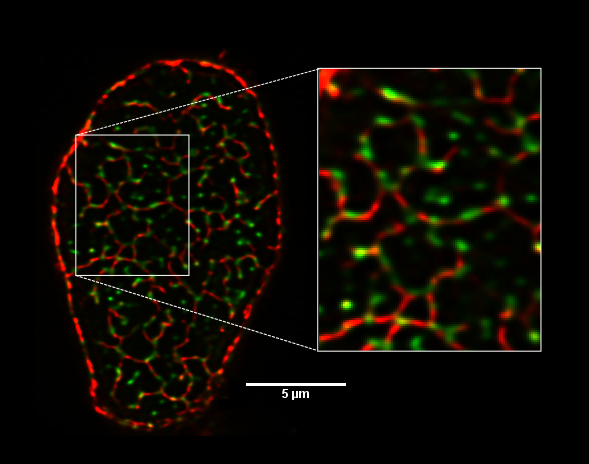
Ca2+ induced release of Ca2+ from the sarcoplasmic reticulum (SR) triggered by voltage-dependent trans-sarcolemmal Ca2+ fluxes is thought to form the basis of excitation-contraction (EC) coupling in cardiac myocytes. Close apposition of the sarcolemmal membrane with the SR Ca2+ release channels or ryanodine receptors (RyRs) is essential to achieving the rapid Ca2+ release that ensures cardiac contraction. The sarcolemmal membrane architecture is complex, with the presence of caveolar invaginations and a dense network of t-tubules near each z-disk. Such complexity in the organization of membrane structures and the compact arrays of key proteins central to EC coupling is only partially resolved when imaged by confocal microscopy. The resolution can be effectively improved by using a recently-demonstrated technique (Chen-Izu et al., 2006) to image these fine elements of the sarcolemma in single cells oriented vertically on the microscope stage. We have used this approach to visualize the distribution of the Na-Ca exchanger (NCX), Caveolin-3 (Cav3, a marker for caveolar membrane invaginations) and RyRs in enzymatically isolated and fixed rat myocytes at high resolution. The distribution of Caveolin-3 (red) and RyR (green) labelling in a single z-disk of a rat ventricular myocyte is shown in the figure below illustrating the level of detail that is achieved.

Recently, it has been suggested that some RyR clusters found distal to z-lines are not associated with junctions (Lukyanenko et al., 2007). Our data show that RyR clusters located between z-lines are generally closely associated with longitudinal elements of the tsystem (Soeller & Cannell, 1999) as identified by NCX and Cav3 labelling. Thus these RyR clusters are likely in junctions with the longitudinal t-system.
Our data provide 3D data sets for detailed modelling studies to improve our understanding of the potential role of NCX in modulating SR release.
Bridge JHB, Inoue M & Cannell MB. (2003) Journal of Molecular and Cellular Cardiology, 35: A14.
Chen-Izu Y, McCulle S, Ward CW, Soeller C, Allen BG, Rabang C, Cannell MB, Balke CW & Izu LT. (2006) Biophysical Journal, 91: 1-13.
Lines GT, Sande JB, Louch WE, Mork HK, Grottum P & Sejersted OM. (2006) Biophysical Journal, 91: 779-92.
Lukyanenko V, Ziman A, Lukyanenko A, Salnikov V & Lederer WJ. (2007) Journal of Physiology, 583: 251-269.
Soeller C & Cannell MB. (1999) Circulation Research, 84: 266-75.
Soeller C, Crossman D, Gilbert R & Cannell MB. (2007) Proceedings of the National Academy of Science of the United States of America, 104: 14958-63.