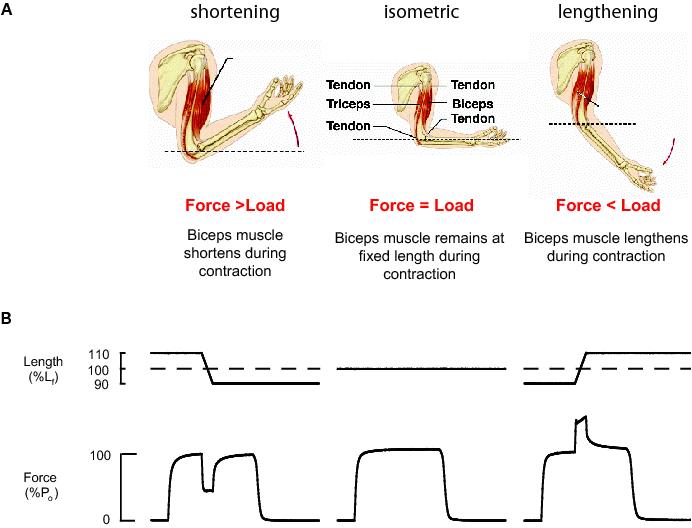
1. For animals of all ages, during activation of skeletal muscles and the subsequent contraction, the balance between the force developed by the muscle and the external load determines whether the muscle shortens, remains at fixed length (isometric), or is lengthened. With maximum activation, the force developed is least during shortening, intermediate when muscle length is fixed, and greatest during lengthening contractions. During lengthening contractions, when force is high, muscles may be injured by the contractions.
2. ‘Frailty’ and ‘failure to thrive’ are most frequently observed in elderly, physically inactive people. A ‘frail’ person is defined as one of small stature, with muscles that are atrophied, weak, and easily fatigued. The condition of ‘failure to thrive’ is typified by a lack of response to well-designed programs of nutrition and physical activity.
3. With aging, skeletal muscle atrophy in humans appears to be inevitable. A gradual loss of muscle fibres begins at approximately age 50 years and continues such that by age 80, ∼50% of the fibres are lost from the limb muscles that have been studied. For both humans and rats, the observation that the timing and magnitude of the loss of motor units is similar to that for muscle fibres, suggests that the mechanism responsible for the loss of fibres and the loss of whole motor units is the same. The degree of atrophy of the fibres that remain is largely dependent on the habitual level of physical activity of the individual.
4. ‘Master athletes’ maintain a high level of fitness throughout their life span. Even among master athletes, performance of marathon runners and weight lifters declines after about 40 years of age, with peak levels of performance decreased by ∼50% by age 80. The success of the master athletes and of previously sedentary elderly who undertake well-designed, carefully administered training programs provide dramatic evidence that age-associated atrophy, weakness, and fatigability can be slowed but not halted.
In animals of all ages and all species, skeletal muscles, when activated by action potentials, may perform three types of contractions.1 If the external load on the muscle is less than the force developed by the fibres within the muscle, the fibres, and consequently the muscle, will shorten. Conversely, if the load is greater than the force developed by the fibres, the fibres will be lengthened, and lastly, if the load and force are equal or if the load is immovable, the fibres will remain “fixed” at the same length, or isometric (Figure 1). Compared with the force developed by a maximally activated skeletal muscle during an isometric contraction, the maximum force developed during a lengthening contraction is 1.5- to 2-fold greater, while the force developed during shortening is always less than the maximum isometric force (Figure 1). The muscle only performs ‘work’ during a shortening contraction and the magnitude of the work performed is a function of the force generated by the muscle and the amount of shortening (Figure 1). Following protocols of repeated isometric or shortening contractions, or stretches without activation, force may be decreased due to fatigue, but recovery of force to control values is fairly rapid and complete and no evidence of injury is observed.2,3 In contrast, the phenomenon of ‘contraction-induced injury’ occurs only after lengthening contractions.2,3 The injury is initiated by the mechanical disruption of individual sarcomeres,4 followed by a sealing off of the focal injury.5 A more severe secondary injury is evidenced several days later due to the invasion of the muscle by inflammatory cells and the generation of free radicals.4,6,7 Injury to skeletal muscle is frequently associated with either a single severe lengthening of a maximally-activated muscle, such as might occur during a fall, or multiple smaller stretches of strongly-activated muscles, as encountered by thigh and knee extensors during a marathon run.4 Complete recovery from the injury caused by a protocol of lengthening contractions requires a period of several weeks, or if severe, even longer.4,6,7 For a given protocol of lengthening contractions, muscles in old animals experience greater injury and following a severe injury, recovery may be incomplete.4 Incomplete recovery from contraction-induced injury may give rise to a permanent loss of muscle mass and force.6,7

Figure 1. The three types of contractions that single fibres, motor units and whole skeletal muscles are able to perform are dependent on the interaction of the force developed by the muscle and the load against which the muscle is attempting to shorten. A shortening contraction occurs when the force is greater than the load. During a shortening contraction, the velocity of shortening is load-dependent with the greater the load the lower the velocity of shortening. During a shortening contraction, a muscle performs ‘work’. An isometric contraction occurs when the force developed by the muscle equals the load, or under conditions when the load is immovable. A lengthening contraction results, when the load on the muscle is greater than the force developed by the muscle. A. The interactions between force and load during each of the three types of contractions. B. Tracings of the displacements initiated by a servo motor lever arm and the forces developed by a maximally activated muscle measured by a force transducer during each of the three types of contractions.
Bortz8 identified the musculoskeletal system as the ‘entry pathway’ for ‘frailty’. Consequently, the structural and functional factors responsible for the physical aspects of frailty are most evident and consequently most effectively investigated directly in skeletal muscles of humans.9-12 Such studies have established unequivocally a 30% to 50% decrease in skeletal muscle mass in both males and females between the ages of 40 and 80 years.12-15 For most elderly people, the decrease in muscle mass12,15 is accompanied by at least an equal, but usually even greater, decrease in strength,12,16,17 and power,18 and an increase in muscle weakness [the strength per unit of cross-sectional area of muscle]19 and fatigability.20,21 The sum total of these effects is that age-related changes in the musculoskeletal system have a significant impact on the everyday activities of the elderly. The impact is even more profound for a ‘frail’ elderly person. For humans, the condition of ‘physical frailty’ is typified by a person of small stature, who displays severe impairments in strength, mobility, balance, and endurance due to muscles that are weak and highly fatigable.22,23 The condition of frailty is observed only rarely in the young, or even middle-aged persons, but is extremely prevalent among the elderly. Commonly cited causes of ‘frailty’ are: genetic, disease, injury, lifestyle, and aging.8 Although the condition of ‘frailty’ is associated with a number of diseases, for the healthy elderly the condition appears to be related primarily to life-style.8 In addition, many frail elderly display the phenomenon of ‘failure to thrive’ wherein the ‘frail elderly person’ does not respond to well-designed programs for conditioning and rehabilitation.24
The conditions of ‘frailty’ and ‘failure to thrive’ constitute two of the most prominent manifestations of the sedentary elderly person. At any age, frailty may be initiated by metabolic or muscle wasting diseases or by a long-term inactive life style,8 but for the younger person the condition is usually reversible. The increased susceptibility to and magnitude of contraction-induced injury constitutes an additional risk factor for the elderly.6,7 Therefore, ‘frailty’ may lead to impaired mobility and balance,25 a higher risk of falling26,27 and increased incidence and severity of contraction-induced injury.4 The condition of frailty is invariably accompanied by declining fitness, health, and quality of life.28 Concomitant with the condition of ‘frailty’, the condition of ‘failure to thrive’ reflects an inability of the frail elderly person to extricate him or herself from the downward spiral of greater and greater frailty even when presented with opportunities to do so.
Muscle mass is a function of the mean volume of the muscle fibres [fibre length multiplied by the fibre cross-sectional area (CSA)] and the number of fibres present in a given skeletal muscle. After maturity, fibre length only changes in conjunction with significant hypertrophy or atrophy of fibres, conditions that initiate decreases and increases in fibre length, respectively.29 Furthermore, although under specific circumstances, branching of fibres may occur,30 the number of fibres in a muscle does not increase.31 Consequently for adults, a change in the mass of a given muscle results primarily from either a change in the CSA of individual fibres or a loss in the number of fibres. In most humans, both factors contribute to the decreased muscle mass with aging, but the proportion of the contribution of each depends on heredity as well as a number of unknown factors in addition to habitual levels of physical activity.15 For many, the atrophy of the skeletal muscles is first noticeable after 40 years of age, and for almost all by 50 years of age.15 Any earlier loss in muscle mass is attributable to a loss in the CSA of individual fibres due to a sedentary life style, since no difference in the number of fibres is observed between 20 and 50 years of age (Figure 2a). Between 50 and 80 years of age, the number of fibres in the large thigh muscle, the vastus lateralis, of men decreased by 50%, from 600,000 fibres to 323,000 fibres (Figure 2a). Although a comparable study has not been undertaken for females, the age-related changes in muscle mass, suggest similar, if not identical, changes occurring.16,18 The loss of fast type 2 fibres appears to be immutable,11,15 but the impact of the fibre loss on muscle mass depends to a substantial degree on the regularity and intensity of the physical activity in which elderly people are engaged. Physical activity impacts directly on the CSA of the fibres that remain, such that rather than atrophying, fibres may maintain CSA,32,33 or even hypertrophy.10,11,34 The tendency is for the mean CSA of the fast type 2 muscle fibres to decrease with aging while slow type 1 fibres tend to maintain their CSA, even in elderly subjects.11,15
The large thigh muscles of humans consist of hundreds of thousands of fibres.15 Despite the substantial sampling problems associated with obtaining small needle-biopsy samples from these large muscles and the impact on the measurements of CSA, absolute and specific forces, and absolute and normalized powers of single permeabilized fibres from the biopsies, these data have proved insightful.32,33,35 Studies of young and elderly persons, both male and female, after carefully designed programs of weight lifting and equally state-of-the-art measures of structure and function of permeabilized single fibres, demonstrate hypertrophy of both slow type 1 and fast type 2 fibres32,35 and on other occasions no change in fibre CSA.33 The discrepancies among these data are likely attributable to differences in the initial fitness level of the individual subjects, the intensity and supervision of the training program, as well as the aforementioned problem of sampling only a few hundred fibres at each biopsy site in the large thigh muscles.32-35
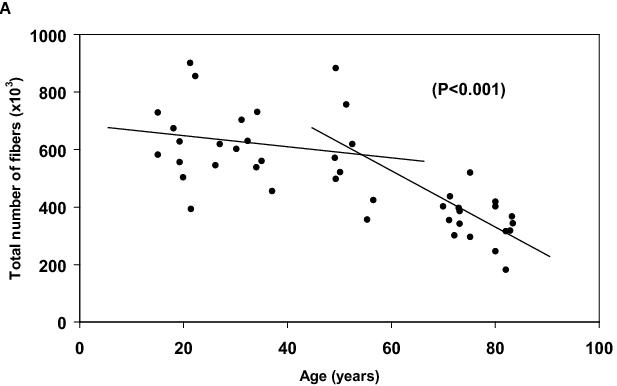
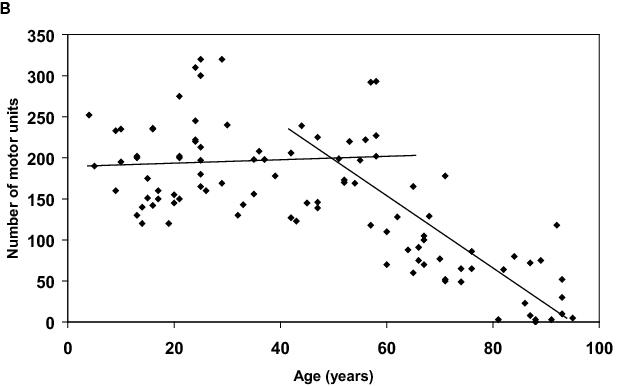
Figure 2. A. The relationship between the total number of fibres in the vastus lateralis muscles and the age of men between 18 to 82 years of age. The average number of fibres in the vastus lateralis muscle does not change between 18 and 50 years of age, but by age 80, the mean number of fibres decreases to 50% of the number for younger men. B. The relationship between the number of motor units in the extensor digitorum brevis muscles and the age of men between the five and 88 years of age. The number of motor units remained constant from 5 years to 50 years of age, but then decreased linearly with a zero intercept at ninety-five years of age.
A decrease in muscle mass and in the number of muscle fibres has been observed in every species in which age-related changes have been studied, which include humans,11,15 rats36-39 and mice.40 Although many of the mechanisms underlying the age-related changes in skeletal muscles remain of unknown origin, progress has been made elucidating the contribution of muscle fibre denervation to the loss of muscle fibres,41 the loss of motor units,42-44 and the remodeling of motor units.45 The major underlying cause of the muscle fibre loss appears to be the loss of motor units (Figure 2b). The phenomenon of a loss of motor units has been reported in muscles of both rats43,45-47 and humans.42,48,49 Although in humans numbers of motor units were measured in small toe42 or thenar48,49 muscles by indirect techniques, whereas the number of fibres in a large thigh muscle of rats were counted directly.15
While observations of age-related changes to skeletal muscles of humans are the most relevant,15,42,48-50 investigations of the underlying mechanisms of age-related changes in skeletal muscles of small, short lived, mammalian species, such as mice20,40 and rats,45 provide many advantages. The advantages of rodent models are clearly evident for those variables for which only less precise, indirect measures are possible in humans. Such variables include: whole muscle mass,40 fibre number,36,39 absolute and specific force of whole muscles,40 absolute power and normalized power of whole muscles,20 and motor unit number, innervation ratio and remodeling.43,45-47 Taking into account the short, two to three year life span of rats, the timing of the onset as well as the rate and magnitude of the loss of motor units in rats,43,47 the relative time course and magnitude of the losses in humans and rats are in exceptionally good agreement for each of the two variables (Figure 2). Furthermore, counts of the number of motor axons innervating skeletal muscles are decreased in both old rats41,46,51 and elderly humans.52 The decreases in the number of motor neurons and in the number of motor units in old compared with adult rats are of similar magnitudes.46 Despite this agreement, no cause-effect relationship between the loss in the number of axons in motor nerves and the loss in the number of motor units or muscle fibres has been established in either rats or human beings.53
In addition to the loss of motor units as a major contributor to muscle atrophy, denervation atrophy of single fibres independent of the motor unit loss is also a possibility.54,55 In rats, the loss of motor units is exclusively from the pool of fast, powerful motor units.45 The loss of the fast motor units leaves some fast fibres within a muscle denervated. Some of these fibres get incorporated into remaining slow motor units by axonal sprouting.55 The result is an increase in the number of muscle fibres in the remaining motor units, i.e. an increase in the innervation ratio.45 Fibres that do not become re-innervated, undergo denervation atrophy and are eventually lost. While the loss of motor units is roughly equivalent between rats43,47 and humans,42,48 the number of muscle fibres lost with aging does not show consistency between these two species. For large limb muscles of humans, a 50% decrease in fibre number occurs by 80 years of age, while hind limb muscles of rats show only a 5% to 10% loss of fibres over a comparable period of their life span.36,39 The similar loss of motor units between the species coupled with the much greater fibre loss from human compared with rodent muscles suggests that in the small muscles of rats, the process of re-innervation is much more effective than in the large muscles of human beings, although this hypothesis has not been tested directly.
For the past quarter of a century, differences of opinion have existed regarding the relative roles in the development of frailty of immutable age-related changes in the structure and function of skeletal muscles15,42,49 and those attributable to a sedentary, low physical activity life style.17,50 For adult men and women, the ‘master athletes’ typify the most physically active in any age group. Throughout their life span, barring injury or illness, the master athlete maintains a high level of fitness and competes in his or her sport or specialized individual event of running, throwing, or weight-lifting. Even among these men and women, performance declines after about 40 years of age. By age 80, the decrease in peak performance is from 35% to 65% for different events (Figure 3). The variability between events is dependent largely on whether an event involves moving the body mass, as in running, or moving a fixed mass as in weight-lifting, shot-putting, or discus throwing. Performance in all events and at all ages has been improved by the advent of ‘plyometric training’.56 ‘Plyometric training’ includes vigorous movements that involve each of the three types of contractions, shortening, isometric and lengthening, as described in the first section. Significant, improvements in muscle mass, strength, power, and endurance have also been achieved in previously sedentary men and women over 75 years of age through participation in conditioning programs that include: (i) participation three times per week on alternative days, (ii) muscle contractions involving each of the three types of contractions, (iii) continuous increments in loading based on improvements in strength, and (iv) a duration of 12 weeks or more.32,33,35,56,57 The improvements in such training programs have been increased considerably by the inclusion of lengthening contractions at >80% of one execution maximum strength, but such programs require the supervision of professional trainers and great care must be exercised not to cause ‘contraction-induced injury’ to participants.6,7,56,57 The success of the master athletes and of previously sedentary elderly, who have undertaken well-designed, carefully administered training programs indicate that the atrophy, weakness, and fatigability usually associated with advancing years, can be slowed. Although the loss in the number of fibres within muscles appears immutable,15 the magnitude of the loss in muscle mass can be ameliorated to some degree by hypertrophy of the fibres that remain.32,35

Figure 3. The performance of Masters athletes for the Marathon run and weight-lifting. The data for the marathon run are taken from the Alan L. Jones website “Age graded running races” 2006. (http://home.stny.rr.com/alanjones/AgeGrade.html) The Masters weightlifting data from IWF Masters Records December 2006 (men’s weight class 85 kg., clean & jerk).
In summary, clearly both immutable changes in skeletal muscle structure and function as well as an increasingly sedentary life style contribute to increasing frailty among the elderly. Young people and even adults appear able to extricate themselves without difficulty from the temporary conditions of frailty induced by physical inactivity, injury, starvation, or illness, whether voluntarily or involuntarily induced.23,50 Although significant gains have been realized in the quality and in the scientific bases for physical conditioning programs specifically for the frail elderly,23,24,58 the phenomenon of the ‘failure to thrive’ for many of the participants continues to be an incomprehensible aspect of even well-designed physical activity programs.8,58 In the United States the estimated cost of physical frailty among elderly people is in the millions of dollars per year, and with the ever increasing numbers of frail elderly, the doubling time is estimated to be 40 years.59 Despite the enormity of the increasing cost and the seriousness of the problem,59 few new insights have been gained and only modest progress has been made toward the provision of successful programs to resolve the conditions of physical frailty,23 or the failure to thrive.8,58
We acknowledge the support of the NIH-NIA grant P01 AG20591 (J.A.F.) and Nathan Shock Center Contractility Core NIA AG13283 (J.A.F.)
Figure 1a. Modified from Vander, Sherman, Luciano Human Physiology, figure 11-31, page 320, McGraw-Hill, 2001. Reproduced with permission of The McGraw-Hill Companies.
Figure 2a. Modified from Journal of the Neurological Sciences, Lexell et al., What is the cause of ageing atrophy? 84: 275-294, figure 6, 1988 with permission from Elsevier BV.
Figure 2b. Adapted from Journal of Neurology, Neurosurgery and Psychiatry, Campbell et al., Physiological changes in ageing muscles. 36:174-182, figure 2, 1973 with permission from BMJ Journals.
1. Faulkner JA. Terminology for contractions of muscles during shortening, while isometric, and during lengthening. J. Appl. Physiol. 2003; 95: 455-9.
2. McCully KK, Faulkner JA. Injury to skeletal muscle fibers of mice following lengthening contractions. J. Appl. Physiol. 1985; 59: 119-26.
3. McCully KK, Faulkner JA. Characteristics of lengthening contractions associated with injury to skeletal muscle fibers. J. Appl. Physiol. 1986; 61: 293-9.
4. Faulkner JA, Brooks SV, Zerba E. Muscle atrophy and weakness with aging: contraction-induced injury as an underlying mechanism. J. Gerontol. 1995; 50A: 124-9.
5. Rader EP, Song W, Van Remmen H, Richardson A, Faulkner JA. Raising the antioxidant levels within mouse muscle fibres does not affect contraction-induced injury. Exp. Physiol. 2006; 91: 781-9.
6. Rader EP, Faulkner JA. Recovery from contraction-induced injury is impaired in weight-bearing muscles of old male mice. J. Appl. Physiol. 2006; 100: 656-61.
7. Rader EP, Faulkner JA. Effect of aging on the recovery following contraction-induced injury in muscles of female mice. J. Appl. Physiol. 2006; 101: 887-92.
8. Bortz WM. A conceptual framework of frailty: a review. J. Gerontol. A. Biol. Sci. Med. Sci. 2002; 57: M283-M288.
9. Aniansson A, Hedberg M, Henning GB, Grimby G. Muscle morphology, enzymatic activity, and muscle strength in elderly men: a follow-up study. Muscle Nerve 1986; 9: 585-91.
10. Grimby G, Saltin B. The ageing muscle. Clin. Physiol. 1983; 3: 209-18.
11. Lexell J. Human aging, muscle mass, and fiber type composition. J. Gerontol. 1995; 50A: 11-6.
12. Young A, Stokes M, Crowe M. The size and strength of the quadriceps muscles of old and young men. Clin. Physiol. 1985; 5: 145-54.
13. Akima H, Kano Y, Enomoto Y, Ishizu M, Okada M, Oishi Y, Katsuta S, Kuno S. Muscle function in 164 men and women aged 20-84 yr. Med. Sci. Sports Exerc. 2001; 33: 220-6.
14. Allen TH, Andersen EC, Langham WH. Total body potassium and gross body composition in relationship to age. J. Gerontol. 1960; 15: 348-57.
15. Lexell J, Taylor CC, Sjostrom M. What is the cause of the ageing atrophy? Total number, size and proportion of different fiber types studied in whole vastus lateralis muscle from 15- to 83-year-old men. J. Neurol. Sci. 1988; 84: 275-94.
16. Frontera WR, Hughes VA, Lutz KJ, Evans WJ. A cross-sectional study of muscle strength and mass in 45- to 78-yr-old men and women. J. Appl. Physiol. 1991; 71: 644-50.
17. Goodpaster BH, Park SW, Harris TB, Kritchevsky SB, Nevitt M, Schwartz AV, Simonsick EM, Tylavsky FA, Visser M, Newman AB. The loss of skeletal muscle strength, mass, and quality in older adults: the health, aging and body composition study. J. Gerontol. A. Biol. Sci. Med. Sci. 2006; 61: 1059-64.
18. Bassey EJ, Fiatarone MA, O'Neill EF, Kelly M, Evans WJ, Lipsitz LA. Leg extensor power and functional performance in very old men and women. Clin. Sci. (Lond) 1992; 82: 321-7.
19. Brooks SV, Faulkner JA. Skeletal muscle weakness in old age: underlying mechanisms. Med. Sci. Sports Exerc. 1994; 26: 432-9.
20. Brooks SV, Faulkner JA. Forces and powers of slow and fast skeletal muscles in mice during repeated contractions. J. Physiol. (Lond) 1991; 436: 701-10.
21. Gandevia SC, Enoka RM, McComas AJ, Stuart DG, Thomas CK. Fatigue: neural and muscular mechanisms. Adv. Exp. Med. Biol. 1995; 384.
22. Fried LP. Conference on the physiologic basis of frailty. Aging (Milano.) 1992; 4: 251-65.
23. Hadley EC, Ory MG, Suzman R, Weindruch R, Fried L. Physical frailty: A treatable cause of dependence in old age. J. Gerontol. 1993; 48: 1-88.
24. Verdery RB. Failure to thrive in the elderly. Clin. Geriatr. Med. 1995; 11: 653-9.
25. Schultz AB. Muscle function and mobility biomechanics in the elderly: an overview of some recent research. J. Gerontol. 1995; 50A: 60-3.
26. Lord SR, Sherrington C, Menz HB. Falls in Older People. Risk Factors and Strategies for Prevention, Cambridge University Press, Cambridge. 2001.
27. Tinetti ME, Williams TF, Mayewski R. Fall risk index for elderly patients based on number of chronic disabilities. Am. J. Med. 1986; 80: 429-34.
28. Baumgartner RN, Koehler KM, Gallagher D, Romero L, Heymsfield SB, Ross RR, Garry PJ, Lindeman RD. Epidemiology of sarcopenia among the elderly in New Mexico. Am. J. Epidemiol. 1998; 147: 755-63.
29. Maxwell LC, Faulkner JA, Hyatt GJ. Estimation of number of fibers in guinea pig skeletal muscles. J. Appl. Physiol. 1974; 37: 259-64.
30. Blaivas M, Carlson BM. Muscle fiber branching--difference between grafts in old and young rats. Mech. Ageing Dev. 1991; 60: 43-53.
31. Gollnick PD, Timson BF, Moore RL, Riedy M. Muscular enlargement and number of fibers in skeletal muscles of rats. J. Appl. Physiol. 1981; 50: 936-43.
32. Trappe S, Williamson D, Godard M, Porter D, Rowden G, Costill D. Effect of resistance training on single muscle fiber contractile function in older men. J. Appl. Physiol. 2000; 89: 143-52.
33. Trappe S, Godard M, Gallagher P, Carroll C, Rowden G, Porter D. Resistance training improves single muscle fiber contractile function in older women. Am. J. Physiol. (Cell) 2001; 281: C398-C406.
34. Lexell J, Taylor CC. Variability in muscle fibre areas in whole human quadriceps muscle: effects of increasing age. J. Anat. 1991; 174: 239-49.
35. Widrick JJ, Stelzer JE, Shoepe TC, Garner DP. Functional properties of human muscle fibers after short-term resistance exercise training. Am. J. Physiol. (Regul. Integr. Comp. Physiol. ) 2002; 283: R408-R416.
36. Daw CK, Starnes JW, White TP. Muscle atrophy and hypoplasia with aging: impact of training and food restriction. J. Appl. Physiol. 1988; 64: 2428-32.
37. Eddinger TJ, Moss RL, Cassens RG. Fiber number and type composition in extensor digitorum longus, soleus, and diaphragm muscles with aging in Fisher 344 rats. J. Histochem. Cytochem. 1985; 33: 1033-41.
38. Hooper AC. Length, diameter and number of ageing skeletal muscle fibres. Gerontology 1981; 27: 121-6.
39. Larkin LM, Kuzon WM, Halter JB. Effects of age and nerve-repair grafts on reinnervation and fiber type distribution of rat medial gastrocnemius muscles. Mech. Ageing Dev. 2003; 124: 653-61.
40. Brooks SV, Faulkner JA. Contractile properties of skeletal muscles from young, adult and aged mice. J. Physiol. (Lond) 1988; 404: 71-82.
41. Ansved T, Larsson L. Quantitative and qualitative morphological properties of the soleus motor nerve and the L5 ventral root in young and old rats. Relation to the number of soleus muscle fibers. J. Neurol. Sci. 1990; 96: 269-82.
42. Campbell MJ, McComas AJ, Petito F. Physiological changes in ageing muscles. J. Neurol. Neurosurg. Psychiatry 1973; 36: 174-82.
43. Cederna PS, Asato H, Gu X, van der MJ, Kuzon WM, Jr., Carlson BM, Faulkner JA. Motor unit properties of nerve-intact extensor digitorum longus muscle grafts in young and old rats. J. Gerontol. A. Biol. Sci. Med. Sci. 2001; 56: B254-B258.
44. McComas AJ, Fawcett PR, Campbell MJ, Sica RE. Electrophysiological estimation of the number of motor units within a human muscle. J. Neurol. Neurosurg. Psychiatry 1971; 34: 121-31.
45. Kadhiresan VA, Hassett CA, Faulkner JA. Properties of single motor units in medial gastrocnemius muscles of adult and old rats. J. Physiol. (Lond) 1996; 493(Pt 2): 543-52.
46. Kanda K, Hashizume K. Changes in properties of the medial gastrocnemius motor units in aging rats. J. Neurophysiol. 1989; 61: 737-46.
47. Sugiura M, Kanda K. Progress of age-related changes in properties of motor units in the gastrocnemius muscle of rats. J. Neurophysiol. 2004; 92: 1357-65.
48. Doherty TJ, Brown WF. The estimated numbers and relative sizes of thenar motor units as selected by multiple point stimulation in young and older adults. Muscle Nerve 1993; 16: 355-66.
49. Doherty TJ, Vandervoort AA, Taylor AW, Brown WF. Effects of motor unit losses on strength in older men and women. J. Appl. Physiol. 1993; 74: 868-74.
50. Holloszy JO. Workshop on Sarcopenia: Muscle Atrophy in Old Age. J. Gerontol. 1995; 50A: 1-161.
51. Ishihara A, Naitoh H, Araki H, Nishihira Y. Soma size and oxidative enzyme activity of motoneurones supplying the fast twitch and slow twitch muscles in the rat. Brain Res. 1988; 446: 195-8.
52. Tomlinson BE, Irving D. The numbers of limb motor neurons in the human lumbosacral cord throughout life. J. Neurol. Sci. 1977; 34: 213-9.
53. Kanda K, Hashizume K, Nomoto E, Asaki S. The effects of aging on physiological properties of fast and slow twitch motor units in the rat gastrocnemius. Neurosci. Res. 1986; 3: 242-6.
54. Andersson AM, Olsen M, Zhernosekov D, Gaardsvoll H, Krog L, Linnemann D, Bock E. Age-related changes in expression of the neural cell adhesion molecule in skeletal muscle: a comparative study of newborn, adult and aged rats. Biochem. J. 1993; 290 ( Pt 3): 641-8.
55. Brown MC, Holland RL, Hopkins WG. Motor nerve sprouting. Annu. Rev. Neurosci. 1981; 4: 17-42.
56. Wilson GJ, Newton RU, Murphy AJ, Humphries BJ. The optimal training load for the development of dynamic athletic performance. Med. Sci. Sports Exerc. 1993; 25: 1279-86.
57. Svantesson U, Grimby G, Thomee R. Potentiation of concentric plantar flexion torque following eccentric and isometric muscle actions. Acta Physiol. Scand. 1994; 152: 287-93.
58. Verdery RB. Failure to thrive in old age: follow-up on a workshop. J. Gerontol. Med. Sci. 1997; 52A: M333-M336.
59. Lopez ME, Zainal TA, Chung SS, Aiken JM, Weindruch R. Oxidative stress and the pathogenesis of sarcopenia. In Sen CK, Packer L and Hanninen O. (eds). Handbook of Oxidants and Antioxidants in Exercise. Elsevier, Oxford. 2000.