1. Synaptic plasticity is thought to underlie learning and memory formation in the brain. However, how synaptic plasticity is induced during these processes remains controversial. An attractive candidate mechanism for learning at the neuronal level is spike-timing-dependent synaptic plasticity (STDP), which depends on the precise (millisecond) timing of the synaptic input and the postsynaptic action potential. This temporal relationship resembles typical features of associative learning. Here we review recent evidence suggesting that STDP is likely to underlie certain forms of learning.
2. First we discuss the cellular mechanisms of STDP elucidated by in vitro experiments. A special focus is put onto aspects known to differ between in vitro preparations and the in vivo situation.
3. Next we review the experimental induction of STDP in vivo, in various systems ranging from Xenopus tectum to human motor cortex.
4. The last part of the review addresses the question whether STDP can be induced by activity patterns occurring during normal behaviour.
5. We conclude that STDP is a robust phenomenon in vivo and a likely mechanism underlying sensory map plasticity in the neocortex. Further experimental evidence is required to determine whether STDP also has a role in more complex forms of learning.
One of the central questions in neuroscience is understanding the mechanisms mediating the enormous information storage capacity of the brain. The discovery of long-term potentiation (LTP) of synaptic transmission1 and its counterpart, long-term depression (LTD),2 sparked off an avalanche of research which ultimately led to the hypothesis that activity-dependent synaptic plasticity is both necessary and sufficient for memory formation.3,4 Although there is still debate about the exact stage(s) of memory formation at which synaptic plasticity is required, it is now generally accepted that synaptic plasticity participates in many forms of learning and memory formation.3-5
With this proviso, the present review addresses the question of what mechanisms are likely to underlie induction of synaptic plasticity in the vertebrate brain in vivo. Experimentally, synaptic plasticity can be induced by a plethora of protocols, which can loosely be grouped into two groups: frequency-dependent and pairing-dependent. In frequency-dependent protocols, the sign and magnitude of the change in synaptic strength depends on the frequency of synaptic activation. Low-frequency activation as a rule causes LTD, whereas high-frequency activation typically elicits LTP. In contrast, pairing-dependent protocols induce plasticity through coincidence of presynaptic activation and postsynaptic depolarisation. The duration of coincident activation during pairing varies greatly in these protocols, from several hundred milliseconds6 to just one pre- and postsynaptic action potential in a specific form of synaptic plasticity termed spike-timing-dependent plasticity (STDP). In contrast to frequency-dependent protocols, during STDP the sign and magnitude of the change in synaptic strength depends on the precise timing of pre- and postsynaptic activation.
Which induction mechanism is employed in vivo will depend on the activity patterns occurring in the brain during learning, and ultimately on how information is represented and processed in the brain. Information theory predicts that information can be coded either exclusively in the rate of action potential firing7 or using both the rate and timing of action potentials.8 Mounting evidence suggests that spike-timing is likely to be important for information processing in both invertebrates9 and vertebrates. In vivo whole-cell recordings from the cortex of rats during anaesthesia,10 quiet wakefulness11 and free exploration12 show surprisingly low action potential rates. This suggests that single action potentials have a much higher information content than previously thought; an interpretation which is further supported by the recent finding that the firing of a single cortical neuron can elicit whisking movement.13 In this study the importance of spike-timing is highlighted by the fact that whisking was phase-locked to the timing of individual action potentials. In summary, there is evidence that at least in some brain areas, such as somatosensory cortex, spike-timing is used to encode information. This makes STDP a prime candidate for induction of synaptic plasticity. In this review we will discuss the cellular mechanisms underlying STDP induction in vitro with a focus on how this might differ in vivo. We will then review recent evidence suggesting that STDP occurs in vivo.
STDP is a form of bi-directional plasticity in which the temporal order of pre- and postsynaptic action potentials on a precise (millisecond) time scale triggers changes in synaptic strength (reviewed in Dan & Poo, 2004,14 200615). The dependence of synaptic plasticity on temporal order was first described in experiments using different stimulation intensities for ipsi- and contralateral projections from the entorhinal cortex to the dentate gyrus.16 Levy and Steward noted that pairing short stimulus trains of a weak input with a strong input caused timing-dependent plasticity in the weak input, with LTP elicited when the weak input preceded the strong stimulus train, whereas LTD was observed for the reverse order.
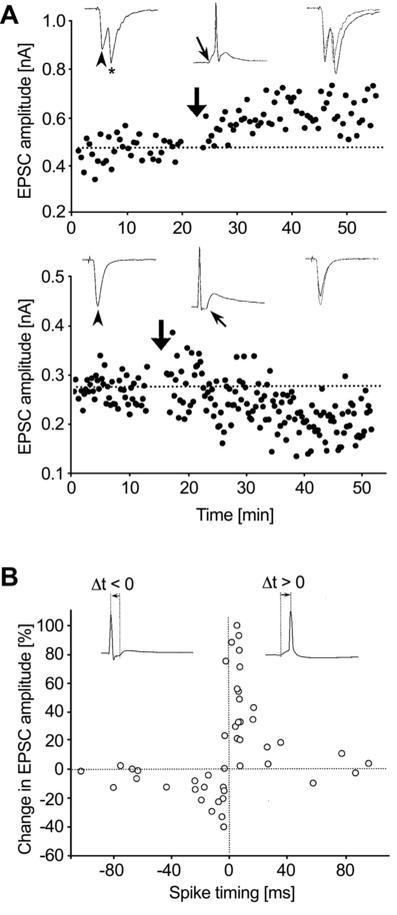
A detailed characterisation of the timing-dependence of STDP (see Figure 1) showed that most synapses undergo LTP when the postsynaptic action potential follows the synaptic input (positive timing), whereas LTD is usually observed when the postsynaptic action potential precedes the synaptic input (negative timing).6,17,18 The functional interpretation of these results is that synaptic inputs that contribute to postsynaptic firing are potentiated, whereas uncorrelated inputs are depressed. This is in essence Hebb’s postulate,19 which is computationally attractive since it directly relates plasticity of an input to its contribution to neuronal output.

Figure 1. STDP learning rules in vitro. Paired recordings were performed in dissociated cultures of rat hippocampal neurons. A: Stimulation of the presynaptic neuron gives rise to an EPSC (top, left inset, arrowhead). After pairing pre- and postsynaptic action potentials in current clamp (top, large arrow), with the EPSP leading the postsynaptic action potentials by +5 ms (top, middle inset, small arrow indicates EPSP onset), synaptic strength is persistently enhanced. Conversely, LTD is induced if the EPSP follows the postsynaptic action potential during pairing (bottom). B: The full STDP timing curve is characterised by induction of LTP for positive and LTD for negative timings. Note the striking transition from maximal LTP to maximal LTD over the very narrow time window around 0 ms. Reproduced with permission from Bi & Poo,18 copyright 1998 by the Society for Neuroscience.
However, STDP timing requirements differ between cell types and synapses: connections between neocortical spiny stellate neurons display LTD over a wide range of positive and negative timings,20 whereas hippocampal inhibitory connections potentiate over a symmetrical time window of ± 20 ms.21 In addition, STDP also depends on the timing of preceding action potentials,22,23 the sub-cellular location of inputs in the dendritic tree,24-26 the firing mode during induction,26-29 and the generation of dendritic spikes.26,29-32
Despite cell type and synapse specific differences, there is little doubt that active backpropagation of action potentials into the dendritic tree33,34 serves as the feedback signal to synapses during STDP induction.35 The common model of STDP induction assumes that depolarisation associated with backpropagating action potentials triggers plasticity via relief of the voltage-dependent block of the NMDA-receptor by magnesium ions.36,37 At positive spike timings this leads to substantial calcium influx through synaptic NMDA-receptors, triggering LTP.38,39 Consistent with this it has recently been shown that brief depolarisations early after glutamate binding are best able to activate NMDA-receptors.40 In contrast, the moderate NMDA-receptor mediated calcium signal evoked by pairings at negative times is thought to trigger LTD.14,41-43 However, LTD induction appears to be more heterogeneous since it has also been reported to depend on NMDA-receptor desensitisation,24 voltage-gated calcium channels,18 metabotropic glutamate receptors20,39,44 and presynaptic NMDA-receptors.44,45
Can the requirements for STDP identified in vitro be observed in vivo? With respect to spike timing there is ample evidence that action potentials in neuronal networks can be activated with precise millisecond timing during physiological stimuli (Figure 2A,B).46-48 The extent of action potential backpropagation in vitro can be regulated by dendritic membrane potential,49,50 and so is likely to be influenced by the level of excitatory and inhibitory synaptic drive in vivo. This would be expected to impact on STDP induction in vivo where network activity is thought to be much greater than in the relatively quiescent slice preparation. Recordings from anaesthetised animals frequently show periodical oscillations in membrane potential termed up- and down-states.51,52 Up-states have been interpreted as ‘high-conductance states’53 (but see Waters & Helmchen54), which might compromise action potential backpropagation.55 In contrast, recent studies have found that action potential backpropagation is similar in vitro and in vivo (Figure 2C),56-59 and can actually be boosted in vivo during up-states (Figure 2D).60 A greater understanding of action potential backpropagation in vivo under behaviourally relevant conditions (if feasible, see Lee et al.12) is required to judge whether STDP is likely to be a robust phenomenon in vivo.

Figure 2. Temporal fidelity and action potential backpropagation in vivo. A,B: Simultaneous extracellular recordings in layer 4 and layer 2/3 of rat barrel cortex. A: Example spike trains in response to multi-whisker deflection (bottom). Note the low overall action potential rates. B: Raster plot of spikes elicited by 100 whisker deflections shows that layer 4 neurons consistently fire before layer 2/3 cells. C: Backpropagating action potentials were recorded at different locations on the apical dendrite of neocortical L2/3 pyramidal neurons in brain slices ( in vitro) and in vivo. The amplitude of backpropagating APs declines with distance from the soma to a similar extent in vitro and in vivo. D: Two-photon imaging of calcium transients evoked by backpropagating action potentials in the apical dendrite of L2/3 pyramidal neurons in vivo. Action potentials fired from an up state (“Up”) elicit larger calcium transients, suggesting that backpropagation is boosted during up states. A,B reproduced with permission from Celikel, Szostak & Feldman.48 C,D reproduced with permission from Waters et al.59 and Waters & Helmchen,60 copyright 2003 and 2004 by the Society for Neuroscience.
Another open question is the relative importance of dendritic spikes for in vivo plasticity induction. Dendritic spikes are regenerative events in the dendrite which can be elicited by strong extracellular stimulation (e.g. Holthoff et al.31 and Schiller et al.61) or by high-frequency bursts of backpropagating action potentials (e.g. Larkum, Kaiser & Sakmann62 and Kampa & Stuart63). Both forms of dendritic spikes are able to induce synaptic plasticity in vitro,26,29-31 but their contribution to in vivo plasticity is controversial.64,65 New techniques like two-photon fluorescence imaging that allow high resolution measurements of dendritic calcium signals in vivo66 can be used to detect dendritic spikes in cortical pyramidal neurons during extracellular stimulation or sensory input.59,67 Further studies will be needed to explore the importance of these dendritic spikes to information processing and induction of synaptic plasticity in the intact brain.
The first characterisation of STDP induced in vivo was provided by Zhang and co-workers using electrical stimulation of retinotectal afferents in the Xenopus tadpole while recording from tectal neurons.68 Pairing was performed using suprathreshold stimulation of one input combined with subthreshold activation of a second, independent input. This was a landmark study since it provided direct evidence for STDP in vivo with timing requirements for induction of LTP and LTD similar to those found in vitro. In addition, it provided a plausible mechanism for refinement of the retinotectal map during development. It was subsequently shown that repetitive visual input elicits LTP at retinotectal afferents.69 LTP induction depended on postsynaptic action potential firing, NMDA-receptor activation and occluded further potentiation, consistent with STDP observed in vitro. However, the light stimulus used typically evoked several action potentials, making it impossible to determine the contribution of single EPSP-action potential pairings. This question was addressed in a recent paper which found that pairing subthreshold visual stimuli with single action potentials evoked in tectal neurons by current injection elicits STDP with conventional timing requirements.70 In summary, this series of studies strongly supports a critical role for STDP in the activity-dependent refinement of retinotectal afferents in Xenopus laevis.
There is also evidence for STDP in the mammalian brain. In the kitten, pairing of orientated gratings of a given orientation with extracellular stimulation of the cortex induced shifts in the orientation preference of this area as assessed by intrinsic optical imaging.71 The observed effects were consistent with STDP learning rules: when the visual stimulus preceded electrical stimulation of the cortex by approximately 20 ms, the orientation preference was shifted towards the paired orientation. Conversely, if the visual stimulus followed electrical activation by ∼10 ms, the orientation preference was shifted away from the paired orientation. A complimentary approach was pursued by Yao and Dan72 who paired gratings of different orientation while electrically recording the orientation preference of single neurons in primary visual cortex. This caused a shift in the orientation tuning of the recorded neurons away from their initially preferred orientation towards the orientation of the stimulus that was presented first. It was subsequently shown that this shift was most likely mediated by STDP of intracortical connections.73
Similar results have been obtained for the representation of visual space.74 The spike-timing of neurons in primary visual cortex was found to be precisely controlled by visual activation of their receptive field. When the receptive fields of two neurons were activated repeatedly in a defined temporal order, the receptive field was found to shift towards the location which was presented first. A similar displacement of the receptive field has recently been demonstrated at the single cell level using pairing of visual input with precisely timed postsynaptic action potentials,75 suggesting that STDP is likely to underlie this phenomenon. Taken together, these data provide strong evidence that STDP can be induced experimentally in the visual cortex, both by pairing visual stimuli with electrical stimulation and by pairing two visual stimuli.
Remarkably, psychophysical experiments suggest that pairing different visual stimuli can have similar effects on both the orientation preference72 and the spatial representation74 in humans. In addition, a timing-dependent form of plasticity can be induced in the human brain during pairing of transcranial magnetic stimulation (TMS) of the motor cortex with peripheral nerve stimulation.76 This study reported a persistent enhancement of motor evoked potentials when nerve stimulation precedes TMS-evoked cortical activation by 25 ms (Figure 3). This potentiation is thought to be expressed in motor cortex, is long-lasting (up to 60 min) and depends on NMDA-receptor activation.77 A later extension of the pairing intervals showed that motor evoked potentials can also be depressed by this paradigm, revealing a timing curve with striking similarity to classical STDP learning rules but offset by approximately +20 ms.78
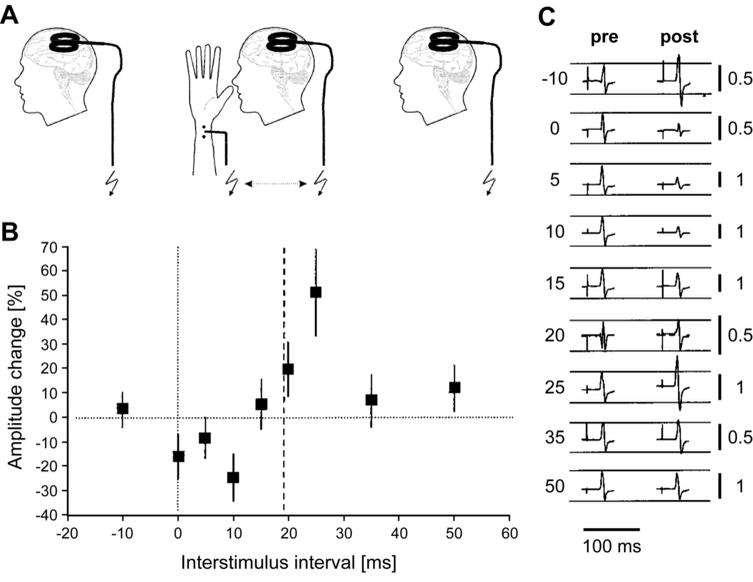
Figure 3. STDP induction in human motor cortex. A: Motor evoked potentials (MEPs) were evoked by transcranial magnetic stimulation (TMS) of the motor cortex. Pairing was carried out between TMS and electrical stimulation of the median nerve in the periphery (middle). B: The effect of pairing depends on inter-stimulus interval: stimulation of the peripheral nerve 10 ms before TMS (+10 ms) depresses MEPs, whereas MEPs are potentiated at +25 ms. The full timing curve has striking similarity to STDP curves observed in vitro (Figure 1), but is offset by +20 ms, which is approximately the time required for excitation from the peripheral nerve to reach the motor cortex. C: Example MEPs before and after pairing at the timings indicated on the left. Calibration bars on the right are in mV. Reproduced with permission from Wolters et al..78
While all of the studies reviewed above have demonstrated that experimental induction of STDP is possible in vivo, a question of equal importance is whether the activity patterns occurring during normal behaviour are able to induce STDP. This question was addressed by recent papers investigating the synaptic mechanisms underlying map plasticity due to sensory deprivation in rat barrel cortex (reviewed in Feldman & Brecht79). The barrel cortex is ideally suited for these studies since it contains an ordered, somatotopic map representing the animal’s facial whiskers (reviewed in Petersen80). This makes it possible to perform sensory deprivation by plucking a row of whiskers and, after several days during which the animal is exposed to its normal environment, the corresponding cortical columns can be identified in vitro in a slice preparation.81 This experimental regime was found to induce LTD of evoked field potentials at L4 to L2/3 feed-forward synapses in deprived barrels,82 occluding further LTD induction and enhancing induction of LTP. This in vivo induction of synaptic plasticity could be due to sensory deprivation affecting either average action potential firing rates or the relative timing of pre- and postsynaptic action potentials. This question was addressed using simultaneous extracellular recordings in L4 and L2/3 of the same barrel in freely behaving rats.48 Acute whisker deprivation caused only a modest reduction in average action potential frequency. The authors then assessed relative spike-timing between L4 and L2/3 in anaesthetised animals. When all whiskers where deflected simultaneously, spike-timing was precise, with L4 neurons spiking several milliseconds before L2/3 neurons (Figure 2A,B). Acute deprivation of the principal whisker caused an immediate reversal of firing order and a drastic decorrelation of spike trains in the two layers, effects that are known to drive synaptic depression during STDP.
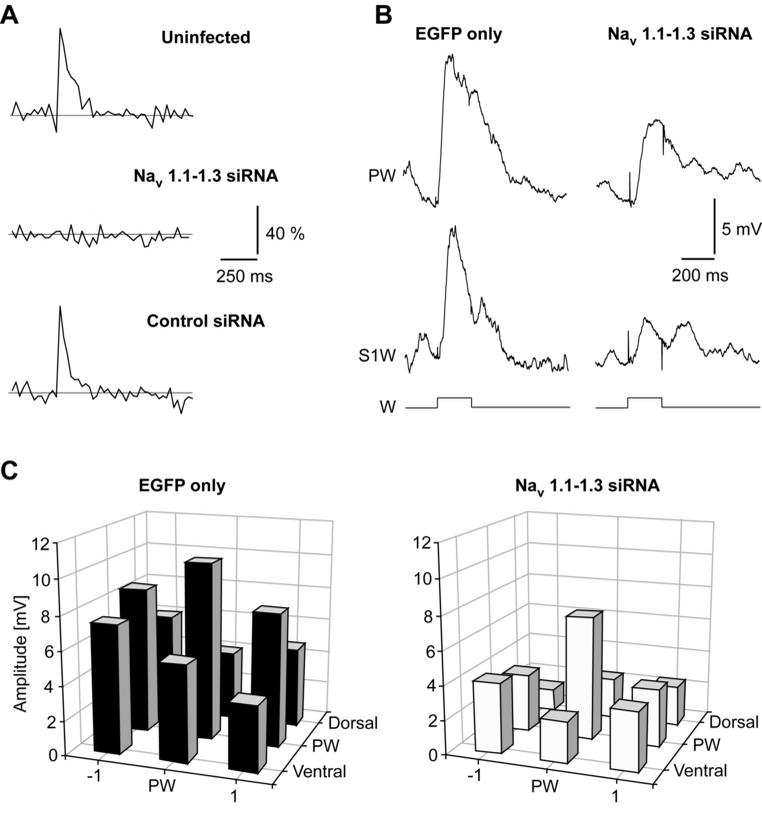
Figure 4. Action potential backpropagation is required for normal developmental strengthening of sensory responses in barrel cortex. A: In vivo imaging of action potential evoked calcium transients in the apical dendrite of layer 2/3 pyramidal neurons in rat barrel cortex. Neurons infected with a viral vector carrying short interfering RNAs (siRNAs) targeting voltage-gated sodium channel subunits (Nav1.1-1.3) displayed no backpropagation (middle), while infection with enhanced GFP alone had no effect (control siRNA). B: Example of sensory responses evoked by deflection of the principal whisker (PW) and surround whiskers (S1W) in neurons with (Nav 1.1-1.3 siRNA) and without sodium channel knock down (EGFP only). C: Sodium channel knock down significantly reduces the sensory responses to both principal whiskers (PW) and surround whiskers (±1, ventral, dorsal), suggesting that action potential backpropagation is crucial for synapse maturation. Reproduced with permission from Komai et al.83
Additional evidence that STDP underlies the development of sensory responses in barrel cortex was recently provided.83 As mentioned above, induction of STDP presumably depends on active action potential backpropagation, which is mediated by dendritic voltage-gated sodium channels.33 Komai and colleagues83 employed lentivirus-based knock down of voltage-gated sodium channel subunits by siRNAs in vivo to assess the role of postsynaptic, somatodendritic excitability in cortical maturation. The knock down effectively abolished action potential backpropagation as assessed by calcium imaging (Figure 4A), and would also have compromised dendritic spike generation while leaving action potential threshold unaltered. After approximately one week of normal behaviour, this caused a marked reduction in the amplitude of sensory postsynaptic potentials evoked by deflection of the principal whisker and the adjacent surround whiskers (Figure 4B,C). The strength of this approach is that only a small number of neurons was infected, which left the network virtually unaltered. The observed effect is therefore only dependent on the excitability of the postsynaptic neuron, which strongly suggests that STDP is crucial during normal development of the barrel cortex.
Over the last decade, evidence has mounted suggesting that STDP is a common mechanism shaping synaptic strength during learning and development. It is readily induced in vitro in several different species and brain areas6,17,18,68,84 and in both glutamatergic (see above) and GABAergic synapses.21,85 In vivo, orientation preference in visual cortex and map plasticity in barrel cortex is likely to be controlled by STDP learning rules. Could STDP also have a role in more complex learning tasks such as classical conditioning performed by pairing an unconditioned stimulus with a conditioned stimulus? This seems likely, as such learning paradigms are remarkably similar to pairing of presynaptic and postsynaptic action potentials during induction of STDP. It has been pointed out, however, that the timescale of the two processes is drastically different:86 STDP occurs within a time window of tens of milliseconds, while classical conditioning takes place over tens of seconds. Simulations suggest that this apparent discrepancy can be resolved if the sensory stimuli elicit sustained responses which decay slowly.86 Further experiments incorporating STDP learning rules in behavioural studies will be required to investigate how information storage and modulation of neural circuits can occur on the basis of a spike-timing based neural code.
1. Bliss TV, Lømo T. Long-lasting potentiation of synaptic transmission in the dentate area of the anaesthetized rabbit following stimulation of the perforant path. J. Physiol. 1973; 232: 331-56.
2. Lynch GS, Dunwiddie T, Gribkoff V. Heterosynaptic depression: a postsynaptic correlate of long-term potentiation. Nature 1977; 266: 737-9.
3. Martin SJ, Grimwood PD, Morris RG. Synaptic plasticity and memory: an evaluation of the hypothesis. Annu. Rev. Neurosci. 2000; 23: 649-711.
4. Morris RG, Moser EI, Riedel G, Martin SJ, Sandin J, Day M, et al. Elements of a neurobiological theory of the hippocampus: the role of activity-dependent synaptic plasticity in memory. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2003; 358: 773-86.
5. Whitlock JR, Heynen AJ, Shuler MG, Bear MF. Learning induces long-term potentiation in the hippocampus. Science 2006; 313: 1093-7.
6. Debanne D, Gahwiler BH, Thompson SM. Long-term synaptic plasticity between pairs of individual CA3 pyramidal cells in rat hippocampal slice cultures. J. Physiol. 1998; 507: 237-47.
7. Sanger TD. Neural population codes. Curr. Opin. Neurobiol. 2003; 13: 238-49.
8. Petersen RS, Panzeri S, Diamond ME. Population coding in somatosensory cortex. Curr. Opin. Neurobiol. 2002; 12: 441-7.
9. Perez-Orive J, Bazhenov M, Laurent G. Intrinsic and circuit properties favor coincidence detection for decoding oscillatory input. J. Neurosci. 2004; 24: 6037-47.
10. Manns ID, Sakmann B, Brecht M. Sub- and suprathreshold receptive field properties of pyramidal neurones in layers 5A and 5B of rat somatosensory barrel cortex. J. Physiol. 2004; 556: 601-22.
11. Margrie TW, Brecht M, Sakmann B. In vivo, low-resistance, whole-cell recordings from neurons in the anaesthetized and awake mammalian brain. Pflügers Arch. 2002; 444: 491-8.
12. Lee AK, Manns ID, Sakmann B, Brecht M. Whole-cell recordings in freely moving rats. Neuron 2006; 51: 399-407.
13. Brecht M, Schneider M, Sakmann B, Margrie TW. Whisker movements evoked by stimulation of single pyramidal cells in rat motor cortex. Nature 2004; 427: 704-10.
14. Dan Y, Poo MM. Spike timing-dependent plasticity of neural circuits. Neuron 2004; 44: 23-30.
15. Dan Y, Poo MM. Spike timing-dependent plasticity: from synapse to perception. Physiol. Rev. 2006; 86: 1033-48.
16. Levy WB, Steward O. Temporal contiguity requirements for long-term associative potentiation/depression in the hippocampus. Neuroscience 1983; 8: 791-7.
17. Markram H, Lubke J, Frotscher M, Sakmann B. Regulation of synaptic efficacy by coincidence of postsynaptic APs and EPSPs. Science 1997; 275: 213-5.
18. Bi GQ, Poo MM. Synaptic modifications in cultured hippocampal neurons: dependence on spike timing, synaptic strength, and postsynaptic cell type. J. Neurosci. 1998; 18: 10464-72.
19. Hebb D. The Organization of Behavior: A Neuropsychological Theory, Wiley, New York. 1949.
20. Egger V, Feldmeyer D, Sakmann B. Coincidence detection and changes of synaptic efficacy in spiny stellate neurons in rat barrel cortex. Nature Neurosci. 1999; 2: 1098-105.
21. Woodin MA, Ganguly K, Poo MM. Coincident pre- and postsynaptic activity modifies GABAergic synapses by postsynaptic changes in Cl− transporter activity. Neuron 2003; 39: 807-20.
22. Froemke RC, Dan Y. Spike-timing-dependent synaptic modification induced by natural spike trains. Nature 2002; 416: 433-8.
23. Wang HX, Gerkin RC, Nauen DW, Bi GQ. Coactivation and timing-dependent integration of synaptic potentiation and depression. Nature Neurosci. 2005; 8: 187-93.
24. Froemke RC, Poo MM, Dan Y. Spike-timing-dependent synaptic plasticity depends on dendritic location. Nature 2005; 434: 221-5.
25. Sjostrom PJ, Hausser M. A cooperative switch determines the sign of synaptic plasticity in distal dendrites of neocortical pyramidal neurons. Neuron 2006; 51: 227-38.
26. Letzkus JJ, Kampa BM, Stuart GJ. Learning rules for spike timing-dependent plasticity depend on dendritic synapse location. J. Neurosci. 2006; 26: 10420-9.
27. Pike FG, Meredith RM, Olding AW, Paulsen O. Rapid report: postsynaptic bursting is essential for ‘Hebbian’ induction of associative long-term potentiation at excitatory synapses in rat hippocampus. J. Physiol. 1999; 518: 571-6.
28. Birtoli B, Ulrich D. Firing mode-dependent synaptic plasticity in rat neocortical pyramidal neurons. J. Neurosci. 2004; 24: 4935-40.
29. Kampa BM, Letzkus JJ, Stuart GJ. Requirement of dendritic calcium spikes for induction of spike-timing-dependent synaptic plasticity. J. Physiol. 2006; 574: 283-90.
30. Golding NL, Staff NP, Spruston N. Dendritic spikes as a mechanism for cooperative long-term potentiation. Nature 2002; 418: 326-31.
31. Holthoff K, Kovalchuk Y, Yuste R, Konnerth A. Single-shock LTD by local dendritic spikes in pyramidal neurons of mouse visual cortex. J. Physiol. 2004; 560: 27-36.
32. Humeau Y, Luthi A. Dendritic calcium spikes induce bi-directional synaptic plasticity in the lateral amygdala. Neuropharmacology 2007; 52: 234-43.
33. Stuart GJ, Sakmann B. Active propagation of somatic action potentials into neocortical pyramidal cell dendrites. Nature 1994; 367: 69-72.
34. Spruston N, Schiller Y, Stuart G, Sakmann B. Activity-dependent action potential invasion and calcium influx into hippocampal CA1 dendrites. Science 1995; 268: 297-300.
35. Magee JC, Johnston D. A synaptically controlled, associative signal for Hebbian plasticity in hippocampal neurons. Science 1997; 275: 209-13.
36. Mayer ML, Westbrook GL, Guthrie PB. Voltage-dependent block by Mg2+ of NMDA responses in spinal cord neurones. Nature 1984; 309: 261-3.
37. Nowak L, Bregestovski P, Ascher P, Herbet A, Prochiantz A. Magnesium gates glutamate-activated channels in mouse central neurones. Nature 1984; 307: 462-5.
38. Koester HJ, Sakmann B. Calcium dynamics in single spines during coincident pre- and postsynaptic activity depend on relative timing of back-propagating action potentials and subthreshold excitatory postsynaptic potentials. Proc. Natl. Acad. Sci. USA. 1998; 95: 9596-601.
39. Nevian T, Sakmann B. Spine Ca2+ signaling in spike-timing-dependent plasticity. J. Neurosci. 2006; 26: 11001-13.
40. Kampa BM, Clements J, Jonas P, Stuart GJ. Kinetics of Mg2+ unblock of NMDA receptors: implications for spike-timing dependent synaptic plasticity. J. Physiol. 2004; 556: 337-45.
41. Yang SN, Tang YG, Zucker RS. Selective induction of LTP and LTD by postsynaptic [Ca2+]i elevation. J. Neurophysiol. 1999; 81: 781-7.
42. Cho K, Aggleton JP, Brown MW, Bashir ZI. An experimental test of the role of postsynaptic calcium levels in determining synaptic strength using perirhinal cortex of rat. J. Physiol. 2001; 532: 459-66.
43. Cormier RJ, Greenwood AC, Connor JA. Bidirectional synaptic plasticity correlated with the magnitude of dendritic calcium transients above a threshold. J. Neurophysiol. 2001; 85: 399-406.
44. Bender VA, Bender KJ, Brasier DJ, Feldman DE. Two coincidence detectors for spike timing-dependent plasticity in somatosensory cortex. J. Neurosci. 2006; 26: 4166-77.
45. Sjostrom PJ, Turrigiano GG, Nelson SB. Neocortical LTD via coincident activation of presynaptic NMDA and cannabinoid receptors. Neuron 2003; 39: 641-54.
46. Riehle A, Grun S, Diesmann M, Aertsen A. Spike synchronization and rate modulation differentially involved in motor cortical function. Science 1997; 278: 1950-3.
47. Prut Y, Vaadia E, Bergman H, Haalman I, Slovin H, Abeles M. Spatiotemporal structure of cortical activity: properties and behavioral relevance. J. Neurophysiol. 1998; 79: 2857-74.
48. Celikel T, Szostak VA, Feldman DE. Modulation of spike timing by sensory deprivation during induction of cortical map plasticity. Nature Neurosci. 2004; 7: 534-41.
49. Stuart GJ, Hausser M. Dendritic coincidence detection of EPSPs and action potentials. Nature Neurosci. 2001; 4: 63-71.
50. Larkum ME, Zhu JJ, Sakmann B. Dendritic mechanisms underlying the coupling of the dendritic with the axonal action potential initiation zone of adult rat layer 5 pyramidal neurons. J. Physiol. 2001; 533: 447-66.
51. Steriade M, Nunez A, Amzica F. A novel slow (<1 Hz) oscillation of neocortical neurons in vivo: depolarizing and hyperpolarizing components. J. Neurosci. 1993; 13: 3252-65.
52. Steriade M, Timofeev I, Grenier F. Natural waking and sleep states: a view from inside neocortical neurons. J. Neurophysiol. 2001; 85: 1969-85.
53. Destexhe A, Rudolph M, Pare D. The high-conductance state of neocortical neurons in vivo. Nature Rev. Neurosci. 2003; 4: 739-51.
54. Waters J, Helmchen F. Background synaptic activity is sparse in neocortex. J. Neurosci. 2006; 26: 8267-77.
55. Williams SR. Spatial compartmentalization and functional impact of conductance in pyramidal neurons. Nature Neurosci. 2004; 7: 961-7.
56. Buzsaki G, Kandel A. Somadendritic backpropagation of action potentials in cortical pyramidal cells of the awake rat. J. Neurophysiol. 1998; 79: 1587-91.
57. Charpak S, Mertz J, Beaurepaire E, Moreaux L, Delaney K. Odor-evoked calcium signals in dendrites of rat mitral cells. Proc. Natl. Acad. Sci. USA. 2001; 98: 1230-4.
58. Quirk MC, Blum KI, Wilson MA. Experience-dependent changes in extracellular spike amplitude may reflect regulation of dendritic action potential back-propagation in rat hippocampal pyramidal cells. J. Neurosci. 2001; 21: 240-8.
59. Waters J, Larkum M, Sakmann B, Helmchen F. Supralinear Ca2+ influx into dendritic tufts of layer 2/3 neocortical pyramidal neurons in vitro and in vivo. J. Neurosci. 2003; 23: 8558-67.
60. Waters J, Helmchen F. Boosting of action potential backpropagation by neocortical network activity in vivo. J. Neurosci. 2004; 24: 11127-36.
61. Schiller J, Schiller Y, Stuart G, Sakmann B. Calcium action potentials restricted to distal apical dendrites of rat neocortical pyramidal neurons. J. Physiol. 1997; 505: 605-16.
62. Larkum ME, Kaiser KM, Sakmann B. Calcium electrogenesis in distal apical dendrites of layer 5 pyramidal cells at a critical frequency of back-propagating action potentials. Proc. Natl. Acad. Sci. USA. 1999; 96: 14600-4.
63. Kampa BM, Stuart GJ. Calcium spikes in basal dendrites of layer 5 pyramidal neurons during action potential bursts. J Neurosci. 2006; 26: 7424-32.
64. Goldberg J, Holthoff K, Yuste R. A problem with Hebb and local spikes. Trends Neurosci. 2002; 25: 433-5.
65. Lisman J, Spruston N. Postsynaptic depolarization requirements for LTP and LTD: a critique of spike timing-dependent plasticity. Nature Neurosci. 2005; 8: 839-41.
66. Svoboda K, Denk W, Kleinfeld D, Tank DW. In vivo dendritic calcium dynamics in neocortical pyramidal neurons. Nature 1997; 385: 161-5.
67. Helmchen F, Svoboda K, Denk W, Tank DW. In vivo dendritic calcium dynamics in deep-layer cortical pyramidal neurons. Nature Neurosci. 1999; 2: 989-96.
68. Zhang LI, Tao HW, Holt CE, Harris WA, Poo M. A critical window for cooperation and competition among developing retinotectal synapses. Nature 1998; 395: 37-44.
69. Zhang LI, Tao HW, Poo M. Visual input induces long-term potentiation of developing retinotectal synapses. Nature Neurosci. 2000; 3: 708-15.
70. Mu Y, Poo MM. Spike timing-dependent LTP/LTD mediates visual experience-dependent plasticity in a developing retinotectal system. Neuron 2006; 50: 115-25.
71. Schuett S, Bonhoeffer T, Hubener M. Pairing-induced changes of orientation maps in cat visual cortex. Neuron 2001; 32: 325-37.
72. Yao H, Dan Y. Stimulus timing-dependent plasticity in cortical processing of orientation. Neuron 2001; 32: 315-23.
73. Yao H, Shen Y, Dan Y. Intracortical mechanism of stimulus-timing-dependent plasticity in visual cortical orientation tuning. Proc. Natl. Acad. Sci. USA. 2004; 101: 5081-6.
74. Fu YX, Djupsund K, Gao H, Hayden B, Shen K, Dan Y. Temporal specificity in the cortical plasticity of visual space representation. Science 2002; 296: 1999-2003.
75. Meliza CD, Dan Y. Receptive-field modification in rat visual cortex induced by paired visual stimulation and single-cell spiking. Neuron 2006; 49: 183-9.
76. Stefan K, Kunesch E, Cohen LG, Benecke R, Classen J. Induction of plasticity in the human motor cortex by paired associative stimulation. Brain 2000; 123: 572-84.
77. Stefan K, Kunesch E, Benecke R, Cohen LG, Classen J. Mechanisms of enhancement of human motor cortex excitability induced by interventional paired associative stimulation. J. Physiol. 2002; 543: 699-708.
78. Wolters A, Sandbrink F, Schlottmann A, Kunesch E, Stefan K, Cohen LG, et al. A temporally asymmetric Hebbian rule governing plasticity in the human motor cortex. J. Neurophysiol. 2003; 89: 2339-45.
79. Feldman DE, Brecht M. Map plasticity in somatosensory cortex. Science 2005; 310: 810-5.
80. Petersen CC. The barrel cortex—integrating molecular, cellular and systems physiology. Pflügers Arch. 2003; 447: 126-34.
81. Finnerty GT, Roberts LS, Connors BW. Sensory experience modifies the short-term dynamics of neocortical synapses. Nature 1999; 400: 367-71.
82. Allen CB, Celikel T, Feldman DE. Long-term depression induced by sensory deprivation during cortical map plasticity in vivo. Nature Neurosci. 2003; 6: 291-9.
83. Komai S, Licznerski P, Cetin A, Waters J, Denk W, Brecht M, et al. Postsynaptic excitability is necessary for strengthening of cortical sensory responses during experience-dependent development. Nature Neurosci. 2006; 9: 1125-33.
84. Sjostrom PJ, Turrigiano GG, Nelson SB. Rate, timing, and cooperativity jointly determine cortical synaptic plasticity. Neuron 2001; 32: 1149-64.
85. Haas JS, Nowotny T, Abarbanel HD. Spike-timing-dependent plasticity of inhibitory synapses in the entorhinal cortex. J. Neurophysiol. 2006.
86. Drew PJ, Abbott LF. Extending the effects of spike-timing-dependent plasticity to behavioral timescales. Proc. Natl. Acad. Sci. USA. 2006; 103: 8876-81.