1. Mammalian eggs are arrested at metaphase of their second meiotic division when ovulated and remain arrested until fertilized. The sperm delivers into the egg phospholipase Cζ, which triggers a series of Ca2+ spikes lasting several hours. The Ca2+ spikes provide the necessary and sufficient trigger for all the events of fertilization, including exit from metaphase II arrest and extrusion of cortical granules that block the entry of other sperm.
2. The oscillatory Ca2+ signal switches on calmodulin-dependent protein kinase II (CamKII), which phosphorylates the egg-specific protein Emi2, earmarking it for degradation. To remain metaphase II arrested eggs must maintain high levels of Maturation-Promoting Factor (MPF) activity, a heterodimer of CDK1 and cyclin B1. Emi2 prevents loss of MPF by blocking cyclin B1 degradation, a process which is achieved by inhibiting the activity of the Anaphase-Promoting Complex/Cyclosome. CamKII is not however the primary initiator in the extrusion of cortical granules.
3. Ca2+ spiking is also observed in mitosis of one-cell embryos, probably because phospholipase Cζ contains a nuclear localisation signal and so is released into the cytoplasm following nuclear envelope breakdown. The function of these mitotic Ca2+ spikes remains obscure, although they are not absolutely required for passage through mitosis.
4. Intriguingly the pattern of Ca2+ spikes observed at fertilization have an effect on both pre- and post-implantation development in a manner that is independent of their ability to activate eggs. This suggests that the Ca2+ spikes which are set in train at fertilization are having effects on processes initiated in the newly fertilized egg but whose influences are only observed several cell divisions later. The nature of the signals remain little explored but their importance is clear and so warrant further investigation.
Ca2+ is a ubiquitous intracellular signalling molecule,1 and intracellular Ca2+ (Ca2+i) changes during signal transduction processes have been widely examined over several decades. Fertilization is one such event where Ca2+i plays a pivotal role.2-5 In most species a rise in Ca2+i is induced by the fertilizing sperm and forms the essential trigger for the egg-embryo transition. This is a remarkable feat of conservation considering that eggs of different species arrest at different points in meiosis, or in some species have completed meiosis.4 Therefore Ca2+i is likely having effects on diverse signalling pathways at fertilization in eggs of different species.
Mammalian eggs are ovulated while arrested at metaphase of the second meiotic division (MetII). They remain arrested at this stage until fertilized, and if not degenerate. It makes physiological sense to prevent entry into the embryonic cell cycles without sperm, firstly because development to term of such parthenotes is not possible due to the need to have genes of paternal origin in mammals, and secondly to prevent growth of potentially cancerous cells in the female genital tract.
It has long been known that Ca2+i is both necessary and sufficient for mammalian fertilization to occur.6 Thus if Ca2+i rises are blocked all the events of fertilization are inhibited. The term ‘egg activation’ is used to describe these events and encompass not only cell cycle resumption out of MetII arrest but also the release of cortical granules (CG), which block the entry of other sperm, and any other events associated with the egg-embryo transition.
This review concentrates on recent developments in our understanding of the signalling pathway used by the Ca2+i signal from the sperm at fertilization to induce release from egg arrest. It then goes on to examine data supporting the hypothesis that Ca2+i changes are also important for longer term embryo development, several divisions after its well-established meiotic role. The review hints at some ideas as to how these two temporally separate processes may be connected.
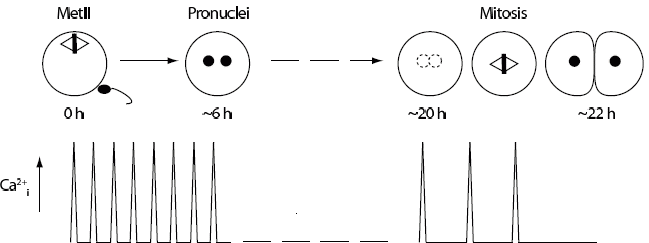
There are two main events which are triggered directly by the Ca2+i rise at fertilization: CG release, which is responsible for the block to polyspermy; and initiation of cell cycle resumption out of meiosis from MetII arrest. Before examining these processes in any detail it is first worth commenting on the spatiotemporal aspects of the Ca2+i signal in mammalian eggs. Some of the first Ca2+i recordings of eggs were performed on species in which a single Ca2+i rise was observed to pass across the egg from the site of sperm fusion. Embryonic development quickly ensued. In striking contrast when similar studies were performed on mammalian eggs it was clear that sperm induced a series of Ca2+i rises (hereafter ‘spikes’) that lasted several hours.7-9 Figure 1 illustrates the sperm-induced Ca2+ signal in mammalian eggs.

Figure 1. Schematic of Ca2+i spiking in mammalian eggs. Met II arrested mammalian eggs show a series of Ca2+i spikes lasting several hours, until pronuclei form in the 1-cell embryo. Nuclear envelope breakdown at the start of mitosis in the 1-cell embryo is the time at which Ca2+i spiking is re-initiated. Cessation of spiking is most likely due to nuclear sequestration of PLCζ, which is then released into the cytoplasm during first mitosis.
The ability of mammalian eggs to respond to sperm with a long lasting Ca2+i signal suggested that the long stimulus was required to induce the egg to activate properly. This has been borne out by elegant work which has demonstrated that in order to get complete CG release and to induce exit from MetII arrest the egg needs to have experienced multiple Ca2+i spikes.10,11 Interestingly fewer spikes are needed to promote CG release than cell cycle resumption10 but this may make physiological sense if the fertilizing sperm has a more immediate need to block the entry of other sperm than it does to trigger the egg to complete its meiotic division.
The initiator of Ca2+i release in mammalian eggs is not fully resolved but is a protein delivered into the egg by the sperm. The most likely candidate is a sperm-specific member of the phospholipase C (PLC) family, PLCζ.12-14 Although other sperm initiating factors have been reported15-17 they remain to be substantiated and there now appears some consensus that the sperm protein is probably PLCζ acting alone. The generation of a PLCζ knockout mouse would help finally to resolve this issue and is expected within the next few years. Interestingly PLCζ appears to be the initiating factor in mammals and birds,18 but not in some fish and primitive chordates, suggesting it is a recently evolved PLC member. Regardless of the actual nature of the initiating factor it is clear that the Ca2+ spikes at fertilization are driven by inositol trisphosphate production acting on its endoplasmic reticulum receptor (InsP3R).19-22 PLCζ contains within it a nuclear localisation signal,14,23,24 which promotes its accumulation in the pronuclei that form at the completion of meiosis II. The nuclear accumulation of PLCζ appears to be the mechanism by which Ca2+i spikes are terminated23,25 and accounts for the fact that when pronuclei formation is blocked Ca2+i spiking continues indefinitely.26
Although it has been established for some time that Ca2+i is required to induce cell cycle resumption, the mechanism by which this is achieved has only recently been described. The most downstream target of Ca2+i is the kinase MPF (Maturation-Promoting Factor or M-Phase Promoting Factor). MPF activity is high in unfertilized eggs and rapidly falls at fertilization.27,28 MPF is a dimer but its catalytic subunit CDK1 is without activity unless bound to its regulatory partner, which in mammals is cyclin B1. If degradation of cyclin B1 is prevented then eggs remain MetII arrested,29 even though sperm have fused and Ca2+i spiking has been initiated. High MPF activity is not just observed during MetII arrest, but is seen in all eukaryotic cells as they pass through mitosis. In mitosis, the decrease in MPF activity at the metaphase-anaphase transition is mediated by the Anaphase-Promoting Complex/Cyclosome (APC/C), a large multimeric protein complex whose activity is essential for exit from mitosis.30 By the nature of its large multisubunit size APC/C is likely subject to a number of control points in its activity; the most described being the spindle assembly checkpoint (SAC). SAC components are members of the Mad and Bub family as well as MpsI kinase.31 These act to inhibit APC/C activity before full congression and microtubule attachment of chromosomes on a metaphase spindle. In this way mis-segregation of chromosomes is avoided at anaphase. Although there is evidence that in frog MetII arrest may be achieved by components of the SAC family inhibiting APC/C, in mammals this mechanism appears less important.32 Recently a novel APC/C inhibitor Early Mitotic Inhibitor I (Emi1) was established as being required to prevent premature APC/C activity as cells enter mitosis.33,34 A related protein Emi2/Erp1 (Emi1-related protein 1), was then later identified, found to be egg specific, and now seems to be the likely target of Ca2+i action.35-38
Loss of Emi2 at fertilization is achieved through phosphorylation by calmodulin-dependent protein kinase II (CamKII). This fits with the observation that CamKII activity increases at fertilization on a Ca2+i signal,39,40 and also constitutively-active CamKII induces cyclin B1 degradation and so cell cycle resumption from MetII arrest.41 This phosphorylation of Emi2 by CamKII creates a docking site for polo kinase which further phosphorylates Emi2 and this second phosphorylation acts as a trigger for Emi2 loss, through its polyubiquitination by the E3 ligase Skpl-Cullin/F-box protein. This mechanism of degradation thus far has been demonstrated only in frog eggs.35-38 However Emi2 appears to be physiologically relevant in mammals for MetII arrest, since when this protein is knocked down in MetII arrested eggs, it induces them to parthenogenetically activate.42 Furthermore when its synthesis is prevented during oocyte maturation, the oocytes fail to arrest at MetII.43 Tagging both cyclin B1 and Emi2 with different fluorescent proteins and imaging their degradation simultaneously in the same egg, reveals that Emi2 loss occurs ahead of cyclin B1.43 Therefore the basic signal transduction pathway which operates at fertilization is likely to be activation of the APC/C through loss in Emi2, stimulated by CamKII phosphorylation. Activation of the APC/C induces degradation of cyclin B1, so MPF falls and oocytes can then resume their second meiotic division.
Interestingly CG release, which occurs ahead of cell cycle resumption, although Ca2+ dependent appears not to involve CamKII.44 Thus a constitutively active mutant of CamKII, although able to induce cell cycle resumption, does not produce the extent of CG release observed with sperm. This suggests that a second, independent signalling pathway is used which is most likely to involve myosin light chain kinase (MLCK), since inhibition of MLCK, blocks CG release.45
By the time pronuclei form in the 1-cell embryo it is committed to passage through the first cell cycle with S-phase starting shortly after pronuclei are observed. Due to sequestration of PLCζ into the pronuclei, Ca2+i spiking also stops at around this time.14,23-26 One would assume that the Ca2+i spiking has now performed its task and can be disregarded in respect of embryo development. However this is not the case.
It was shown some years ago that a transferred nucleus from a fertilized one- or two- cell embryo had the ability to induce Ca2+i spiking and so activate an unfertilized MetII egg following fusion of the two.46,47 However when the fusion experiment was performed with the pronucleus of a parthenote no egg activation from MetII arrest was seen. These observations are now easily explained by the nuclear sequestration of PLCζ into the pronuclei of the fertilized embryo, and its obvious absence from the pronuclei of parthenotes. The fact that PLCζ is stable enough in the pronucleus during the first two cell divisions, also readily explains the observation that Ca2+i spiking is seen again during the first mitotic division of fertilized embryos but not parthenotes.48 Presumably during mitosis PLCζ is released into the cytoplasm where it can generate inositol trisphosphate and so Ca2+i spikes. These spikes however appear non-essential for passage through mitosis because if they are blocked by Ca2+ buffers then mitosis proceeds with normal timings.49 Furthermore parthenotes do not show these spikes and yet readily undergo mitosis, confirming the non-essential nature of Ca2+i in the mitotic division.
There are however caveats in dismissing the importance of Ca2+i spikes to the first mitotic division. The first is that raising Ca2+ in G2 embryos accelerates entry into mitosis,49 suggesting that although Ca2+ is non-essential it can actually affect passage through this cell cycle transition. In sea urchin embryos Ca2+i does play an essential role, as shown by the block to anaphase when Ca2+i changes are buffered.50 In mammals one could argue that Emi2 should be absent at first mitosis since it is degraded on exit from MetII, and if so passage through mitosis dictated by activation of the APC/C would not be dependent on Ca2+i as it is during MetII arrest. Emi2 levels have not been assessed during the first mitotic division, however they do increase again following the cessation of Ca2+i spikes at pronucleus formation,42,43 making it possible that Emi2 is actually present during the first mitotic division. This stimulates the question of what strategies the embryo uses to degrade Emi2, because presumably this must happen in order for the APC/C to be active during the first mitotic division. So far only a Ca2+ dependent mechanism of Emi2 degradation has been described. Future studies are therefore required to assess the way in which the embryo controls Emi2 activity.
Single monotonic rises in Ca2+i are generally poor activators of mammalian eggs. Eggs may partially complete their second meiotic division, so extrude a polar body, but re-arrest at a new metaphase and stay arrested without the formation of a pronucleus.51 This is likely due to poor or transient degradation of Emi2, whose activity comes back and inhibits the APC/C allowing cyclin B1 reaccumulation and MPF activity. The general exception to this is the ability with a single Ca2+i rise to activate eggs that have passed the window of their normal in vivo time of fertilization.52 Here this is best explained by the aged eggs being less able to maintain high MPF levels. Thus the essential nature of the oscillatory Ca2+i signal may well be to provide a long enough signal in which to ensure the egg escapes meiosis.53,54 This cannot however be the sole function of Ca2+i because differing regimens of experimentally-induced Ca2+i spiking designed to give very high rates of egg activation, do not all induce good quality embryo development.10,55-60
In one study eggs were incubated in Sr2+-containing medium to experimentally induce spiking in Ca2+i for varying periods up to 24 h.60 Here Sr2+ is acting as a Ca2+ mimetic on the egg InsP3R, which in terms of channel opening contains both activating and inhibitory Ca2+ binding sites. Importantly Sr2+ is very much less (600-fold) potent than Ca2+ in inhibiting the InsP3R, so effectively sensitizes the receptor.61 It was shown that eggs incubated for varying times in Sr2+-containing medium to produce high rates of egg activation, go on to form blastocysts with different numbers of inner cell mass cells and trophectodermal cells.60 The embryos with the least inner cell mass cells, which go on to form all the embryonic structures, were those exposed to Sr2+ for the least amount of time. The regime which produced embryos that were most similar in composition to fertilized eggs, were those placed in Sr2+ media for 24 h; this period encompassing meiotic exit and first mitosis, both periods in which Ca2+i spiking is normally observed. More elegant studies have been performed by Jean-Pierre Ozil and his collaborators, using an activation chamber in which eggs can be exposed to a series of artificially induced Ca2+i spikes of varying amplitude and duration.62,63 Using such a chamber in which spike frequency and duration are altered it is clear that for protocols which all give high rates of egg activation longer term development is not uniform, with the quality of postimplantation development falling off dramatically for some Ca2+i spiking regimes.10,56-59
At present it is not resolved how Ca2+i spiking at fertilization, and possibly first mitosis, are having their effects several cell divisions later. It is proposed here that there may be two possibilities (summarized in Figure 2). The first is that a suboptimal Ca2+i signal fails to stimulate fully the degradation of proteins whose function is essentially to help maintain MetII arrest and whose presence could hinder the cell divisions of the embryo. Given that MetII arrest is so successful, for good physiological reason, and so protracted, it is likely that other signalling pathways are recruited to maintain arrest that are independent of Emi2. One such pathway is likely to be the mos…MAPKinase pathway. In mouse loss of mos, a MAPKinase Kinase Kinase, allows eggs to escape arrest after just a few hours.64 Furthermore MetII arrest in frog eggs appears to involve more than one signalling pathway.65 Therefore maintenance of arrest at MetII may involve multiple pathways all of which need to be successfully downregulated for optimal embryonic cell division.
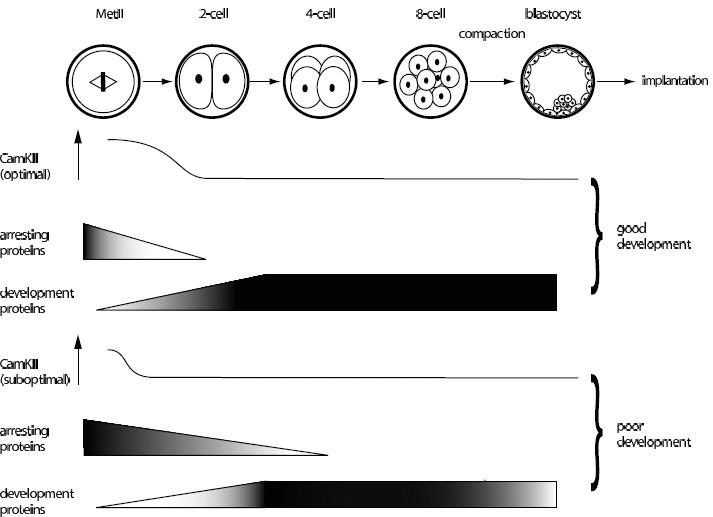
Figure 2. Schematic model of how early events of fertilization can influence embryo development. Some of the stages of pre-implantation development are depicted leading up to implantation. At fertilization, Ca2+i spiking switches on CamKII, which has an important role to play in promoting loss, probably through degradation, of a number of proteins responsible for arresting the egg at MetII before fertilization, and also in promoting the synthesis of a number of new embryonic proteins through either transcription or translation. Suboptimal CamKII activation may influence embryo development by hindering both these processes. See text for further details.
The second possibility is that the Ca2+i signal needs to switch on the expression of various proteins, whose early expression is needed for good embryo development. This hypothesis is supported by the observations that the number of Ca2+i spikes experienced by an activating egg can influence the expression of new proteins in the early embryo10 and that new protein synthesis is required for zygotic genomic activation.66 In regard of the ability of Ca2+i to influence protein expression, it is important to note that CamKII can phosphorylate and so switch on cytoplasmic polyadenylation element binding protein (CPEB)67,68 to stimulate protein expression in hippocampal dendrites through increased mRNA polyadenylation. Given the importance of CPEB in the translational efficiency of certain mRNA’s in oocytes before fertilization69 it is tempting to speculate that Ca2+i activated CamKII is also able to affect protein expression through CPEB in eggs. Zygotic transcription begins much earlier than previously thought during the 1-cell embryo stage70-72 so it remains possible that CamKII is involved in the expression of zygotic proteins. Of course these two possibilities for how Ca2+i has long-term effects are not mutually exclusive: Ca2+i could be both involved in stimulating the expression of nascent proteins (e.g. through CPEB) and also in degrading others (e.g. Emi2). Assuming both processes are reliant on Ca2+i activation of CamKII, then such hypotheses makes the actual dynamics of the signal important, since CamKII activation by Ca2+i is sensitive to frequency and amplitude.73,74
Knowledge of how the oscillatory Ca2+i signal at fertilization is initiated and the downstream signalling pathways it affects have been elucidated over recent years. The establishment of CamKII as an important transducer of Ca2+i action in cell cycle resumption opens the possibility of solving the phenomenon of how Ca2+i can have a much longer term effect on embryo development, several cell divisions later, from its established role in exit from MetII arrest. We may find that optimal embryo development is made possible in eggs by the CamKII-mediated switching on of genes through transcription and/or translation during fertilization combined with the stimulated degradation of proteins required in the protracted cell cycle arrest at MetII.
The author expresses thanks to members of the lab who been involved in this research over recent years, the Wellcome Trust to continued financial support, and Tom Ducibella for critical reading.
1. Berridge MJ, Lipp P, Bootman MD. The versatility and universality of calcium signalling. Nat. Rev. Mol. Cell Biol. 2000; 1:11-21.
2. Miyazaki S. Thirty years of calcium signals at fertilization. Semin. Cell Dev. Biol. 2006; 17:233-43.
3. Runft LL, Jaffe LA, Mehlmann LM. Egg activation at fertilization: where it all begins. Dev. Biol. 2002; 245:237-54.
4. Stricker SA. Comparative biology of calcium signaling during fertilization and egg activation in animals. Dev. Biol. 1999; 211:157-76.
5. Whitaker M. Calcium at fertilization and in early development. Physiol. Rev. 2006; 86:25-88.
6. Kline D, Kline JT. Repetitive calcium transients and the role of calcium in exocytosis and cell cycle activation in the mouse egg. Dev. Biol. 1992; 149:80-9.
7. Cuthbertson KS, Whittingham DG, Cobbold PH. Free Ca2+ increases in exponential phases during mouse oocyte activation. Nature 1981; 294:754-7.
8. Cuthbertson KS, Cobbold PH. Phorbol ester and sperm activate mouse oocytes by inducing sustained oscillations in cell Ca2+. Nature 1985; 316:541-2.
9. Miyazaki S, Igusa Y. Fertilization potential in golden hamster eggs consists of recurring hyperpolarizations. Nature 1981; 290:702-4.
10. Ducibella T, Huneau D, Angelichio E, et al. Egg-to-embryo transition is driven by differential responses to Ca2+ oscillation number. Dev. Biol. 2002; 250:280-91.
11. Lawrence Y, Ozil JP, Swann K. The effects of a Ca2+ chelator and heavy-metal-ion chelators upon Ca2+ oscillations and activation at fertilization in mouse eggs suggest a role for repetitive Ca2+ increases. Biochem. J. 1998; 335:335-42.
12. Saunders CM, Larman MG, Parrington J, et al. PLCζ: a sperm-specific trigger of Ca2+ oscillations in eggs and embryo development. Development 2002; 129:3533-44.
13. Knott JG, Kurokawa M, Fissore RA, Schultz RM, Williams CJ. Transgenic RNA interference reveals role for mouse sperm phospholipase Cζ in triggering Ca2+ oscillations during fertilization. Biol. Reprod. 2005; 72:992-6.
14. Yoda A, Oda S, Shikano T, et al. Ca2+ oscillation-inducing phospholipase C zeta expressed in mouse eggs is accumulated to the pronucleus during egg activation. Dev. Biol. 2004; 268:245-57.
15. Sette C, Paronetto MP, Barchi M, Bevilacqua A, Geremia R, Rossi P. Tr-kit-induced resumption of the cell cycle in mouse eggs requires activation of a Src-like kinase. EMBO J. 2002; 21:5386-95.
16. Perry AC, Wakayama T, Cooke IM, Yanagimachi R. Mammalian oocyte activation by the synergistic action of discrete sperm head components: induction of calcium transients and involvement of proteolysis. Dev. Biol. 2000; 217:386-93.
17. Perry AC, Wakayama T, Yanagimachi R. A novel trans-complementation assay suggests full mammalian oocyte activation is coordinately initiated by multiple, submembrane sperm components. Biol. Reprod. 1999; 60:747-55.
18. Coward K, Ponting CP, Chang HY, et al. Phospholipase Cζ, the trigger of egg activation in mammals, is present in a non-mammalian species. Reproduction 2005; 130:157-63.
19. Miyazaki S, Yuzaki M, Nakada K, et al. Block of Ca2+ wave and Ca2+ oscillation by antibody to the inositol 1,4,5-trisphosphate receptor in fertilized hamster eggs. Science 1992; 257:251-5.
20. Miyazaki S, Shirakawa H, Nakada K, Honda Y. Essential role of the inositol 1,4,5-trisphosphate receptor/Ca2+ release channel in Ca2+ waves and Ca2+ oscillations at fertilization of mammalian eggs. Dev. Biol. 1993; 158:62-78.
21. Jones KT, Nixon VL. Sperm-induced Ca2+ oscillations in mouse oocytes and eggs can be mimicked by photolysis of caged inositol 1,4,5-trisphosphate: evidence to support a continuous low level production of inositol 1,4,5-trisphosphate during mammalian fertilization. Dev. Biol. 2000; 225:1-12.
22. Shirakawa H, Ito M, Sato M, Umezawa Y, Miyazaki S. Measurement of intracellular IP3 during Ca2+ oscillations in mouse eggs with GFP-based FRET probe. Biochem. Biophys. Res. Commun. 2006; 345:781-8.
23. Larman MG, Saunders CM, Carroll J, Lai FA, Swann K. Cell cycle-dependent Ca2+ oscillations in mouse embryos are regulated by nuclear targeting of PLCζ. J. Cell Sci. 2004; 117:2513-21.
24. Kuroda K, Ito M, Shikano T, et al. The role of X/Y linker region and N-terminal EF-hand domain in nuclear translocation and Ca2+ oscillation-inducing activities of phospholipase Cζ, a mammalian egg-activating factor. J. Biol. Chem. 2006; 281:27794-805.
25. Marangos P, FitzHarris G, Carroll J. Ca2+ oscillations at fertilization in mammals are regulated by the formation of pronuclei. Development 2003; 130:1461-72.
26. Jones KT, Carroll J, Merriman JA, Whittingham DG, Kono T. Repetitive sperm-induced Ca2+ transients in mouse oocytes are cell cycle dependent. Development 1995; 121:3259-66.
27. Jones KT. Turning it on and off: M-phase promoting factor during meiotic maturation and fertilization. Mol. Hum. Reprod. 2004; 10:1-5.
28. Doree M, Hunt T. From Cdc2 to Cdk1: when did the cell cycle kinase join its cyclin partner? J. Cell Sci. 2002; 115:2461-4.
29. Madgwick S, Nixon VL, Chang HY, Herbert M, Levasseur M, Jones KT. Maintenance of sister chromatid attachment in mouse eggs through maturation-promoting factor activity. Dev. Biol. 2004; 275:68-81.
30. Peters JM. The anaphase promoting complex/cyclosome: a machine designed to destroy. Nat. Rev. Mol. Cell Biol. 2006; 7:644-56.
31. Shah JV, Cleveland DW. Waiting for anaphase: Mad2 and the spindle assembly checkpoint. Cell 2000; 103:997-1000.
32. Tsurumi C, Hoffmann S, Geley S, Graeser R, Polanski Z. The spindle assembly checkpoint is not essential for CSF arrest of mouse oocytes. J. Cell Biol. 2004; 167:1037-50.
33. Reimann JD, Freed E, Hsu JY, Kramer ER, Peters JM, Jackson PK. Emi1 is a mitotic regulator that interacts with Cdc20 and inhibits the anaphase promoting complex. Cell 2001; 105:645-55.
34. Reimann JD, Gardner BE, Margottin-Goguet F, Jackson PK. Emi1 regulates the anaphase-promoting complex by a different mechanism than Mad2 proteins. Genes Dev. 2001; 15:3278-85.
35. Liu J, Maller JL. Calcium elevation at fertilization coordinates phosphorylation of XErp1/Emi2 by Plx1 and CaMK II to release metaphase arrest by cytostatic factor. Curr. Biol. 2005; 15:1458-68.
36. Rauh NR, Schmidt A, Bormann J, Nigg EA, Mayer TU. Calcium triggers exit from meiosis II by targeting the APC/C inhibitor XErp1 for degradation. Nature 2005; 437:1048-52.
37. Tung JJ, Hansen DV, Ban KH, et al. A role for the anaphase-promoting complex inhibitor Emi2/XErp1, a homolog of early mitotic inhibitor 1, in cytostatic factor arrest of Xenopus eggs. Proc. Natl. Acad. Sci. USA 2005; 102:4318-23.
38. Hansen DV, Tung JJ, Jackson PK. CaMKII and polo-like kinase 1 sequentially phosphorylate the cytostatic factor Emi2/XErp1 to trigger its destruction and meiotic exit. Proc. Natl. Acad. Sci. USA 2006; 103:608-13.
39. Markoulaki S, Matson S, Abbott AL, Ducibella T. Oscillatory CaMKII activity in mouse egg activation. Dev. Biol. 2003; 258:464-74.
40. Markoulaki S, Matson S, Ducibella T. Fertilization stimulates long-lasting oscillations of CaMKII activity in mouse eggs. Dev. Biol. 2004; 272:15-25.
41. Madgwick S, Levasseur M, Jones KT. Calmodulin-dependent protein kinase II, and not protein kinase C, is sufficient for triggering cell-cycle resumption in mammalian eggs. J. Cell Sci. 2005; 118:3849-59.
42. Shoji S, Yoshida N, Amanai M, et al. Mammalian Emi2 mediates cytostatic arrest and transduces the signal for meiotic exit via Cdc20. EMBO J. 2006; 25:834-45.
43. Madgwick S, Hansen DV, Levasseur M, Jackson PK, Jones KT. Mouse Emi2 establishes a metaphase II spindle by re-stabilizing cyclin B1 during interkinesis. J. Cell Biol. 2006; 174:791-801.
44. Knott JG, Gardner AJ, Madgwick S, Jones KT, Williams CJ, Schultz RM. Calmodulin-dependent protein kinase II triggers mouse egg activation and embryo development in the absence of Ca2+ oscillations. Dev. Biol. 2006; 296:388-95.
45. Matson S, Markoulaki S, Ducibella T. Antagonists of myosin light chain kinase and of myosin II inhibit specific events of egg activation in fertilized mouse eggs. Biol. Reprod. 2006; 74:169-76.
46. Kono T, Carroll J, Swann K, Whittingham DG. Nuclei from fertilized mouse embryos have calcium-releasing activity. Development 1995; 121:1123-8.
47. Zernicka-Goetz M, Ciemerych MA, Kubiak JZ, Tarkowski AK, Maro B. Cytostatic factor inactivation is induced by a calcium-dependent mechanism present until the second cell cycle in fertilized but not in parthenogenetically activated mouse eggs. J. Cell Sci. 1995; 108:469-74.
48. Kono T, Jones KT, Bos-Mikich A, Whittingham DG, Carroll J. A cell cycle-associated change in Ca2+ releasing activity leads to the generation of Ca2+ transients in mouse embryos during the first mitotic division. J. Cell Biol. 1996; 132:915-23.
49. FitzHarris G, Larman M, Richards C, Carroll J. An increase in [Ca2+]i is sufficient but not necessary for driving mitosis in early mouse embryos. J. Cell Sci. 2005; 118:4563-75.
50. Groigno L, Whitaker M. An anaphase calcium signal controls chromosome disjunction in early sea urchin embryos. Cell 1998; 92:193-204.
51. Kubiak JZ. Mouse oocytes gradually develop the capacity for activation during the metaphase II arrest. Dev. Biol. 1989; 136:537-45.
52. Fulton BP, Whittingham DG. Activation of mammalian oocytes by intracellular injection of calcium. Nature 1978; 273:149-51.
53. Jones KT. Ca2+ oscillations in the activation of the egg and development of the embryo in mammals. Int J. Dev. Biol. 1998; 42:1-10.
54. Jones KT. Mammalian egg activation: from Ca2+ spiking to cell cycle progression. Reproduction 2005; 130:813-23.
55. Ozil JP. The parthenogenetic development of rabbit oocytes after repetitive pulsatile electrical stimulation. Development 1990; 109:117-27.
56. Ozil JP, Huneau D. Activation of rabbit oocytes: the impact of the Ca2+ signal regime on development. Development 2001; 128:917-28.
57. Ozil JP, Markoulaki S, Toth S, et al. Egg activation events are regulated by the duration of a sustained [Ca2+]cyt signal in the mouse. Dev. Biol. 2005; 282:39-54.
58. Ozil JP, Banrezes B, Toth S, Pan H, Schultz RM. Ca2+ oscillatory pattern in fertilized mouse eggs affects gene expression and development to term. Dev. Biol. 2006; 300:534-44.
59. Toth S, Huneau D, Banrezes B, Ozil JP. Egg activation is the result of calcium signal summation in the mouse. Reproduction 2006; 131:27-34.
60. Bos-Mikich A, Whittingham DG, Jones KT. Meiotic and mitotic Ca2+ oscillations affect cell composition in resulting blastocysts. Dev. Biol. 1997; 182:172-9.
61. Marshall IC, Taylor CW. Two calcium-binding sites mediate the interconversion of liver inositol 1,4,5-trisphosphate receptors between three conformational states. Biochem. J. 1994; 301:591-8.
62. Ozil JP. Role of calcium oscillations in mammalian egg activation: experimental approach. Biophys. Chem. 1998; 72:141-52.
63. Ducibella T, Schultz RM, Ozil JP. Role of calcium signals in early development. Semin. Cell Dev. Biol. 2006; 17:324-32.
64. Verlhac MH, Kubiak JZ, Weber M, et al. Mos is required for MAP kinase activation and is involved in microtubule organization during meiotic maturation in the mouse. Development 1996; 122:815-22.
65. Tunquist BJ, Maller JL. Under arrest: cytostatic factor (CSF)-mediated metaphase arrest in vertebrate eggs. Genes Dev. 2003; 17:683-710.
66. Aoki F, Hara KT, Schultz RM. Acquisition of transcriptional competence in the 1-cell mouse embryo: requirement for recruitment of maternal mRNAs. Mol. Reprod. Dev. 2003; 64:270-4.
67. Atkins CM, Davare MA, Oh MC, Derkach V, Soderling TR. Bidirectional regulation of cytoplasmic polyadenylation element-binding protein phosphorylation by Ca2+/calmodulin-dependent protein kinase II and protein phosphatase 1 during hippocampal long-term potentiation. J. Neurosci. 2005; 25:5604-10.
68. Atkins CM, Nozaki N, Shigeri Y, Soderling TR. Cytoplasmic polyadenylation element binding protein-dependent protein synthesis is regulated by calcium/calmodulin-dependent protein kinase II. J Neurosci. 2004; 24:5193-201.
69. Racki WJ, Richter JD. CPEB controls oocyte growth and follicle development in the mouse. Development 2006; 133:4527-37.
70. Zeng F, Baldwin DA, Schultz RM. Transcript profiling during preimplantation mouse development. Dev. Biol. 2004; 272:483-96.
71. Hamatani T, Carter MG, Sharov AA, Ko MS. Dynamics of global gene expression changes during mouse preimplantation development. Dev. Cell 2004; 6:117-31.
72. Wang QT, Piotrowska K, Ciemerych MA, et al. A genome-wide study of gene activity reveals developmental signaling pathways in the preimplantation mouse embryo. Dev. Cell 2004; 6:133-44.
73. Hudmon A, Schulman H. Structure-function of the multifunctional Ca2+/calmodulin-dependent protein kinase II. Biochem. J. 2002; 364:593-611.
74. De Koninck P, Schulman H. Sensitivity of CaM kinase II to the frequency of Ca2+ oscillations. Science 1998; 279:227-30.