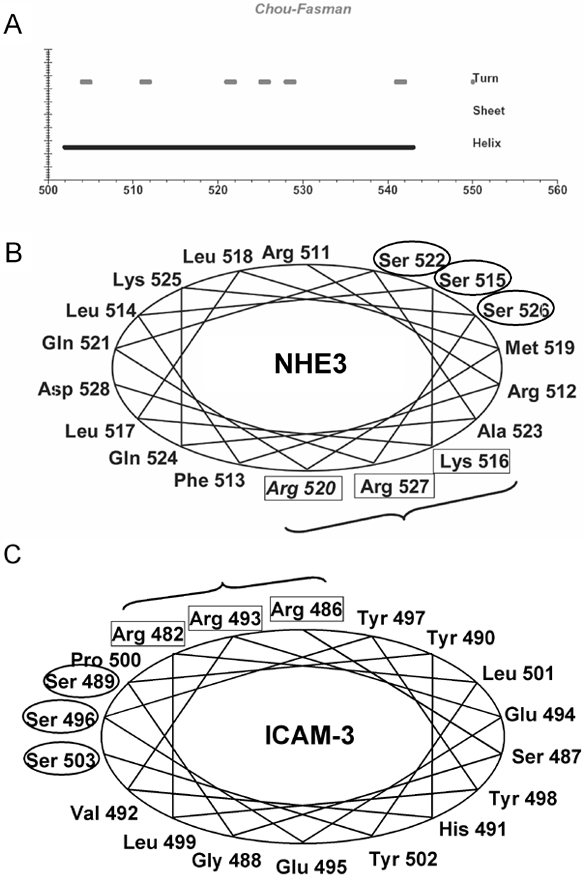
While multiple ERM binding proteins and this type of binding sequence have been identified, we recently contributed that the grouped positively charged amino acids could be presented via an α-helix. We demonstrated this for NHE322 and suggested this is true for other proteins as well. NHE3 has a direct ezrin binding domain in its COOH-terminus, which is predicted (Chou-Fasman algorithm) to form an α-helical structure between residues 502-543. There is a cluster of three positive amino acids (K516, R520 and R527) on one face of the α-helix and it has a high isoelectric point (theoretical pI = 12.1 between aa 511-528; Figure 1A,B). Other ERM binding domains also have a similar α-helical domain at the ERM binding site. ICAM-3 binds ERM at positive amino acids R482, R486, and R493 (Figure 1C; boxed amino acids) which are clustered at one side of a putative α-helix. ICAM-3 also has a serine cluster S489, S496, and S503 (Figure 1C; circled amino acids) on the opposite side of the α-helix. This serine-rich motif in ICAM3 was critical for its ERM-directed subcellular targeting.24 Similarly, NHE3 has a serine-rich cluster (S515, S522, and S526 (Figure. 1B; circled amino acids) near the ezrin binding positive amino acid cluster. The role of this serine cluster in ezrin in NHE3 function or binding to ezrin is unknown.

Figure 1. α-Helical juxtamembrane domain of the NHE3 C-terminus contains a positively charged amino acid cluster with high isoelectric point that is involved in ezrin binding. A: Secondary structure predictions of NHE3 juxtamembrane region of the COOH-terminus (Gene Runner; Chou-Fasman analysis). This area of the NHE3 contains a long α-helical rich domain, has some turns but no β-sheet structure (thick lines are predicted regions of stated domains). B: Since ezrin binding is often to an α-helical area and to positive amino acid clusters, this NHE3 α-helical domain was examined for positive amino acid clusters. K516, R520, and R527 (boxed amino acids) form a positive amino acid cluster on one side of a putative α-helix between aa 511-528. The calculated isoelectric point of this putative α-helix was 12.1. Isoelectric points were calculated using a program from Gene Runner software. NHE3 has a serine cluster near the ezrin binding positive amino acid cluster (circled serines; S515, S522, S525). C: Like NHE3, ICAM-3 has a positively charged amino acid cluster (boxed amino acids; R482, R486, and R493) in the juxtamembrane region of its C-terminus, in an area predicted to be α-helical (Gene Runner; Chou-Fasman analysis). It has also a serine cluster near the ezrin binding cluster (circled serines; S489, S496, and S503).
NHE1 associates with the actin cytoskeleton by direct binding to ezrin, although unlike NHE3, it has only a single direct interaction with ERM proteins. The COOH-terminal juxtamembrane region of NHE1 was predicted to be α-helical by secondary structure programs (by GeneRunner; Chou-Fasman). Furthermore, the crystal structure of the NHE1 juxtamembrane domain where CHP binds (aa 503–545), which includes the region of ezrin binding, was solved and shown to contain an α-helical domain.25 The ezrin binding domain (aa 553-564) of NHE1, identified by Barber and collaborators16 has a positive amino acid cluster (H545, K549, R552, and K556) in an α-helical domain (by helical wheel analysis via GeneRunner) and a high theoretical isoelectric point (pI = 10.7 from region of aa 553-564). NHE1 also interacts with ERM proteins, which tethers NHE1 to cortical actin cytoskeleton to regulate cell shape, adhesion and motility. NHE1 defends against apoptotic stress by physical association with the actin cytoskeleton by binding activated ERM proteins, resulting in activation of the pro-survival kinase, Akt.26 This result shows that NHE1-regulated ERM protein binding is critical for the PI3-kinase/Akt signaling pathway dependent cell survival.
Another ezrin binding protein is podocalyxin, which is a single-transmembrane protein and is the major sialoglycoprotein of glomerular epithelial cells. It is known to directly bind to ezrin through its COOH-terminal juxtamembrane region14 and also binds ezrin indirectly, through NHERF1 or NHERF2.15 Sequence alignment revealed that podocalyxin possesses a sequence, 411HQRIS, in the juxtamembrane region that is similar to the 484HQRS sequence found in intercellular adhesion molecule-3 (ICAM-3).24 The direct ezrin binding domain of podocalyxin involves its last 18 aa, between aa 405 and 422.14 Chou-Fasman analysis showed that the ezrin binding juxtamembrane region of podocalyxin, including aa 405-422, is an α-helical domain. Helical wheel analysis showed that it has a positive amino acid cluster and a high isoelectric point (pI = 11.8 of aa 411-422). Thus, as with NHE3, secondary structure analysis of ezrin ligand binding sites indicates that the clustered positive charges for ezrin binding can be arranged in an α-helical as well as in a linear configuration. Consequently, both linear and α-helical secondary structures must be considered when searching for ezrin binding sites. Moreover, clustered positively charged amino acids are not sufficient for ezrin binding (the NHE3 COOH-terminal positive amino acid cluster, 607RRR, does not bind ezrin). A high isoelectric point also appears to be necessary.
Importantly, whether positively charged residues are involved in direct ezrin contact is not established. Indeed, in crystal structure to 2.4 Å of the radixin FERM (band 4.1 protein and ERM homology) domain with the cytoplasmic domain of ICAM-2, binding involves a β-sheet peptide of ICAM-2. Whereas positively charged residues are involved in this binding, they flank but are not in the binding domain, and their suggested role is in interacting with membrane phosphatidylinositol bisphosphate.27
Many membrane proteins interact with ezrin directly or indirectly, the latter often via PDZ domain containing proteins, including but not limited to NHERF1 or NHERF2 (Figure 2). In addition, ezrin also has direct and indirect association with several cytosolic proteins (Figure 2).
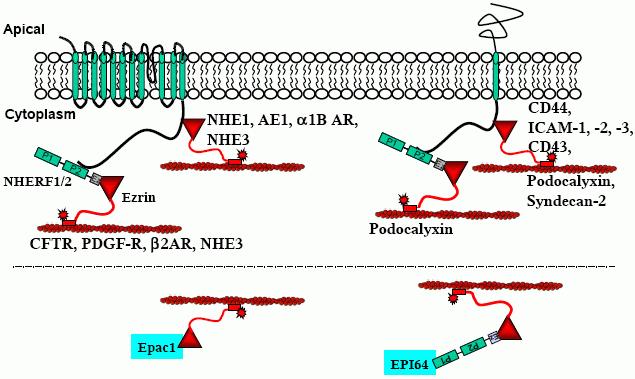
Figure 2. Two different types of ezrin interactions with proteins leads to actin cytoskeleton association. Many single transmembrane proteins such as CD44, ICAM-1,-2,-3, CD43, and syndecan associate with the actin cytoskeleton by directly interacting with ezrin. A few, such as podocalyxin, also bind indirectly to ezrin. Many multiple transmembrane proteins associate with the actin cytoskeleton by indirectly interacting with ezrin by PDZ domain containing proteins as intermediates. Examples include CFTR, PDGF-R, β2AR, and NHE3. Some multi-membrane spanning domain proteins interact directly with ezrin. These include NHE1, AE1, α1b-AR, and NHE3. Several non-transmembrane proteins can associate with the actin cytoskeleton via ezrin. Epac1 directly binds to ezrin and EPI64 indirectly binds to ezrin via NHERF1.
Membrane proteins can associate indirectly with ezrin via PDZ domain containing proteins. Indirect associations have been described with the β2-adrenergic receptor (β2-AR), cystic fibrosis transmembrane regulator (CFTR), podocalyxin, platelet-derived growth factor receptor (PDGFR) and NHE3 through several PDZ domain containing adaptors, NHERF1, NHERF2, and synapse-associated protein 97 (SAP97).11,12,13,14,15,28,22
G protein-coupled receptors (GPCRs) can act as scaffolds binding a variety of proteins involved in receptor regulation and signaling. An indirect interaction between ERM proteins and GPCRs has been suggested by previous findings showing that some GPCRs can interact with ERM proteins with NHERF1 as an intermediate.29,30,31 This interaction seems to play a role in GPCR trafficking. Recent studies showed that ezrin can also directly interact with the α1b-AR through a positive amino acid cluster located on the juxtamembrane region of COOH-terminus of α1b-AR and this interaction contributes to receptor recycling to the plasma membrane.32
The cytoplasmic tail of podocalyxin contains a COOH-terminal PDZ binding motif (482DTHL) that binds to NHERF1 or NHERF2.15,14 Podocalyxin is linked to ezrin and the actin cytoskeleton via NHERF-2 and the podocalyxin/NHERF-2/ezrin multiple-protein complex is localized along the apical domain of the podocyte plasma membrane.15,33 The multiple protein signaling complex including, podocalyxin, NHERF-2, ezrin, and actin are disrupted in podocytes from puromycin aminonucleoside (PAN), protamine sulfate, or sialidase-treated rats, conditions that result in a dramatic loss of foot processes and a marked increase in urinary protein excretion, comparable to that seen in nephrotic syndrome.15 PDZ-binding domains are therefore also critical in maintaining the glomerular cell integrity and permselectivity and avoiding glomerular protein leak.
The FERM domain of ERM proteins bind directly to multiple membrane proteins, thereby linking the actin cytoskeleton to the plasma membrane. At the membrane, the ERM proteins can directly interact with the juxtamembrane cytoplasmic region of a variety of single spanning transmembrane domain proteins, such as CD44, ICAM-1, -2, -3, and CD43,34,23,35 and multiple spanning transmembrane domain proteins, such as AE1, NHE1, and α1b-AR.36,17,32 This occurs via a series of basic amino acids clusters on the juxtamembrane cytoplasmic region of these proteins.23 Recently it was demonstrated that ezrin can directly interact with the α1b-AR through a poly-arginine motif located on the COOH-terminal tail of the receptor and that this interaction contributes to receptor recycling to the plasma membrane.32
NHE1 scaffolding to the basolateral membrane of polarized epithelial cells occurs independently of PDZ domain interactions. NHE1 does not bind NHERFs and the COOH-terminal cytoplasmic domain of NHE1 lacks canonical binding sites for PDZ domains. In contrast, NHE1 associates directly with ezrin, thereby scaffolding NHE1 to the plasma membrane by associating with the actin cytoskeleton. The anion exchanger AE1 is also structurally linked to the actin cytoskeleton. The band 4.1 protein (an ERM family protein) binds to AE1 at a motif of clustered positively charged amino acids in the juxtamembrane region of the COOH-domain.36 A similar ERM binding motif is present in CD4437 and is also found in NHE1.16 The NHE1-ERM association has a role similar to AE1 in regulating cell shape that can be understood based on the ability of ERM proteins to tether cortical actin filaments to the plasma membrane. The NHE1-ezrin direct interaction is a key determinant in the induction of stress fibres in response to activation of the small GTPase RhoA by lysophosphatidic acid (LPA). It is likely that RhoA and the RhoA-kinase ROCK regulate the association of NHE1 and ERM proteins.16,38
Most well known ezrin binding of non-transmembrane proteins involve the PDZ domain containing proteins, NHERF1 or NHERF2. They have two PDZ domains and have an ERM binding domain in the last 30 amino acids of the COOH-terminus.39,6 Similarly to the ezrin binding domain of membrane proteins, the ERM binding domain of NHERF1 or NHERF2 contains a positive amino acids cluster (K331, H335, R338, R342 in NHERF1; K310, R314, R317, R321 in NHERF2; unpublished data by Cha and Donowitz) located in the last 30 amino acids of the COOH-terminus and are critical for ezrin binding. Recently, Cha et al.22 showed that the FERM domain III of N-terminal ezrin binds to NHERF1 or NHERF2. The interaction of ezrin with NHERF proteins occurs via an ezrin N-terminal membrane targeting site that binds cargo molecules, whereas the COOH-terminus of ezrin contains an F-actin binding site.
Other multiple ezrin-interacting proteins have been reported. The phosphoinositide-3 kinase (PI3-kinase) p85 subunit binds to ezrin at two different binding sites: the N-terminal domain and pY353, which binds to the c-Src homology 2 (SH2) domain of p85.13 Also, the Drosophila ERM-related protein coracle, which is present in epithelial cells and helps organize the septate junction, requires binding to its ligands with both its N-terminal (FERM domain) and its non-actin binding COOH-terminus for normal septate junction formation.40 The increasing recognition of more than one ezrin molecule or multiple sites on one ezrin molecule binding to a single partner is consistent with ezrin existing as a dimer2,41 and suggests this pattern should be sought to determine how frequently it is involved in ERM function.
Recent studies have shown that Epac1 (exchange protein directly activated by cAMP) interacts with ezrin through its N-terminal 49 amino acids.42 Interestingly, this N-terminal 49 amino acids of Epac1 contains a positive amino acids cluster, 20HQRPS, which is an ezrin binding motif similar to 411HQRIS in podocalyxin14 and 484HQRS in ICAM-3.24 In addition, this putative ezrin binding region in Epac1 has a high isoelectric point (pI = 11.5).
Another non-transmembrane protein, EPI64 (EBO50-PDZ interactor of 64 kD) is known to indirectly interact with ezrin via NHERF1. Bretscher and his collaborators showed that EPI64, a 508 aa cytosolic protein extracted from placenta microvilli that contains a TBC (Tre-2/Bub2/Cdc16) domain, indirectly interacts with ezrin by binding NHERF1.43 EPI64 ends in the amino acid sequence DTYL, which binds to the first PDZ domain of NHERF1.44
We have shown that direct binding of NHE3 to ezrin is necessary for NHE3 trafficking and mobility in the BB. Although NHE1/direct ezrin binding has been shown to be important for organizing the cytoskeleton,16,17,45 there is no evidence yet for a role of NHE3/direct ezrin binding in similar organization of either the lamellipodia of fibroblasts or the epithelial cell (OK cell) apical cytoskeleton, although this has not been systematically examined.
Based on physiological studies, NHE3 associates with the actin cytoskeleton both by binding to and independently of NHERF1 and NHERF2. We have shown that NHE3 directly binds ezrin at a site in its COOH-terminus between aa 475-589, which is separate from the PDZ interacting domain.6 This is an area predicted to be α-helical, with a positive aa cluster on one side of the α-helix (K516, R520, and R527). Point mutations of these positively charged amino acids abolished ezrin binding. The functional consequences of these NHE3 ezrin binding mutants include: 1) A marked decrease in surface expression with a greater decrease in NHE3 activity, indicating a dependence of turnover number as well as trafficking on direct ezrin binding; 2) Decreased surface expression due to decreased rates of exocytosis and plasma membrane delivery of newly synthesized NHE3, with normal total expression levels and slightly reduced endocytosis rates; 3) A longer plasma membrane half-life of mutant NHE3 with normal total half-life; 4) Decreased NHE3 BB mobile fraction. Therefore the functional role of the direct interaction of ezrin with NHE3 is related to the exocytic trafficking of and plasma membrane delivery of newly synthesized NHE3, which determines the amount of plasma membrane NHE3 and partially determines NHE3 activity and BB mobility, the latter of which probably increases NHE3 delivery from microvilli to the intervillus clefts, and may be important for NHE3-regulated endocytosis.
The indirect association of ezrin with NHE3 controls the macromolecular complexes formed between NHERF1/2 and NHE3. NHERF1 and NHERF2 are docking proteins that assemble multi-protein signaling complexes, including NHE3, ezrin, and protein kinase A II (PKAII), through their multiple PDZ domains. These multi-protein signaling complexes facilitate the phosphorylation of NHE3 by PKA, which is necessary for cAMP mediated inhibition of NHE3 activity.6,7 Here the interaction between NHERF proteins and ezrin is critical for cAMP mediated inhibition of NHE3 activity. The ezrin binding domain deletion mutant NHERF1Δ307 or NHERF2Δ3046 abolished cAMP mediated inhibition of NHE3 activity. In the cGMP inhibition of BB NHE3, the complex of NHE3, NHERF2, and cyclic GMP kinase II (cGKII) is necessary for cGMP mediated NHE3 inhibition. In this case, NHERF2 directly binds cGKII in the BB to form an NHE3 complex, with cGKII also associating with the BB via its myristoylation. However, unlike cAMP mediated inhibition of NHE3, cGMP mediated inhibition of NHE3 does not require ezrin indirect interaction through NHERF2 since the cGMP mediated inhibition of NHE3 was not abolished by the ezrin binding domain deletion mutant, NHERF2 Δ30.47
Another functional consequence of this NHERF binding to NHE3 also includes Ca2+ inhibition of NHE3. NHERF2 has been demonstrated to be involved with NHE3 inhibition by elevated intracellular calcium by a mechanism that involves both complex formation and changes in trafficking. NHERF2 but not NHERF1 reconstitutes Ca2+ inhibition of NHE3.48,49,50 In this effect, NHERF2 scaffolds a complex involving NHE3, ezrin, α-actinin-4 and protein kinase Ca (PKCα). The ezrin binding domain deletion mutant NHERF2Δ30 abolished Ca2+ mediated inhibition of NHE3 activity (Cha and Donowitz, unpublished data). This result shows that Ca2+inhibition of NHE3 requires ezrin indirect interaction through NHERF2 specifically, but not NHERF1, with the likely explanation being that NHERF2 but not NHERF1 binds α-actinin-4, and actinin-4 is needed for Ca2+ inhibition of NHE3. Both NHERF1 and NHERF2 bind PKCα.48,51
The functional consequences of ezrin indirect association via NHERF binding also include limitating the mobility of NHE3 in epithelial cell BB. The role of indirect ezrin binding via PDZ domain containing proteins in lateral mobility on the plasma membrane has been studied in NHE3. The work of Cha and colleagues showed that the truncation mutant (NHE3585; truncated to aa 1-585) or the point mutant (NHE3 F2LM; unpublished data by Cha and Donowitz), which prevents NHERF1/2 binding, increase the lateral mobility of NHE3, and that the overexpression of NHERF2 significantly decreased the lateral mobility of NHE3. Based on these results we conclude that NHERF1/2 play a role as anchoring proteins which immobilize NHE3 on the plasma membrane.52 Similarly, Haggie et al. showed that lateral mobility of CFTR was increased at the plasma membrane after deletion of its C-terminal PDZ binding motif.53 This means that indirect ezrin association via NHERF proteins limits CFTR and NHE3 movement in the BB, most likely through an anchoring mechanism.
Ezrin activation is necessary to associate with binding partners, including membrane proteins. Cytosolic inactive ezrin does not interact with membrane proteins. There are three types of recognized ezrin activation signaling pathways relevant to NHE3 regulation (Figure 3).
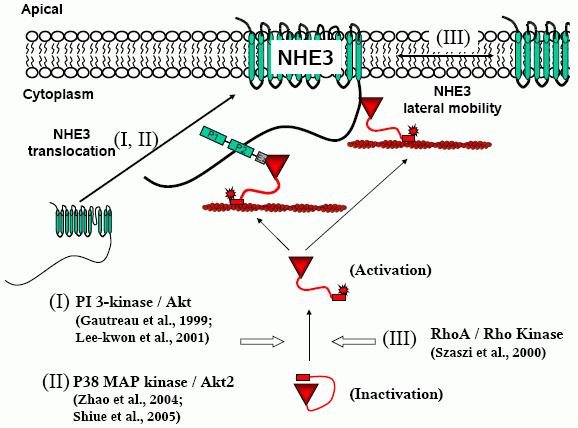
Figure 3. Ezrin activation signal pathways in NHE3 translocation. Ezrin activation in NHE3 regulation involves three different pathways, PI3-kinase/Akt, p38 MAPK/Akt2, and RhoA/Rho kinase pathways. PI3-kinase/Akt and p38 MAPK/Akt2 are involved in NHE3 translocation to the apical plasma membrane and Rho/Rho kinase limits NHE3 lateral mobility in the plasma membrane.
PI3-kinase activity is necessary for basal NHE3 activity in the polarized epithelial cell lines Caco-2 and OK, as well as in the fibroblast cell lines PS120 and in AP-1 cells. PI3-kinase is also necessary for epidermal growth factor (EGF)/fibroblast growth factor (FGF) stimulation of NHE3 in fibroblasts and EGF stimulation of NHE3 in ileal brush border. PI3-kinase inhibitors wortmannin or LY294002 lowered NHE3 basal activity by 50% and also lowered FGF stimulated NHE3 in PS120 cells by 50%. Thus, EGF/FGF stimulate NHE3 by both PI3-kinase dependent and PI3-kinase independent mechanisms.54,55,56
ERM proteins are regulated by tyrosine phosphorylation in response to EGF, platelet derived growth factor (PDGF) and hepatocyte growth factor (HGF). In ezrin, two tyrosine residues, corresponding to amino acids Tyr-145 and Tyr-353, are phosphorylated by the EGF receptor.57 Ezrin interacts with p85, the regulatory subunit of PI3-kinase. Two distinct sites of ezrin are involved in this interaction, the NH2-terminal domain aa 1-309 and the phosphorylated Tyr-353 residue, which binds to the COOH-terminal SH2 domain of p85. A point mutation in ezrin, Y535F, induces cell apoptosis by impairing the activation of the serine/threonine protein kinase Akt (PKB), a downstream effector of PI3-kinase. These results show a novel function for ezrin in determining survival of epithelial cells by activating the PI3-kinase/Akt pathway.58
Addition of luminal D-glucose in intestinal absorptive cells acts via a Na+-dependent glucose cotransporter (SGLT1) to cause NHE3 to be translocated to the apical plasma membrane and leads to cytoplasmic alkalization. Turner and his collaborators showed that ezrin activation is required for this NHE3 translocation to the apical plasma membrane.59,60 They showed that ezrin activation was a consequence of linkage of SGLT1 to stimulation of a p38 mitogen-activated protein kinase (MAPK) pathway acting via MAPK-activated protein kinase 2 (MAPKAPK-2), Akt2 and ezrin, but was independent of Rho kinase.61,62 Additionally, they showed that inhibition of Rho kinase did not inhibit ezrin phosphorylation at T567. Inhibition of p38 MAP kinase prevented Akt activation and blocked ezrin phosphorylation. MAPKAPK-2 phosphorylates an Akt2-derived peptide in vitro. siRNA-mediated MAPKAPK-2 knockdown inhibited phosphorylation of Akt2 and ezrin but not p38 MAPK. MAPKAPK-2 knockdown also blocked NHE3 translocation. Thus, MAPKAPK-2 links p38 MAPK to Akt2, ezrin, and NHE3 activation after SGLT1-mediated transport.63
Cytoplasm-to-plasma membrane translocation of ERM proteins is partially under the control of the small GTPase Rho. Rho-dependent formation of microvilli-like structures requires ERM proteins.64 ERM proteins are phosphorylated at a C-terminal threonine by Rho kinase (ROCK), and this phosphorylation interferes with their head-to-tail interaction.65 The phosphorylation of moesin T558 by ROCK in vivo plays a necessary role in the formation of microvilli-like structures.64 In addition, binding of GDP-dissociation inhibitor Rho-GDI to a cryptic site in the N-terminal domain of ERM activates Rho by releasing Rho-GDI inhibition.66
Mobility of NHE3 on the apical plasma membrane in epithelial cells is decreased by Rho GTPases.67 Tetanus toxin B (TxB) impairs Rho GTPase function. When compared with control cells, TxB-treated cells showed markedly increased mobility of NHE3 and suggested that NHE3 mobility on the apical plasma membrane is dependent on Rho-dependent ezrin activation. In contrast, NHE3 mobility was not changed by the PI 3-kinase inhibitor LY294002.22 In fibroblasts and OK cells, wortmannin or LY294002 reduced the basal transport activity and the surface amount of NHE3 by ∼50% which was shown to be due to inhibition of the exocytosis of the exchanger back to the plasma membrane.68,54,55 These results suggest that Rho/ROCK dependent but not PI3-Kinase/Akt dependent ezrin activation regulates the lateral mobility of NHE3 on the apical plasma membrane.
We showed that ezrin binds to NHE3 at two sites. At one site it binds indirectly via the NHERF family of PDZ domain-containing proteins, NHERF1 or NHERF2, through their COOH-terminal ERM binding domain and at the second site by direct binding in an α-helical area in the juxtamembrane domain of NHE3, involving a positive amino acid cluster containing K516, R520, and R527. This is the first direct evidence that the NHE3 C-terminus exists as an α-helix.
The same ezrin domain (FERM III) is used in both types of binding to NHE3, which indicates the involvement of two separate ezrin molecules with a single NHE3 molecule. This concept seems to apply to ezrin interactions with multiple ligands. How ezrin interacts with its partners has recently been partially clarified. There are two patterns of ezrin interactions, direct binding and indirect binding via NHERF interactions. Substrates that only directly bind ezrin include single spanning transmembrane domain proteins (ICAM-1, -2, and -3, CD44 and CD43) and multiple spanning transmembrane domain proteins (AE1, NHE1, and α1b-AR). Indirect ezrin association has been described with the multiple transmembrane domain proteins, including β2-AR, CFTR, PDGFR, and NHE3 as well as the single transmembrane domain protein, podocalyxin. NHE3 and podocalyxin bind ezrin both directly and indirectly via NHERF1 or NHERF2.6,14,15,22 It appears that many transmembrane proteins associate with the actin cytoskeleton by interacting with ezrin both directly via juxtamembrane ezrin binding of their cytoplasmic COOH-terminus and indirectly through cytoplasmic scaffolding PDZ domain-containing proteins, particularly NHERF1, NHERF2, or SAP97.
There are also several non-transmembrane proteins that directly interact with ezrin or indirectly interact with ezrin via NHERF proteins. Ezrin not only links plasma membrane proteins and the actin cytoskeleton but also links cytosolic proteins with the actin cytoskeleton.
NHE3 is regulated by ezrin activation by three different pathways: Rho/ROCK,69 PI 3-kinase/Akt,70 and p38 MAP kinase/Akt pathways.60 P38 MAP kinase/Akt and PI 3-kinase/Akt pathway regulate NHE3 trafficking and regulate the NHE3 activity by changing the surface amount of NHE3 related to LPA, EGF or D-glucose.70,50,60 However, the Rho/ROCK pathway regulates functional activity of NHE3 at the plasma membrane without changing surface amount in fibroblasts,67,69 while it increases surface NHE3 amount in polarized epithelial cells.71
Our studies of NHE3 have identified and characterized the functional consequences of the two types of ezrin interaction. The direct ezrin binding domain of NHE3 involves a positively charged amino acid cluster in an area of high isoelectric point. Our studies indicated that these positively charged amino acid clusters can be arranged in either a linear or an α-helical conformation. The functional consequences of the two types of interaction with ezrin affect multiple functional aspects of NHE3. However, both are necessary for acute changes in NHE3 activity which are a critical component of the major functions of NHE3. It will be important to characterize the functions of the direct and indirect binding of ezrin to NHE3 and other proteins to determine whether there are any characteristic functions associated with direct vs indirect ezrin binding to membrane proteins.
We acknowledge the expert editorial assistance of Ms. H. McCann. Supported in part by the National Institutes of Health, NIDDK Grants RO1-DK26523, RO1-DK061765, PO1-DK072084, and R24-DK64388 (The Hopkins Basic Research Digestive Diseases Development Core Center), and the Hopkins Center for Epithelial Disorders.
1. Fievet B, Louvard D, Arpin M. ERM proteins in epithelial cell organization and functions. Biochim. Biophys. Acta 2007; 1773: 653-60.
2. Bretscher A, Edwards K, Fehon RG. ERM proteins and merlin: Integrators at the cell cortex. Nat. Rev. Mol. Cell Biol. 2002; 3: 586-99.
3. Bretscher A, Chambers D, Nguyen R, Reczek D. ERM-merlin and EBP50 protein families in plasma membrane organization and function. Annu. Rev. Cell Dev. Biol. 2000; 16: 113-43.
4. Skoudy A, Van Nhieu GT, Mantis N et al. A functional role for ezrin during Shigella flexneri entry into epithelial cells. J. Cell Sci. 1999; 112: 2059-68.
5. Eugene E, Hoffmann I, Pujol C, Couraud PO, Bourdoulous S, Nassif X. Microvilli-like structures are associated with the internalization of virulent capsulated Neisseria meningitidis into vascular endothelial cells. J. Cell Sci. 2002; 115: 1231-41.
6. Yun CHC, Lamprecht G, Forster DV, Sider A. NHE3 kinase A regulatory protein E3KARP binds the epithelial brush border Na+/H+ exchanger NHE3 and the cytoskeletal protein ezrin. J. Biol. Chem. 1998; 273: 25856-63.
7. Weinman EJ, Steplock D, Donowitz M, Shenolikar S. NHERF associations with sodium-hydrogen exchanger isoform 3 (NHE3) and ezrin are essential for cAMP-mediated phosphorylation and inhibition of NHE3. Biochemistry 2000; 39: 6123-29.
8. Khanna C, Wan XL, Bose S et al. The membrane-cytoskeleton linker ezrin is necessary for osteosarcoma metastasis. Nat. Med. 2004; 10: 182-86.
9. Hunter KW. Ezrin, a key component in tumor metastasis. Trends Mol. Med. 2004; 10: 201-4.
10. Saotome I, Curto M, McClatchey AI. Ezrin is essential for epithelial organization and villus morphogenesis in the developing intestine. Dev. Cell 2004; 6: 855-64.
11. Hall RA, Premont RT, Chow CW et al. The β2-adrenergic receptor interacts with the Na+/H+-exchanger regulatory factor to control Na+/H+ exchange. Nature 1998; 392: 626-30.
12. Wang SS, Raab RW, Schatz PJ, Guggino WB, Li M. Peptide binding consensus of the NHE-RF-PDZ1 domain matches the C-terminal sequence of cystic fibrosis transmembrane conductance regulator (CFTR). Febs Lett. 1998; 427: 103-8.
13. Sun F, Hug MJ, Lewarchik CM, Yun CHC, Bradbury NA, Frizzell RA. E3KARP mediates the association of ezrin and protein kinase A with the cystic fibrosis transmembrane conductance regulator in airway cells. J. Biol. Chem. 2000; 275: 29539-46.
14. Schmieder S, Nagai M, Orlando RA, Takeda T, Farquhar MG. Podocalyxin activates RhoA and induces actin reorganization through NHERF1 and ezrin in MDCK cells. J. Am. Soc. Nephrol. 2004; 15: 2289-98.
15. Takeda T, McQuistan T, Orlando RA, Farquhar MG. Loss of glomerular foot processes is associated with uncoupling of podocalyxin from the actin cytoskeleton. J. Clin. Invest. 2001; 108: 289-301.
16. Denker SP, Huang DC, Orlowski J, Furthmayr H, Barber DL. Direct binding of the Na-H exchanger NHE1 to ERM proteins regulates the cortical cytoskeleton and cell shape independently of H+ translocation. Mol. Cell 2000; 6: 1425-36.
17. SP, Barber DL. Cell migration requires both ion translocation and cytoskeletal anchoring by the Na-H exchanger NHE1. J. Cell Biol. 2002; 159: 1087-96.
18. Baumgartner M, Sillman AL, Blackwood EM et al. The Nck-interactina kinase ohowhorviates ERM proteins for formation of lamellipodium by growth factors. Proc. Natl. Acad. Sci. U.S.A. 2006; 103: 13391-13396.
19. Yun CHC, Oh SY, Zizak M et al. cAMP-mediated inhibition of the epithelial brush border Na+/H+ exchanger, NHe3, requires an associated regulatory protein. [published erratum appears in Proc. Natl. Acad. Sci. U.S.A. 1997; 94: 10006] Proc. Natl. Acad. Sci. U.S.A. 1997; 94: 3010-15.
20. Shenolikar S, Voltz JW, Cunningham R, Weinman EJ. Regulation of ion transport by the NHERF family of PDZ proteins. Physiology 2004; 19: 362-69.
21. Donowitz M, Cha B, Zachos NC et al. NHERF family and NHE3 regulation. [published erratum appears in: J. Physiol. 2005; 567: 1079] J. Physiol. 2005; 567: 3-11.
22. Cha B, Tse M, Yun C et al. The NHE3 juxtamembrane cytoplasmic domain directly binds ezrin: Dual role in NHE3 trafficking and mobility in the brush border. Mol. Biol. Cell 2006; 17: 2661-73.
23. Yonemura S, Hirao M, Doi Y et al. Ezrin/radixin/moesin (ERM) proteins bind to a positively charged amino acid cluster in the juxta-membrane cytoplasmic domain of CD44, CD43, and ICAM-2. J. Cell Biol. 1998; 140: 885-95.
24. Serrador JM, Vicente-Manzanares M, Calvo J et al. A novel serine-rich motif in the intercellular adhesion molecule 3 is critical for its ezrin/radixin/moesin-directed subcellular targeting. J. Biol. Chem. 2002; 277: 10400-409.
25. Ben Ammar Y, Takeda S, Hisamitsu T, Mori H, Wakabayashi S. Crystal structure of CHP2 complexed with NHE1-cytosolic region and an implication for pH regulation. EMBO J. 2006; 25: 2315-25.
26. Wu KL, Khan S, Lakhe-Reddy S et al. The NHE1 Na+/H+ exchanger recruits ezrin/radixin/moesin proteins to regulate Akt-dependent cell survival. J. Biol. Chem. 2004; 279: 26280-86.
27. Hamada K, Shimizu T, Yonemura S, Tsukita S, Tsukita S, Hakoshima T. Structural basis of adhesion-molecule recognition by ERM proteins revealed by the crystal structure of the radixin-ICAM-2 complex. EMBO J. 2003; 22: 502-14.
28. Bonilha VL, Rodriguez-Boulan E. Polarity and developmental regulation of two PDZ proteins in the retinal pigment epithelium. Invest. Ophth. Vis. Sci. 2001; 42: 3274-82.
29. Weinman EJ, Cunningham R, Shenolikar S. NHERF and regulation of the renal sodium-hydrogen exchanger NHE3. Pflugers Arch. 2005; 450: 137-44.
30. Cao TT, Deacon HW, Reczek D, Bretscher A, von Zastrow M. A kinase-regulated PDZ-domain interaction controls endocytic sorting of the β2-adrenergic receptor. Nature 1999; 401: 286-90.
31. Li JG, Chen CG, Liu-Chen LY. Ezrin-radixin-moesin-binding phosphoprotein-50/Na+/H+ exchanger regulatory factor (EBP50/NHERF) blocks U50,488H-induced down-regulation of the human κ opioid receptor by enhancing its recycling rate. J. Biol. Chem. 2002; 277: 27545-52.
32. Stanasila L, Abuin L, Diviani D, Cotecchia S. Ezrin directly interacts with the α1b-adrenergic receptor and plays a role in receptor recycling. J. Biol. Chem. 2006; 281: 4354-63.
33. Orlando RA, Takeda T, Zak B et al. The glomerular epithelial cell anti-adhesin podocalyxin associates with the actin cytoskeleton through interactions with ezrin. J. Am. Soc. Nephrol. 2001; 12: 1589-98.
34. Hirao M, Sato N, Kondo T et al. Regulation mechanism of ERM (ezrin/radixin/moesin) protein/plasma membrane association: Possible involvement of phosphatidylinositol turnover and Rho-dependent signaling pathway. J. Cell Biol. 1996; 135: 37-51.
35. Heiska L, Alfthan K, Gronholm M, Vilja P, Vaheri A, Carpen O. Association of ezrin with intercellular adhesion molecule-1 and -2 (ICAM-1 and ICAM-2). Regulation by phosphatidylinositol 4, 5-bisphosphate. J. Biol. Chem. 1998; 273: 21893-900.
36. Jons T, Drenckhahn D. Identification of the binding interface involved in linkage of cytoskeletal protein 4.1 to the erythrocyte anion-exchanger. EMBO J. 1992; 11: 2863-67.
37. Legg JW, Isacke CM. Identification and functional analysis of the ezrin-binding site in the hyaluronan receptor, CD44. Curr. Biol. 1998; 8: 705-708.
38. Tominaga T, Barber DL. Na-H exchange acts downstream of RhoA to regulate integrin-induced cell adhesion and spreading. Mol. Biol. Cell 1998; 9: 2287-303.
39. Reczek D, Berryman M, Bretscher A. Identification of EBP50: A PDZ-containing phosphoprotein that associates with members of the ezrin-radoxin-moesin family. J. Biol. Chem.1997; 139: 169-79.
40. Ward RE, Schweizer L, Lamb RS, Fehon RG. The protein 4.1, ezrin, radixin, moesin (FERM) domain of drosophila Coracle, a cytoplasmic component of the septate junction, provides functions essential for embryonic development and imaginal cell proliferation. Genetics 2001; 159: 219-28.
41. Chambers DN, Bretscher A. Ezrin mutants affecting dimerization and activation. Biochemistry 2005; 44: 3926-32.
42. Zhao J. Subcellular localisation of Epac [dissertation]. Utrecht: Utrecht University; 2006. http://igitur-archive.library.uu.nl/dissertations/2006-0912-202142/
43. Hanono A, Garbett D, Reczek D, Chambers DN, Bretscher A. EPI64 regulates microvillar subdomains and structure. J. Cell Biol. 2006; 175: 803-13.
44. Reczek D, Bretscher A. Identification of EPI64, a TBC/rabGAP domain-containing microvillar protein that binds to the first PDZ domain of EBP50 and E3KARP. J. Cell Biol. 2001; 153: 191-205.
45. Baumgartner M, Patel H, Barber DL. Na+/H+ exchanger NHE1 as plasma membrane scaffold in the assembly of signaling complexes. Am. J. Physiol. Cell Physiol. 2004; 287: C844-50.
46. Cha BY, Kim JH, Hutt H et al. Differences in function and structure of NHERF1 and NHERF2 in cAMP and cGMP regulation of NHE3. Gastroenterology 2004; 126: A300.
47. Cha B, Kim JH, Hut H et al. cGMP inhibition of Na+/H+ antiporter 3 (NHE3) requires PDZ domain adapter NHERF2, a broad specificity protein kinase G-anchoring protein. J. Biol. Chem. 2005; 280: 16642-50.
48. Kim JH, Lee-Kwon W, Park JB, Ryu SH, Yun CHC, Donowitz M. Ca2+-dependent inhibition of Na+/H+ exchanger 3 (NHE3) requires an NHE3-E3KARP-α-actinin-4 complex for oligomerization and endocytosis. J. Biol. Chem. 2002; 277: 23714-24.
49. Lee-Kwon W, Kim JH, Choi JW et al. Ca2+-dependent inhibition of NHE3 requires PKCα which binds to E3KARP to decrease surface NHE3 containing plasma membrane complexes. Am. J. Physiol. Cell Physiol. 2003; 285: C1527-36.
50. Li X, Leu S, Cheong A et al. Akt2, phosphatidylinositol 3-kinase, and PTEN are in lipid rafts of intestinal cells: Role in absorption and differentiation. Gastroenterology 2004; 126: 122-35.
51. Lee-Kwon W, Kawano K, Choi JW, Kim JH, Donowitz M. Lysophosphatidic acid stimulates brush border Na+/H+ exchanger 3 (NHE3) activity by increasing its exocytosis by an NHE3 kinase A regulatory protein-dependent mechanism. J. Biol. Chem. 2003; 278: 16494-501.
52. Cha B, Kenworthy A, Murtazinal R, Donowitz M. The lateral mobility of NHE3 on the apical membrane of renal epithelial OK cells is limited by the PDZ domain proteins NHERF1/2, but is dependent on an intact actin cytoskeleton as determined by FRAP. J. Cell Sci. 2004; 117: 3353-65.
53. Haggie PM, Stanton BA, Verkman AS. Increased diffusional mobility of CFTR at the plasma membrane after deletion of its C-terminal PDZ binding motif. J. Biol. Chem. 2004; 279: 5494-500.
54. Janecki AJ, Janecki M, Akhter S, Donowitz M. Basic fibroblast growth factor stimulates surface expression and activity of Na+/H+ exchanger NHE3 via mechanism involving phosphatidylinositol 3-kinase. J. Biol. Chem. 2000; 275: 8133-42.
55. Akhter S, Cavet ME, Tse CM, Donowitz M. C-terminal domains of Na+/H+ exchanger isoform 3 are involved in the basal and serum-stimulated membrane trafficking of the exchanger. Biochemistry 2000; 39: 1990-2000.
56. Khurana S, Nath SK, Levine SA et al. Brush border phosphatidylinositol 3-kinase mediates epidermal growth factor stimulation of intestinal NaCl absorption and Na+/H+ exchange. J. Biol. Chem. 1996; 271: 9919-27.
57. Krieg J, Hunter T. Identification of the two major epidermal growth factor-induced tyrosine phosphorylation sites in the microvillar core protein ezrin. J. Biol. Chem. 1992; 267: 19258-65.
58. Gautreau A, Poullet PR, Louvard D, Arpin M. Ezrin, a plasma membrane-microfilament linker, signals cell survival through the phosphatidyl inositol 3-kinase/Akt pathway. Proc. Natl. Acad. Sci. U.S.A. 1999; 96: 7300-305.
59. Zhao HR, Shiue H, Palkon S et al. Ezrin regulates NHE3 translocation and activation after Na+-glucose cotransport. Proc. Natl. Acad. Sci. U.S.A 2004; 101: 9485-90.
60. Shiue H, Musch MW, Wang YM, Chang EB, Turner JR. Akt2 phosphorylates ezrin to trigger NHE3 translocation and activation. J. Biol. Chem. 2005; 280: 1688-95.
61. Turner JR, Black ED. SGLT1-mediated Na+- glucose cotransport activates NHE3 via a p38 MAPK-dependent pathway. Gastroenterology 2000; 118: A870-A871.
62. Turner JR, Black ED. NHE3-dependent cytoplasmic alkalinization is triggered by Na+-glucose cotransport in intestinal epithelia. Am. J. Physiol. Cell Physiol. 2001; 281: C1533-41.
63. Shiue H, Musch MW, Wang YM, Chang EB, Turner JR. Akt2 phosphorylates ezrin to trigger NHE3 translocation and activation. J. Biol. Chem. 2005; 280: 1688-95.
64. Oshiro N, Fukata Y, Kaibuchi K. Phosphorylation of moesin by Rho-associated kinase (Rho-kinase) plays a crucial role in the formation of microvilli-like structures. J. Biol. Chem. 1998; 273: 34663-66.
65. Matsui T, Maeda M, Doi Y et al. Rho-kinase phosphorylates COOH-terminal threonines of ezrin/radixin/moesin (ERM) proteins and regulates their head-to-tail association. J. Cell Biol. 1998; 140: 647-57.
66. Takahashi K, Sasaki T, Mammoto A et al. Direct interaction of the Rho GDP dissociation inhibitor with ezrin/radixin/moesin initiates the activation of the Rho small G protein. J. Biol. Chem. 1997; 272: 23371-75.
67. Alexander RT, Furuya W, Szaszi K, Orlowski J, Grinstein S. Rho GTPases dictate the mobility of the Na/H exchanger NHE3 in epithelia: Role in apical retention and targeting. Proc. Natl. Acad. Sci. U.S.A. 2005; 102: 12253-58.
68. Kurashima K, Chow CW, Grinstein S, Orlowski J. Na+/H+ exchanger (NHE3) activity is regulated by a phosphatidylinositol 3′-kinase dependent pathway. FASEB J. 1998; 12: A1025.
69. Szaszi K, Kurashima K, Kapus A et al. RhoA and Rho kinase regulate the epithelial Na+/H+ exchanger NHE3 - Role of myosin light chain phosphorylation. J. Biol. Chem. 2000; 275: 28599-606.
70. Lee-Kwon W, Johns DC, Cha BY et al. Constitutively active phosphatidylinositol 3-kinase and AKT are sufficient to stimulate the epithelial Na+/H+ exchanger 3. J. Biol. Chem. 2001; 276: 31296-304.
71. Hayashi H, Szaszi K, Coady-Osberg N et al. Inhibition and redistribution of NHE3, the apical Na+/H+ exchanger, by Clostridium difficile toxin B. J. Gen. Physiol. 2004; 123: 491-504.