Aquaporins (AQPs) are expressed in tissues in which oedema and fluid imbalances are of major concern. In the mammalian brain, AQP4 water channels are localised in astroglial cells at the blood-brain-barrier interface; AQP1 channels are expressed in choroid plexus and serve in cerebral spinal fluid secretion. Potential roles in brain water homeostasis, oedema, angiogenesis, cell migration, development, neuropathological diseases, and cancer, suggest that AQPs are attractive drug targets. AQP1 is well known as a water channel and, under permissive conditions, also functions as a cGMP-gated cation channel; development of therapeutic strategies that involve differential targeting of AQP1 dual ion-and-water channel functions also is of interest. A need for pharmacological agents to dissect the roles of aquaporins in physiological and pathological processes is clear; however, to date little is known regarding the pharmacology of AQP channels. The classic AQP blocker mercury is toxic, and candidate agents such as tetraethylammonium and phloretin lack specificity and potency.
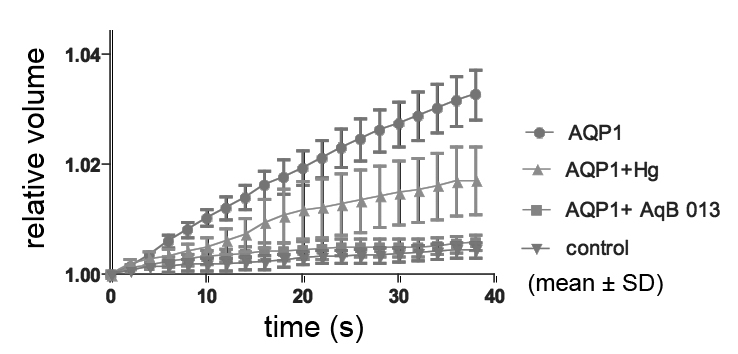
Our research has focused on the structure and function of AQPs, and on novel drug discovery. We have now identified a compound that has promise as a first-in-class lead compound (Migliati, DuBois, Meurice, Fang, Ritter, Flynn & Yool, 2008, submitted). An impressive array of crystal structure data is available for AQPs. From our structural modelling, two intracellular candidate binding sites on AQP4 were predicted from computational modelling. Synthetic derivatives were designed to enhance intracellular delivery and steric fit based on predicted binding in a homology model of AQP4. More than 40 novel derivative compounds were tested on AQP1 and -4 channels expressed in Xenopus oocytes, assayed by videomicroscopic analysis of swelling rates using a double-swell protocol in which each oocyte serves as its own control. One of the series of compounds, AqB013, proved to be an effective blocker of AQPs -1 and -4, with half maximal block (IC50) of ∼20 μM in the oocyte expression system (Figure). AqB013 is more effective in blocking AQP1 than is mercury at the same dose (50 μM). Altered efficacy of block after site-directed mutagenesis of amino acid residues in the candidate binding site supports the identification of the aquaporin channel as the direct molecular target of the blocking effect of AqB013. Collaborative studies using AqB013 in vivo and in vitro are in progress to assess effects of this compound in cell and systems models of pathophysiology. This novel class of pharmacological agents for aquaporins could be valuable as adjuncts in treating oedema and other conditions involving fluid imbalance in aquaporin-expressing tissues.
