1. Cardiovascular diseases most commonly occur in the elderly and are a frequent cause of disability or death. However, the effect of age itself on cardiac function is not well understood.
2. Studies in both human and animal hearts indicate that contractile function is unaffected by age while at rest. However, the ability to increase cardiac contractile force during strenuous activities such as exercise declines with age.
3. Similar findings have been observed in individual ventricular myocytes isolated from aged hearts. When myocytes are stimulated with β-adrenergic agonists or rapid pacing frequencies, aged cells show a much smaller increase in peak contractions and Ca2+ transients than young adult cells. In addition, contractions and Ca2+ transients are prolonged in aged cells compared to younger cells under these conditions.
4. These observations suggest that the age-related decline in cardiac contractile function originates at the cellular level and may reflect modifications in processes involved in excitation-contraction (EC) coupling.
5. Biochemical studies have shown that there are age-related modifications in the expression, regulation and function of a number of proteins essential to EC-coupling in the heart.
6. Functional studies indicate that these changes in EC-coupling proteins disrupt Ca2+ homeostasis and contribute to decrease in peak contraction and prolongation of contraction duration observed in myocytes from aged hearts.
7. This review describes modifications in cardiac contractile function that occur in the ageing heart and evaluates underlying alterations in the EC-coupling pathway that may be responsible for this decline in contractile function in ageing.
Most experimental studies of cardiovascular disease use young adult or even juvenile animals, which are very far removed from the human ages where cardiac pathophysiology becomes clinically important. However, the ageing process affects both the structure and function of the heart. This leads to an age-associated decline in cardiac function, which may predispose older adults towards the development of various cardiovascular diseases. To understand the impact of age on cardiac contractile function, effects of age on mechanisms involved in cardiac excitation-contraction (EC) coupling have been investigated at the level of the individual ventricular myocyte. This brief review describes our current understanding of the impact of age on cardiac contraction and evaluates underlying alterations to the EC coupling pathway that may provoke contractile decline in the ageing heart.
In humans, ageing causes significant changes in the heart, even in the absence of overt cardiovascular disease.1 Left ventricular wall thickness increases with age in the human heart.1 This occurs even though the total number of viable ventricular myocytes actually declines with age because the remaining cells hypertrophy.1,2 Increased accumulation of collagen and fibrous tissue also contributes to the thickening of the ventricle.1 These structural changes are thought to contribute to the reduction in cardiac output and decrease in fractional shortening with age.1,3 Although contractility at rest does not appear to be affected by age,4-6 the ability to increase ejection fraction in response to activities such as exercise declines in older adults.1 Myocardial contraction is also prolonged and relaxation is incomplete in aged individuals compared to younger adults.1,7
The impact of age on cardiac contractile function also has been investigated in various animal models of ageing. Most studies have used mice and rats that are approximately 24 months of age to model aged humans, and compared responses to data obtained in younger adult animals, typically aged 3 to 8 months. Based on survival data, the 50% mortality rate for humans occurs near the age of 85 years,8 while the 50% mortality rate in mice and rats occurs at approximately 24 months of age.9 Therefore, 24-month old rodents have been used as models of 85-year old humans.
Studies in aged rodent models have shown that left ventricular mass increases10 and individual ventricular myocytes are hypertrophied across various species.11-16 Also, the total number of ventricular myocytes decreases with age in the rat heart, likely as a result of an increase in necrotic and apoptotic cell death.17 Contractile function also appears to change with age in animal models. In intact hearts and isolated cardiac tissues, peak contractions are unaffected by age at low stimulation rates, but fractional shortening declines with age and the rates of shortening and re-lengthening are prolonged at more rapid pacing rates.18-27 A similar pattern is seen in in vivo studies when β-adrenergic receptors are stimulated to mimic the effects of exercise.10,28 Aged hearts show a much smaller increase in contractile force in response to β-adrenergic receptor stimulation than their younger counterparts. Since contractions are initiated by an increase in intracellular free Ca2+ at the level of the individual myocyte,29 these observations suggest that contractile decline may result from impaired Ca2+ handling due to age-related modifications in components of EC-coupling.
Cardiac contraction is activated by a transient rise in intracellular free Ca2+. The Ca2+ transient arises when Ca2+ influx, primarily as L-type Ca2+ current (ICaL), triggers Ca2+ release from the sarcoplasmic reticulum (SR)30 through Ca2+ release channels, known as cardiac ryanodine receptors (RyR).31 This process is called Ca2+-induced Ca2+ release (CICR).32-34 Ca2+ release from the SR is proportional to the magnitude of ICaL, and the degree to which this signal is amplified is known as the “gain” of CICR.29 Experimentally, gain is defined as the amount of SR Ca2+ release divided by the amount of trigger Ca2+ influx (total Ca2+ release/ICaL).29 Gain can be modulated by temperature and by SR Ca2+ load35,36 and is thought to play a role in the regulation of cardiac contraction.37,38 During relaxation, most of the released Ca2+ is transported back into the SR by the SR Ca2+ ATPase (SERCA),30 although some Ca2+ is removed from the cell by the Na+/Ca2+ exchanger (NCX) with a minor contribution from the sarcolemmal Ca2+ ATPase.39-41
SR Ca2+ is released in discrete Ca2+ release units called Ca2+ sparks.42-44 These Ca2+ sparks originate near specialized junctions between the sarcolemma (t-tubule or surface membrane) and the SR.45-47 At these junctions, L-type Ca2+ channels and RyRs are located in close proximity.45-47 Ca2+ sparks are thought to represent coordinated Ca2+ release through a cluster of RyRs which become activated by one or more L-type Ca2+ channels.29,45,48 Normally, spontaneous Ca2+ release from one release unit does not activate neighbouring release units, as released Ca2+ diffuses away from adjacent units.29 However, upon depolarisation, many release units are simultaneously activated by ICaL and individual Ca2+ sparks fuse to form the Ca2+ transient.29,49 Spontaneous Ca2+ sparks also can occur in quiescent cells, even in the absence of L-channel openings.42,44,50 Spark frequency increases as SR Ca2+ load increases, which suggests that spontaneous Ca2+ sparks represent a leak pathway for Ca2+ that limits SR Ca2+ content.51 Thus, changes in these unitary Ca2+ release events can impact upon SR Ca2+ content and affect the magnitude of the Ca2+ transient.
Since the cardiac contraction largely reflects the magnitude and time course of the Ca2+ transient,52,53 processes which affect the Ca2+ transient have clearly been of interest in studies of the impact of age on cardiac contractile function. Many studies have focussed on the effect of age on contractions and Ca2+ homeostasis at the level of the individual ventricular myocyte. As described in detail below, ageing results in significant biochemical and physiological changes in the EC-coupling pathway that are believed to be linked to the decline in contractile function in the ageing heart.

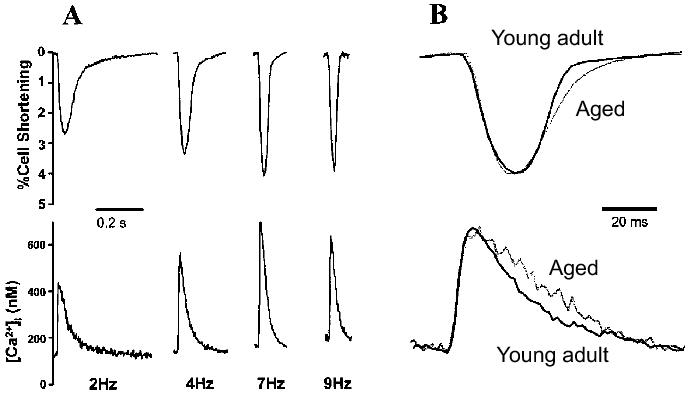
Figure 1. Contractions and Ca2+ transients are prolonged at rapid stimulation rates in ventricular myocytes from aged mice compared to cells from younger animals. A: Contractions and Ca2+ transients were recorded from myocytes isolated from young adult (5 mo) and aged (34 mo) mice. Cells were loaded with fura-2 and field-stimulated at a range of different frequencies at 37°C. Representative recordings of cell shortening (top) and Ca2+ transients bottom) from a young adult myocyte paced at 2, 4, 7, and 9 Hz. B: Examples of contractions (top) and Ca2+ transients (bottom) recorded from young adult and aged myocytes paced at 9 Hz. Responses were normalized to the peak value in each case to show changes in time course. Each recording represents the average of ten original recordings. Reprinted from Lim et al.14 with permission.
A decrease in the ability of individual ventricular myocytes to contract is thought to contribute importantly to the age-associated decline in cardiac contractile function. When myocytes are paced at slow stimulation rates (<1 Hz), peak contractions appear similar in young adult and aged myocytes from mice and rats.13,14,54,55 However at higher stimulation frequencies (> 2 Hz), the extent of cell shortening is lower in aged mouse ventricular myocytes than in young adult cells.14 In addition, re-lengthening is prolonged in cells from aged animals.14 The decline in cardiac contractile function also is reflected in Ca2+ transients recorded from aged rodent myocytes.13,14,56 Figure 1 shows results from a study by Lim et al.14 that compares contractions and Ca2+ transients recorded from ventricular myocytes isolated from young adult and aged mice. In young adult myocytes, peak contractions and Ca2+ transients increase and responses decay more rapidly at higher stimulation frequencies14,56 (Figure 1A). However, aged myocytes produce much smaller increases in peak Ca2+ transients than younger cells when myocytes are paced at rapid rates.14,56 In addition, rates of decay are prolonged in aged cells when compared to younger cells under these experimental conditions14,56 (Figure 1B). These results suggest that the ability of individual ventricular myocytes to contract declines with age. This functional decline at the level of the myocyte would be expected to decrease the overall contractile performance of the ageing heart.
Ventricular myocytes from aged animals also show a decrease in their ability to augment contractions and Ca2+ transients when β-adrenergic receptors are stimulated by catecholamines.13,55,57 In addition, the rates of decay of contractions and Ca2+ transients are prolonged in aged cells when compared to younger cells in the presence of β-adenergic receptor stimulation.13 This may be due to a decrease in the density of β-adrenergic receptors with age,58 though most studies have reported no effect of age on β-adrenergic receptor density.59-61 Recently however, Farrell & Howlett57 reported that a decrease in adenylate cyclase activity with age leads to less cAMP production in ventricular myocytes from aged rats.57 Figure 2 shows that, when ventricular myocytes are stimulated with the non-selective β-agonist isoproterenol, aged cells produce significantly less cAMP than younger cells.57 This decrease in β-adenergic receptor signalling may help explain the loss of sensitivity to catecholamines that occurs with age. These age-associated modifications in contractions, Ca2+ transients and β-adrenergic receptor signalling in various animal models of ageing are summarized in Table 1.
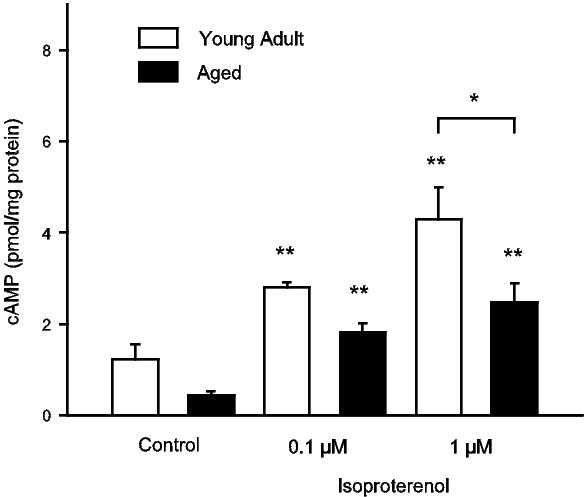
Figure 2. Intracellular cAMP formation in response to increasing concentrations of the non-selective β-adrenergic agonist isoproterenol was significantly greater in ventricular myocytes from young adult rats compared to myocytes from aged rats. Intracellular cAMP formation was measured in isolated intact ventricular myocytes isolated from young adult (3 mo) and aged (24 mo) rats. Intracellular cAMP production in response to administration of isoproterenol was increased over control levels in both young adult and aged myocytes. However, this increase was significantly greater in young adult myocytes when compared to aged cells (** denotes significantly different from age-matched control group, p < 0.05; * denotes significantly different from young adult group, p < 0.05). Reprinted from Farrell & Howlett57 with permission.
Table 1: Age-associated decline in contractile function and β-adrenergic receptor signalling in ventricular myocytes| Parameter | Functional Change | Model/References |
| Contraction | ↓ cell shortening, relaxation slowed, w/ rapid stimulation | mice, 5 vs 24 & 34 mo;14 |
| Contraction | ↓ cell shortening, relaxation slowed w/ β-AR stimulation |
rats, 1-4 vs 24 mo;13
3 mo vs 24 mo55,57 |
| Ca2+ Transient | ↓ peak amplitude, decay slowed, w/ rapid stimulation |
mice, 5 vs 24 & 34 mo;14
mice, 2 vs 20-26 mo56 |
| Ca2+ Transient | ↓ peak amplitude, decay slowed, w/ β-AR stimulation |
mice, 5 vs 24 & 34 mo;14
mice, 2 vs 20-26 mo56 |
| β-AR signalling | ↔ receptor density |
rats, 3 vs 24 mo;59
rats, 3 vs 24 mo;60 rats, 3 vs 24 mo61 |
| ↓ receptor density | rats, 2 vs 24 mo58 | |
| ↓ cAMP production | 3 mo vs 24 mo57 |
To understand mechanisms that suppress contractile function in the ageing heart, a number of studies have investigated the impact of age on proteins involved in EC-coupling. Contractions are slowed in the ageing heart, in part due to changes in myofilament proteins including a shift from α myosin heavy chain to β myosin heavy chain.62 This results in a decrease in myosin ATPase activity in the ageing heart.62 Changes in SERCA2a, the primary SERCA isoform expressed in the heart, also may affect relaxation in the ageing heart. An age-related decrease in the ability of SERCA2a to sequester Ca2+ in the SR may prolong the Ca2+ transient and slow contraction in the ageing heart.20,22,63 Reduced expression of SERCA2a in aged myocytes may be responsible for the slowing of Ca2+ reuptake and prolongation of contraction,63 although this is controversial.62,64,65 Age-related modifications in the regulation of SERCA2a by the endogenous inhibitor phospholamban (PLB) also may affect contraction in the ageing heart. PLB is expressed at elevated levels in aged mice,64 which would be expected to slow Ca2+ reuptake in the ageing heart. Furthermore, phosphorylation of PLB by protein kinase A (PKA) appears to decrease with age.65 As phosphorylation of PLB by PKA normally increases the activity of SERCA2a and speeds relaxation,66 a reduction in phosphorylation of PLB by PKA would slow contraction in the ageing heart. Further, the ability of Ca2+/calmodulin dependent (CaM) kinase to increase SERCA2a activity through phosphorylation is reduced in the ageing heart.65 Together, these findings indicate that SERCA2a activity may be decreased in the ageing heart due to a decline in pump density and an increase in inhibitory regulation. These changes would be expected to slow SR Ca2+ uptake, reduce SR Ca2+ content and prolong the Ca2+ transient, all of which would contribute to a decline in contractile function in the ageing heart.
Other studies have investigated NCX activity in the ageing heart. Results of studies with membrane vesicles or cardiac muscles have been inconsistent, with reports that NCX activity is either decreased64,67 or unchanged68 in the ageing heart. The reasons for these diverse results are unclear, but might be due to differences in membrane preparations or experimental models. However, a recent study of the function of NCX has shown that NCX activity actually increases with age in intact ventricular myocytes.69 As NCX functions primarily to remove Ca2+ from the cell, an increase in NCX activity might help remove Ca2+ from the ageing cardiac myocyte during relaxation. This could serve to compensate, at least in part, for the age-related decline in SERCA2a activity. The increased expression of NCX also may enhance Ca2+ influx during the action potential, which may contribute to the prolonged contraction observed in aged myocytes.7 However, the relative contributions of NCX and SERCA2a to myocardial relaxation in the ageing heart remain uncertain.
Proteins involved in SR Ca2+ release have also been investigated in the ageing heart. Levels of calsequestrin, the major SR Ca2+ binding protein, are similar in young adult and aged hearts.63,64 In contrast, proteins involved in SR Ca2+ release have been shown to change with age. Changes in RyR2, the major RyR expressed in heart, also may affect contractile function in the ageing heart. Some studies have observed an age-associated reduction in RyR2 levels in the ageing heart,18,70 although this has not been reported in all models of ageing.65 In addition, phosphorylation of RyR2 by CaM kinase is reduced in the ageing heart.65 The physiological consequences of phosphorylation of RyR2 remain highly controversial,71 but the decrease in phosphorylation of RyR2 with age may affect SR Ca2+ release in the ageing heart. The major age-associated modifications in components of cardiac EC coupling in different animal models of ageing are illustrated in Table 2.
Table 2: Molecular components of the EC-coupling pathway in the ageing heart| Component | Modification | Model/references |
| Myosin ATPase | ↓ATPase activity | rats, 4 vs 24 mo62 |
| α to β myosin heavy chain | rats, 4 vs 24 mo62 | |
| SERCA2a | ↓ sequestration of Ca2+ | rats, 6-8 vs 24-26 mo;20 rats, 1-2 vs 24 mo;63 rats, 6-8 vs 24-26 mo22 |
| ↓ expression | rats, 1-2 vs 24 mo63 | |
| ↔ expression | rats, 6-8 vs 26-28 mo;65 mice, 5 vs 24 & 34 mo;64 rats, 4 vs 24 mo62 | |
| ↓ phosphorylation | rats, 6-8 vs 26-28 mo65 | |
| PLB | ↑ protein expression | mice, 5 vs 24 & 34 mo64 |
| ↓ phosphorylation | rats, 6-8 vs 26-28 mo65 | |
| NCX | ↑ activity | 14-15 vs 27-31 mo69 |
| ↔ or ↓ activity | mice, 5 vs 24 & 34 mo;64 rats, 6 vs 24 mo;68 rats, 4-6 vs 24-2767 | |
| Calsequestrin | ↔ | rats, 1-2 vs 24 mo;63 mice, 5 vs 24 & 34 mo64 |
| RyR | ↓ Receptor density | rats, 4 vs. 24 mo;18 hamsters, 4 vs 10 mo70 |
| ↔ density | rats, 6-8 vs 26-28 mo65 | |
| ↓ phosphorylation | rats, 6-8 vs 26-28 mo65 |
To determine whether age-related modifications in proteins affect cardiac function, physiological properties of ventricular myocytes have been compared in cells from young adult and aged animals. Some studies have explored the impact of age on spontaneous Ca2+ sparks to establish whether the decrease in RyR2 density and reduction in RyR2 phosphorylation might affect unitary Ca2+ release events. Studies have shown that the frequency of spontaneous Ca2+ sparks increases with age in mouse ventricular myocytes, although the duration of individual Ca2+ sparks declines.72 An increase in spark frequency along with a reduction in Ca2+ spark duration also has been reported in aged rat myocytes, along with a decline in the width and amplitude of Ca2+ sparks.73 These findings suggest that age-associated changes in RyR2 may affect Ca2+ spark activity and Ca2+ spark properties in the ageing heart. A reduction in spark duration and/or spark widths and amplitudes might be expected to disrupt SR Ca2+ release in the ageing heart. In addition, increased Ca2+ spark activity in ageing cardiac myocytes may reduce SR Ca2+ content and disrupt Ca2+ transients in the ageing heart.
Other studies have investigated electrophysiological properties of
intact ventricular myocytes isolated from young adult and aged
animals. Studies have shown that the cardiac action potential, which
initiates the Ca2+ transient, is prolonged in the ageing
heart.12,15,22 On the other hand, resting membrane
potentials in ventricular myocytes and tissues are not affected by
age.12,15,22 Voltage clamp studies have shown that the
increased action potential duration in the ageing heart results from
age-dependent changes in transmembrane currents. Ageing is associated
with a decrease in peak density of the repolarising transient outward
K+ current (ITO) and a modest slowing of the
rate of inactivation of ITO.12,15 Forward-mode
NCX current also increases with age.69 The inactivation
of ICaL also is slowed in aged rat ventricular myocytes
compared to younger cells.12,15 Collectively, the
decrease in ITO, increase in inward NCX current and slowed
inactivation of ICaL can account for the increase in
action potential duration observed in aged ventricular myocytes.22
This increase in action potential duration would be expected to
prolong depolarisation and could slow Ca2+ release and
contractions in aged cardiac myocytes. Interestingly, many previous
studies that reported prolongation of Ca2+ transients and
contractions in ageing myocytes were conducted in field-stimulated
cells, where Ca2+ release and contractions were activated
by action potentials.13,14,55 Thus, it is possible that
the age-related increase in action potential duration contributes to
the slowed contractions and prolonged Ca2+ release
reported in these studies.13,14,55 Key findings of
functional studies of cardiac EC coupling in rodent models of ageing
are summarized in Table 3 and illustrated in Figure 3.
Table 3: Functional studies of EC coupling in aged ventricular myocytes
| Parameter | Functional Change | Model/references |
| Ca2+ Sparks | ↑ spontaneous spark frequency | mice, 5 vs 24 mo;72 rats, 6 vs 24 mo73 |
| ↓ spark duration | mice, 5 vs 24 mo;72 rats, 6 vs 24 mo73 | |
| ↓ spark width | rats, 6 vs 24 mo73 | |
| ↓ spark amplitude | rats, 6 vs 24 mo73 | |
| Action Potential | ↑ duration | rats, 6-8 vs 24-26 mo;22 rats, 2-3 vs 24-25 mo;12 rats, 6 vs 27+ mo15 |
| RMP | ↔ RMP | rats, 6-8 vs 24-26 mo;22 rats, 2-3 vs 24-25 mo;12 rats, 6 vs 27+ mo15 |
| ITO | ↓ peak current density | rats, 2-3 vs 24-25 mo12 |
| ↓ rate of inactivation | rats, 6 vs 27+ mo15 | |
| NCX | ↑ forward mode current | rats, 14-15 vs 27-31 mo69 |
| ICaL | ↓ rate of inactivation | rats, 6 vs 27+ mo;15 rats, 2-3 vs 24-25 mo;12 mice, 7 vs 24 mo75 |
| ↓ peak current density | rats, 6 vs 27+ mo;15 mice, 7 vs 24 mo75 | |
| ↓ Ca2+ channel density | hamsters, 4 vs 20 mo74 |
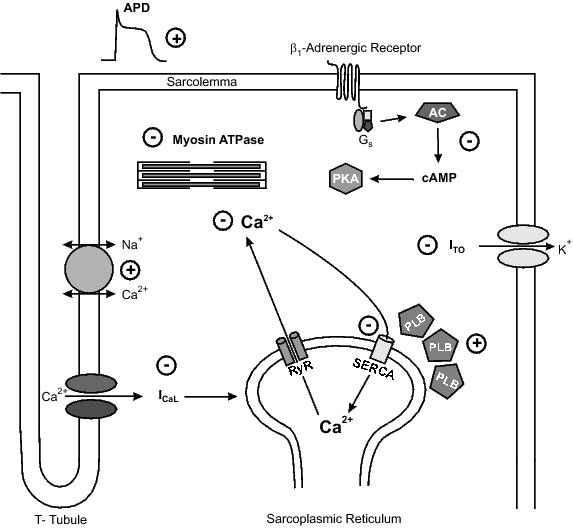
Figure 3. Changes in major components of cardiac excitation-contraction coupling in the ageing heart. In the ageing heart, action potential duration, contraction duration, and Ca2+ decay rate are all prolonged. These changes occur as a result of reduced Ca2+ influx, reduced Ca2+ release, and depressed cardiac contractile efficiency, due to a decline in expression or activity, of proteins involved in cardiac EC coupling. An age-related decrease in the effect of β-adenergic stimulation, due to a reduction in cAMP production in the ageing heart also are seen. Alterations in these mechanisms are thought be responsible for the cardiac contractile decline observed in ageing heart.
Studies have shown that peak density of ICaL declines with age in rat ventricular myocytes, at least when experiments are conducted under physiological conditions.15 Receptor binding studies also have shown that the density of dihydropyridine receptors (L-type Ca2+ channels) declines with age, although properties of these channels are unchanged.74 As ICaL is the predominant trigger for CICR, the observed decrease in ICaL may account for the reduction in SR Ca2+ release and decrease in the size of contractions reported previously in aged myocytes.13,14,56 However, these previous studies of contractile function in ventricular myocytes did not measure ICaL together with contractions and/or SR Ca2+ release. A recent study used voltage clamp techniques to directly investigate effects of age on contractions, Ca2+ transients and transmembrane currents.75 With voltage clamp techniques, the duration and magnitude of depolarisation can be controlled and variations in action potential duration can be eliminated. Under these conditions, the amplitudes of contractions and Ca2+ transients are smaller in ventricular myocytes from aged mice (∼24 mo) compared to responses in cells from young adult animals (5-6 mo).75 However, this study also showed that the age-related decline in the size of contractions and Ca2+ transients occurred only in myocytes from male animals and not in cells from female animals.75 These findings suggest that age-related changes in cardiac EC-coupling may be more prominent in myocytes from males than in cells from females. Consistent with this idea, another study in ventricular myocytes from female sheep reported no evidence of contractile decline with age.16 However, most other studies of the impact of age on cardiac EC-coupling have used hearts and myocytes from male animals only or the sex of the animals has not been specified. As a result, most changes considered "typical" of the ageing process reflect observations in hearts and myocytes from male animals. This is an intriguing finding that suggests the impact of age on cardiac EC coupling is markedly affected by the sex of the animal. Further research considering sex as a variable may improve understanding of how the heart changes with age in men and women, and may help explain sex differences in the expression of various cardiovascular diseases.
Age-related modifications in cardiac structure and function contribute to the decline in cardiac contractile function associated with ageing. Although this is not evident at rest, stimuli that augment contractile function are less effective in aged hearts than in young adult hearts. At the cellular level, contractions are smaller and slower in aged myocytes than in younger cells when myocytes are exposed to rapid pacing rates or β-adrenergic agonists. Evidence suggests that this decline in contractile function originates from modifications in the expression, regulation, and function of proteins associated with myocardial Ca2+ handling. An age-related reduction in the density and activity of RyRs may contribute to this decline. A decrease in SERCA expression, activity and function as well as an increase in SERCA inhibition by PLB also could play a crucial role in slowing contraction. A decrease in ITO, increase in inward NCX current and prolongation of the inactivation of ICaL may increase action potential duration and slow contraction in aged ventricular myocytes. In addition, reduced L-type Ca2+ channel density and a decrease in peak ICaL may suppress CICR, which would depress cardiac contraction. Further investigation of these age-related changes should help explain why cardiac contractile function declines with age. Studies in animals of both sexes may reveal important sex differences in the effects of age in the heart, which might help explain why men and women develop different heart diseases later in life.
The authors express their appreciation to Peter Nicholl for assistance with the figures. This work was supported in part by grants from the Canadian Institutes for Health Research and the Heart and Stroke Foundation of Nova Scotia. Elias Fares is supported by a graduate scholarship from the Nova Scotia Health Research Foundation.
1. Lakatta EG, Levy D. Arterial and cardiac aging: major shareholders in cardiovascular disease enterprises: Part II: the aging heart in health: links to heart disease. Circulation 2003; 107: 346-354.
2. Olivetti G, Melissari M, Capasso JM, Anversa P. Cardiomyopathy of the aging human heart. Myocyte loss and reactive cellular hypertrophy. Circ. Res. 1991; 68(6): 1560-8.
3. Bernhard D, Laufer G. The aging cardiomyocyte: a mini-review. Gerontology 2008; 54(1): 24-31.
4. Fleg JL, O'Connor F, Gerstenblith G, Becker LC, Clulow J, Schulman SP, et al. Impact of age on the cardiovascular response to dynamic upright exercise in healthy men and women. J. Appl. Physiol. 1995; 78: 890-900.
5. Najjar SS, Schulman SP, Gerstenblith G, Fleg JL, Kass DA, O'Connor F, et al. Age and gender affect ventricular-vascular coupling during aerobic exercise. Am. Coll. Cardiol. 2004; 44: 611-7.
6. Ruan Q, Nagueh SF. Effect of age on left ventricular systolic function in humans: a study of systolic isovolumic acceleration rate. Exp. Physiol. 2005; 90: 527-34.
7. Lakatta EG, Sollott SJ. Perspectives on mammalian cardiovascular aging: humans to molecules. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2002; 132: 699-721.
8. Grundy EMD. The Epidemiology of aging. In: Tallis R, Fillit H (eds). Brocklehurst’s textbook of geriatric medicine and gerontology, 6th edition. Churchill Livingstone, London. 2003; 4-20.
9. Turturro A, Witt WW, Lewis S, Hass BS, Lipman RD, Hart RW. Growth curves and survival characteristics of the animals used in the biomarkers of aging program. J. Gerontol. Biol. Sci. 1999; 54A: B492–B501.
10. Hacker TA, McKiernan SH, Douglas PS, Wanagat J, Aiken JM. Age-related changes in cardiac structure and function in Fischer 344 x Brown Norway hybrid rats. Am. J. Physiol. 2006; 290: H304-11.
11. Fraticelli A, Josephson R, Danziger R, Lakatta E, Spurgeon H. Morphological and contractile characteristics of rat cardiac myocytes from maturation to senescence. Am. J. Physiol. 1989; 257: H259-65.
12. Walker KE, Lakatta EG, Houser SR. Age associated changes in membrane currents in rat ventricular myocytes. Cardiovasc. Res. 1993; 27: 1968-77.
13. Xiao RP, Spurgeon HA, O'Connor F, Lakatta EG. Age-associated changes in β-adrenergic modulation on rat cardiac excitation-contraction coupling. J. Clin. Invest. 1994; 94: 2051-9.
14. Lim CC, Apstein CS, Colucci WS, Liao R. Impaired cell shortening and relengthening with increased pacing frequency are intrinsic to the senescent mouse cardiomyocyte. J. Mol. Cell. Cardiol. 2000; 32: 2075-82.
15. Liu SJ, Wyeth RP, Melchert RB, Kennedy RH. Aging-associated changes in whole cell K+ and L-type Ca2+ currents in rat ventricular myocytes. Am. J. Physiol. 2000; 279: H889-900.
16. Dibb KM, Rueckschloss U, Eisner DA, Isenberg G, Trafford AW. Mechanisms underlying enhanced cardiac excitation contraction coupling observed in the senescent sheep myocardium. J. Mol. Cell. Cardiol. 2004; 37: 1171-81.
17. Kajstura J, Cheng W, Sarangarajan R, Li P, Li B, Nitahara JA, et al. Necrotic and apoptotic myocyte cell death in the aging heart of Fischer 344 rats. Am. J. Physiol. 1996; 271: H1215-28.
18. Assayag P, Charlemagne D, Marty I, de Leiris J, Lompré AM, Boucher F, et al. Effects of sustained low-flow ischemia on myocardial function and calcium-regulating proteins in adult and senescent rat hearts. Cardiovasc. Res. 1998; 38: 169-80.
19. Lakatta EG, Gerstenblith G, Angell CS, Shock NW, Weisfeldt ML. Diminished inotropic response of aged myocardium to catecholamines. Circ. Res. 1975; 36: 262-9.
20. Froehlich JP, Lakatta EG, Beard E, Spurgeon HA, Weisfeldt ML, Gerstenblith G. Studies of sarcoplasmic reticulum function and contraction duration in young adult and aged rat myocardium. J. Mol. Cell. Cardiol. 1978; 10: 427-438.
21. Capasso JM, Malhotra A, Remily RM, Scheuer J, Sonnenblick EH. Effects of age on mechanical and electrical performance of rat myocardium. Am. J. Physiol. 1983; 245: H72-81.
22. Wei JY, Spurgeon HA, Lakatta EG. Excitation-contraction in rat myocardium: alterations with adult aging. Am. J. Physiol. 1984; 246: H784-791.
23. Orchard CH, Lakatta EG. Intracellular calcium transients and developed tension in rat heart muscle. A mechanism for the negative interval-strength relationship. J. Gen. Physiol. 1985; 86: 637-651.
24. Anversa P, Capasso JM, Puntillo E, Sonnenblick EH, Olivetti G. Morphometric analysis of the infarcted heart. Pathol. Res. Pract. 1989; 185: 544-50.
25. Rozenberg S, Tavernier B, Riou B, Swynghedauw B, Page CL, Boucher F, et al. Severe impairment of ventricular compliance accounts for advanced age-associated hemodynamic dysfunction in rats. Exp. Gerontol. 2006; 41: 289-95.
26. Hano O, Bogdanov KY, Sakai M, Danziger RG, Spurgeon HA, Lakatta EG. Reduced threshold for myocardial cell calcium intolerance in the rat heart with aging. Am. J. Physiol. 1995; 269: H1607-1612.
27. Schmidt U, del Monte F, Miyamoto MI, Matsui T, Gwathmey JK, Rosenzweig A, et al. Restoration of diastolic function in senescent rat hearts through adenoviral gene transfer of sarcoplasmic reticulum Ca2+-ATPase. Circulation 2000; 101: 790-6.
28. Lieber SC, Qiu H, Chen L, Shen YT, Hong C, Hunter WC, et al. Cardiac dysfunction in aging conscious rats: altered cardiac cytoskeletal proteins as a potential mechanism. Am. J. Physiol. 2008; 295: H860-6.
29. Bers DM. Calcium cycling and signaling in cardiac myocytes. Ann. Rev. Physiol. 2008; 70: 23-49.
30. Bers DM. Cardiac excitation-contraction coupling. Nature 2002; 415: 198-205.
31. Meissner G. Molecular regulation of cardiac ryanodine receptor ion channel. Cell Calcium 2004; 35: 621-8.
32. Fabiato A. Rapid ionic modifications during the aequorin-detected calcium transient in a skinned canine cardiac Purkinje cell. J. Gen. Physiol. 1985; 85: 189-246.
33. Fabiato A. Simulated calcium current can both cause calcium loading and in and trigger calcium release from the sarcoplasmic reticulum of a skinned canine cardiac Purkinje cell. J. Gen. Physiol. 1985; 85: 291-320.
34. Fabiato A. Time and calcium dependence of activation and inactivation of calcium-induced release of calcium from the sarcoplasmic reticulum of a skinned canine cardiac Purkinje cell. J. Gen. Physiol. 1985; 85: 247-290.
35. Shutt RH, Howlett SE. Hypothermia increases the gain of excitation-contraction coupling in guinea pig ventricular myocytes. Am. J. Physiol. 2008; 295: C692-700.
36. Ginsburg KS, Bers DM. Modulation of excitation-contraction coupling by isoproterenol in cardiomyocytes with controlled SR Ca2+ load and Ca2+ current trigger. J. Physiol. 2004; 556: 463-80.
37. Sjaastad I, Wasserstrom JA, Sejersted OM. Heart failure - a challenge to our current concepts of excitation-contraction coupling. J. Physiol. 2003; 546: 33-47.
38. Altamirano J, Bers DM. Voltage dependence of cardiac excitation-contraction coupling: unitary Ca2+ current amplitude and open channel probability. Circ. Res. 2007; 101: 590-7.
39. Periasamy M, Bhupathy P, Babu GJ. Regulation of sarcoplasmic reticulum Ca2+ ATPase pump expression and its relevance to cardiac muscle physiology and pathology. Cardiovasc. Res. 2008; 77: 265-73.
40. Reuter H, Pott C, Goldhaber JI, Henderson SA, Philipson KD, Schwinger RH. Na+-Ca2+ exchange in the regulation of cardiac excitation-contraction coupling. Cardiovasc. Res. 2005; 67: 198-207.
41. Eisner DA, Sipido KR. Sodium calcium exchange in the heart: necessity or luxury? Circ. Res. 2004; 95: 549-51.
42. Cheng H, Lederer WJ, Cannell MB. Calcium sparks: elementary events underlying excitation-contraction coupling in heart muscle. Science 1993; 262: 740-44.
43. Lopez-Lopez JR, Shacklock PS, Balke CW, Wier WG. Local, stochastic release of Ca2+ in voltage-clamped rat heart cells: visualization with confocal microscopy. J. Physiol. 1994; 480: 21-9.
44. Santana LF, Cheng H, Gomez AM, Cannell MB, Lederer WJ. Relation between the sarcolemmal Ca2+ current and Ca2+ sparks and local control theories for cardiac excitation-contraction coupling. Circ. Res. 1996; 78: 166-171.
45. Inoue M, Bridge JH. Variability in couplon size in rabbit ventricular myocytes. Biophys. J. 2005; 89: 3102-10.
46. Bootman MD, Higazi DR, Coombes S, Roderick HL. Calcium signalling during excitation-contraction coupling in mammalian atrial myocytes. J. Cell Sci. 2006; 119: 3915-25.
47. Scriven DR, Klimek A, Lee KL, Moore ED. The molecular architecture of calcium microdomains in rat cardiomyocytes. Ann. N.Y. Acad. Sci. 2002; 976: 488-99.
48. Wang SQ, Song LS, Lakatta EG, Cheng H. Ca2+ signalling between single L-type Ca2+ channels and ryanodine receptors in heart cells. Nature 2001; 410: 592-6.
49. Guatimosim S, Dilly K, Santana LF, Saleet Jafri M, Sobie EA, Lederer WJ. Local Ca2+ signaling and EC coupling in heart: Ca2+ sparks and the regulation of the [Ca2+]i transient. J. Mol. Cell. Cardiol. 2002; 34: 941-50.
50. Ferrier GR, Smith RH, Howlett SE. Calcium sparks in mouse ventricular myocytes at physiological temperature. Am. J. Physiol. 2003; 285: H1495-505.
51. Satoh H, Blatter LA, Bers DM. Effects of [Ca2+]i, SR Ca2+ load, and rest on Ca2+ spark frequency in ventricular myocytes. Am. J. Physiol. 1997; 272: H657-668.
52. Bers DM. Excitation-Contraction Coupling and Cardiac Contractile Force, 2nd edn. Kluwer Academic Publishers, Dordrecht. 2001.
53. Endoh M. Signal transduction and Ca2+ signaling in intact myocardium. J. Pharmacol. Sci. 2006; 100: 525-37.
54. Capasso JM, Fitzpatrick D, Anversa P. Cellular mechanisms of ventricular failure: myocyte kinetics and geometry with age. Am. J. Physiol. 1992; 262: H1770-81.
55. Farrell SR, Howlett SE. The effects of isoproterenol on abnormal electrical and contractile activity and diastolic calcium are attenuated in myocytes from aged Fischer 344 rats. Mech. Ageing Dev. 2007; 128: 566-73.
56. Isenberg G, Borschke B, Rueckschloss U. Ca2+ transients of cardiomyocytes from senescent mice peak late and decay slowly. Cell Calcium 2003; 34: 271-80.
57. Farrell SR, Howlett SE. The age-related decrease in catecholamine sensitivity is mediated by β-adrenergic receptors linked to a decrease in adenylate cyclase activity in ventricular myocytes from male Fischer 344 rats. Mech. Ageing Dev. 2008; 129: 735-44.
58. Xiao RP, Tomhave ED, Wang DJ, Ji X, Boluyt MO, Cheng H, et al. Age-associated reductions in cardiac β1- and β2-adrenergic responses without changes in inhibitory G proteins or receptor kinases. J. Clin. Invest. 1998; 101: 1273–1282.
59. Raitt MH, Tamblyn C, Allen JM, Abrass IB. β-adrenergic receptor downregulation and recovery in heart and lung in the Fischer 344 rat: effects of aging and correlation with mRNA levels. J. Gerontol. 1995; 50: B213–B217.
60. Scarpase PJ. Decreased β-adrenergic responsiveness during senescence. Fed. Proc. 1986; 45: 51–54.
61. Abrass IB, Davis JL, Scarpace PJ. Isoproterenol responsiveness and myocardial β-adrenergic receptors in young and old rats. J. Gerontol. 1982; 37: 156–160.
62. Buttrick P, Malhotra A, Factor S, Greenen D, Leinwand L, Scheuer J. Effect of aging and hypertension on myosin biochemistry and gene expression in the rat heart. Circ. Res. 1991; 68: 645-52.
63. Lompré AM, Lambert F, Lakatta EG, Schwartz K. Expression of sarcoplasmic reticulum Ca2+-ATPase and calsequestrin genes in rat heart during ontogenic development and aging. Circ. Res. 1991; 69: 1380-8.
64. Lim CC, Liao R, Varma N, Apstein CS. Impaired lusitropy-frequency in the aging mouse: role of Ca2+-handling proteins and effects of isoproterenol. Am. J. Physiol. 1999; 277: H2083-2090.
65. Xu A, Narayanan N. Effects of aging on sarcoplasmic reticulum Ca2+ cycling proteins and their phosphorylation in rat myocardium. Am. J. Physiol. 1998; 275: H2087-94.
66. Rodriguez P, Kranias EG. Phospholamban: a key determinant of cardiac function and dysfunction. Arch. Mal. Coeur. Vaiss. 2005; 98(12): 1239-43.
67. Heyliger CE, Prakash AR, McNeill JH. Alterations in membrane Na+-Ca2+ exchange in the aging myocardium. Age 1988; 11: 1-6.
68. Abete P, Ferrara N, Cioppa A, Ferrara P, Bianco S, Calabrese C, et al. The role of aging on the control of contractile force by Na+-Ca2+ exchange in rat papillary muscle. J. Gerontol. A Biol. Sci. Med. Sci. 1996; 51: M251-9.
69. Mace LC, Palmer BM, Brown DA, Jew KN, Lynch JM, Glunt JM, et al. Influence of age and run training on cardiac Na+/Ca2+ exchange. J. Appl. Physiol. 2003; 95: 1994-2003.
70. Nicholl PA, Howlett SE. Sarcoplasmic reticulum calcium release channels in ventricles of older adult hamsters. Can. J. Aging 2006; 25: 107-13.
71. Györke S, Carnes C. Dysregulated sarcoplasmic reticulum calcium release: potential pharmacological target in cardiac disease. Pharmacol. Ther. 2008; 119: 340-54.
72. Howlett SE, Grandy SA, Ferrier GR. Calcium spark properties in ventricular myocytes are altered in aged mice. Am. J. Physiol. 2006; 290: H1566-74.
73. Zhu X, Altschafl BA, Hajjar RJ, Valdivia HH, Schmidt U. Altered Ca2+ sparks and gating properties of ryanodine receptors in aging cardiomyocytes. Cell Calcium 2005; 37: 583-91.
74. Howlett SE, Nicholl PA. Density of 1,4-dihydropyridine receptors decreases in the hearts of aging hamsters. J. Mol. Cell. Cardiol. 1992; 24: 885-94.
75. Grandy SA, Howlett SE. Cardiac excitation-contraction coupling is altered in myocytes from aged male mice but not in cells from aged female mice. Am. J. Physiol. 2006; 291: H2362-70.