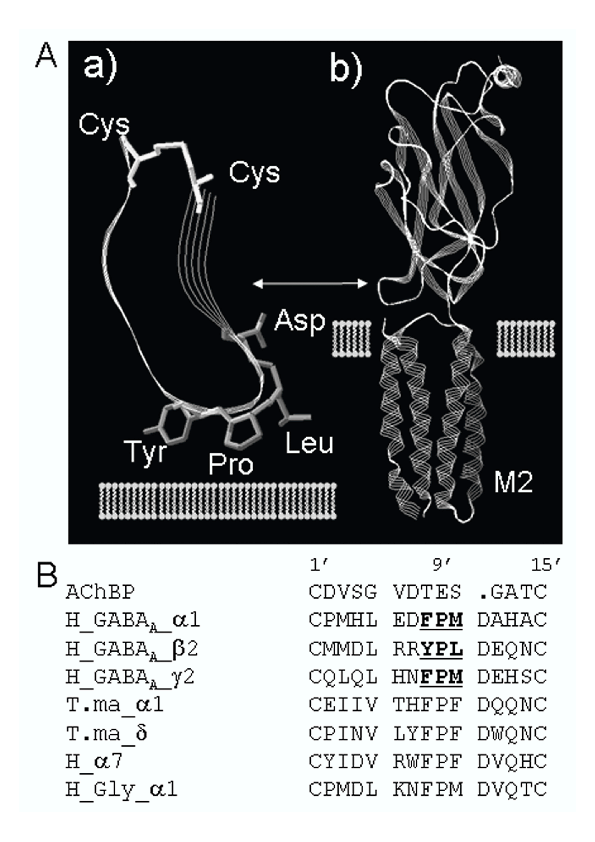
Ligand-gated ion channels respond to specific neurotransmitters by transiently opening an integral membrane ion-selective pore, allowing ions to move down their electrochemical gradient. A distinguishing feature of all members of the ligand-gated ion channel superfamily is the presence of a 13 amino acid disulfide loop (Cys-loop) in the extracellular ligand-binding domain. Structural data derived from the acetylcholine receptor place this loop at the interface between the ligand binding domain and the transmembrane pore-forming domain where it is ideally located to participate in coupling ligand binding to channel opening. We have introduced specific mutations into a conserved motif at the mid-point of the Cys-loop of the GABAA receptor subunits α1, β2 and γ2S where the sequence reads aromatic, proline, aliphatic (ArPAl motif). Receptors carrying a mutation in the Cys-loop of one of their subunits were expressed in L929 cells and responses to both GABA and drugs were assessed using the whole-cell patch clamp technique. Drug potentiation and direct activation were significantly enhanced by mutations in this Cys-loop but these effects were subunit-dependent. Currents in response to agonists were larger when mutations were carried in the α and β subunits but not in the γ subunit. In contrast, potentiation of current responses by diazepam, etomidate and pentobarbital were all enhanced when mutations were carried in the α and γ subunits, but not the β subunit. Since the disruption of interactions mediated through the ArPAl motif enhances the mutant receptor's response to both agonist and drugs we suggest that this motif in the Cys-loop of the wild-type receptor participates in interactions that create activation barriers to conformational changes during channel gating. The participation of subunits in channel gating is however, asymmetrical.
