1. Here we review past work that has studied the composition of luminal fluid in organs, with a focus on measures of calcium and pH in the exocrine glands. This luminal environment is “external” to the mammalian body and is not subject to the usual mechanisms of homeostatic control. Instead it is controlled by the behaviour of the cells that line the lumen.
2. We discuss the likely possibility that rapid and local changes in calcium and pH occur within microdomains in the lumen. Further we present preliminary evidence, using live-cell imaging of intact pancreatic fragments that support the idea that pH changes do occur. Our evidence indicates that exocytosis of secretory granules in pancreatic acinar cells leads to a loss of protons from the granule and a subsequent local acidification of the lumen.
3. These changes in luminal composition are placed in the context of diseases of the pancreas, such as cystic fibrosis and pancreatitis, both of which are known to result in perturbations of luminal fluid composition.
We are used to thinking of the extracellular environment surrounding mammalian cells as a homeostatically controlled environment which, under normal circumstances, shows only moderate changes in ion composition. However the luminal fluid environment in many organs effectively lies “outside” the body. It is a product of the cells of the organ and is not directly controlled by whole-body homeostasis. A good example of a hollow ducted organ is the pancreas where the luminal fluid outflow from the exocrine part of the pancreas is generated through the coordinated secretory and absorptive activities of the exocrine epithelial cells. We know very little about the composition of this luminal fluid environment, and in particular have no knowledge of possible local and dynamic changes. This paper discusses past work and shows an example of the recent methods we are using in our studies of the luminal microenvironment.
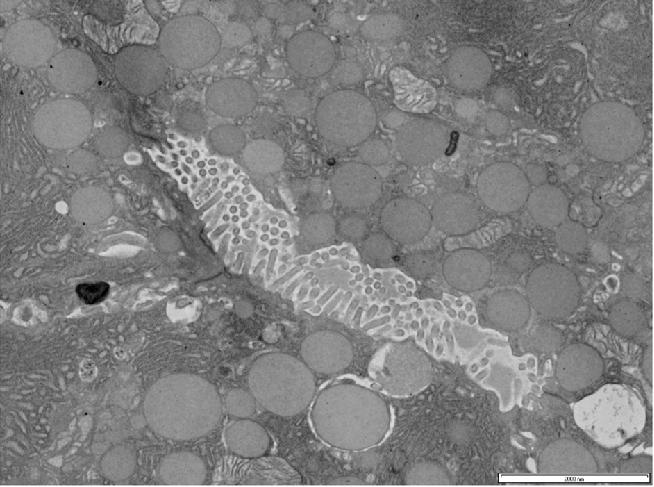
In vivo the exocrine gland lumen is a low-volume, complex structure with many microvilli projecting into the space from the surrounding epithelial cells1,2 that would act as a physical barrier to free diffusion (Figure 1). So although intralumenal fluid has been analyzed as it exits the gland3 or by sampling along the ducts4-6 these methods miss possible local and fast changes in luminal fluid composition.

Figure 1. Transmission electron micrograph of the acinar lumen in end-pieces of exocrine mouse pancreas. This section is taken at the terminal secretory endpiece with the lumen defined by the surrounding acinar cells. The lumen in not an empty space but is a complex small volume with many protruding microvilli and convoluted cell boundaries. Scale bar 2μm.
Looking at other hollow organs, where a mucous layer is present, a luminal microenvironment is also created here. The lungs are bounded on one side by the apical cell membrane and on the other side by air, leaving an aqueous mucous layer ∼30μm deep.7 Within this layer ion concentrations are under the control of the surface epithelial cells8 and likely to be part of a process of regulation. For example, in the cervix, the pH within the mucous lining is under the regulation of the oestrus cycle9 and is thought to be governed by an apically located proton pump the V-ATPase. In the male reproductive system a luminal acid environment in the epididymis is critical to normal function, immobilizing the sperm in this region as they mature.10 Once again it is thought to be controlled by apical V-ATPase activity that is part of a complex regulation that likely includes other apical transporters like the cystic fibrosis transmembrane regulator (CFTR).11
In all exocrine glands the secretory outflow exits the gland through a duct (Figure 2). Upstream the duct branches multiple times, each branch lined by a single layer of duct cells. Further upstream the duct branching continues with divisions getting finer and finer and terminating in a closed-ended lumen at the secretory end-pieces that are surrounded by the enzyme-secreting acinar cells. This tree like ductal structure is generated by tight junction links between neighbouring epithelial cells. The contents of the lumen are therefore separated from the main organ and entirely created by the characteristic permeabilities of the tight junctions and the action of ion channels and pumps on the cells’ apical membranes.1
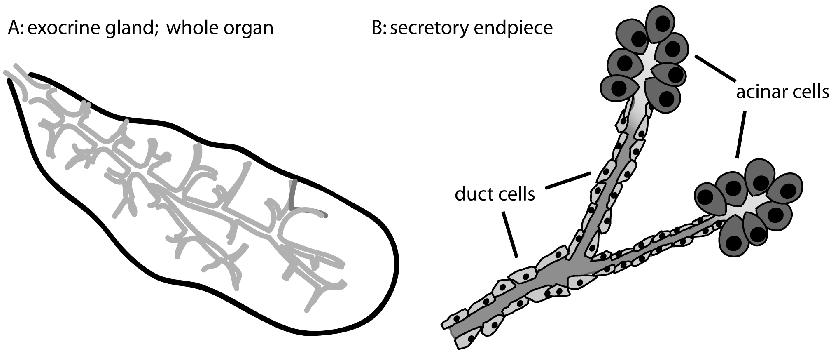
Figure 2. Anatomy of exocrine glands. A. shows the branching duct system that runs through the organ. B. The ducts end at secretory endpieces that are closed and terminate with the secretory acinar cells. The luminal environment is entirely generated by the duct and acinar cells and the nature of the intercellular barrier.
At a relatively low spatial resolution there is evidence from micropuncture studies to support regional differences in the composition of luminal fluid.4,5 In the salivary glands it has been shown that the so-called primary output, largely that of the acinar cells and measured at the intercalated ducts, is isotonic with the serum.12,13 However, the final salivary output is hypotonic to the serum with measured decreases in chloride, potassium and sodium12 with Young (on a CJ Martin Travelling Fellowship) and Schogel13 showing that the ion changes occur in the major ducts of the salivary gland. We now know that CFTR is highly enriched on the duct cell apical membrane9 and is a key player in a multi-step process of chloride-bicarbonate exchange that reduces luminal chloride concentrations in the duct in exchange for cellular bicarbonate.14,15
Two important ions, known to play a signalling role in the extracellular environment, are calcium and protons. While it is known that they are part of the composition of the lumenal fluid their regulation and any physiological consequences of their presence remain poorly understood.
Extracellular calcium in the serum is homeostatically regulated at a concentration of around 2 mM. But in the exocrine gland lumen, the calcium concentration measured statically, has been described as anywhere from 400 nM and 2 mM.16 Ceccarelli et al.17 measured the calcium concentration in the outflow from the whole pancreas and demonstrated a differential contribution of the cell types, with duct cells providing relatively little calcium compared to acinar cells. Very little is known about the mechanisms moving calcium into and out of the pancreatic lumen except that the secretory granules of the acinar cells contain a high calcium concentration17-19 (with estimates up to 200 mM total calcium19) that, on granule fusion, would be released into the lumen potentially promoting large local changes in extracellular calcium.20 The other source of luminal calcium is the possible outflow from the cell and across the lumenal membrane due to the activity of the plasma membrane calcium ATPase (PMCA) which has been shown to be directly coupled to the oscillatory changes in the agonist-evoked intracellular calcium concentration.21 In order for calcium to appear in the lumen it would have to be specifically moved across the apical cell membrane. It is not clear which PMCA isoform might be located on the apical membrane but immunolocalization experiments show PMCA4 as a possible candidate.22
In the pancreas both an apical PMCA and granule fusion could act in consort to promote increases in luminal calcium. These changes in luminal calcium concentration might target the G-protein coupled calcium-receptor (CaR – which is sensitive to changes in extracellular calcium) known to be present in the apical membrane of acinar and duct cells.23 Activation of the CaR can be coupled to elevations of intracellular calcium, decreases in cAMP, as well as the activation of various kinases.24 Stimulation of the CaR of the acinar cells in an autocrine manner may therefore be an important positive or negative feedback regulator of acinar cell function.25 Here it is useful to mention recent work on cell behaviour in stomach pits where the CaR is found on the apical domain26 and where a local feedback mechanism of calcium exit via the PMCA is sufficient to activate the CaR and stimulate cellular responses.27
Increased lumenal calcium might also affect granule fusion dynamics which have been shown, in other cell types, to be dependent on extracellular calcium.28 We have shown that post-fusion behaviour of the secretory granules in the acinar cells is complex with the fusion pore, that forms between the granule and the cell membrane, capable of opening and closing.29 In endocrine cells, where fusion pore dynamics have been well studied, it has been shown that high extracellular calcium promotes closure of this fusion pore.28 If a similar behaviour were found in acinar cells then a rise in luminal calcium could provide a negative feedback signal shutting the fusion pore and preventing further release of enzymes.
The pH of the lumen is also not well defined, again the outflow of the gland can be measured (pH 8.5 from the whole pancreas30) but it is likely that pH shows regional changes along the lumen. Unlike calcium, the mechanisms that might regulate luminal pH are better understood and include sodium-proton exchange, bicarbonate and chloride transporters31,32 and the plasma membrane calcium pump33 (PMCA).
On acinar cell stimulation a rise in intracellular calcium, triggers the opening of apical chloride channels and movement of chloride from the cell into the lumen.34 This chloride outflow is balanced by a basolateral chloride influx through the sodium-potassium-2 chloride cotransporter and through a chloride-bicarbonate exchanger.35 In this way trans-epithelial chloride transport can be maintained over extended periods of time. It has been shown that during this stimulated chloride movement, intracellular pH is maintained by basolateral proton efflux through a sodium-proton counter transporter, NHE1.36 This model for secretion suggests that luminal pH changes in the secretory end-pieces are not under direct cellular control (see Figure 4 for proposed model).
In the downstream duct cells, apical bicarbonate movement is tightly linked to chloride movement and it is the relatively high luminal bicarbonate concentration that gives rise to the alkaline pH of the total organ outflow.6 The precise mechanism of bicarbonate secretion is still unknown but it is believed that bicarbonate either enters the cell through a basolateral sodium-bicarbonate co-transporter or is produced in the cell via the action of carbonic anhydrase.6 Bicarbonate outflow into the lumen is balanced by basolateral proton loss either through the NHE or through the V-ATPase.37 At the apical membrane of the duct cells chloride efflux is most probably largely mediated by CFTR and this acts in consort with a chloride-bicarbonate exchanger to raise luminal bicarbonate.38
While bicarbonate is the most commonly thought of luminal pH buffer, work in the salivary gland has shown that phosphate and secreted proteins are also significant pH buffers.39 In particular the proteins secreted from the acinar cells, when the salivary glands are stimulated, are thought to have a significant pH buffering action.
The above discussion suggests that the luminal environment of the ducts might possess significant pH buffering power but buffering in the lumen in the secretory endpieces remains unknown. Thus local gradients and transients of pH might still be established, especially in the secretory endpieces where the contents of the highly acidic secretory granules (pH < 6.040) would release protons in to the lumen. Here the pH shifts are likely to be large and this has the potential to locally regulate exocytosis (inhibits below pH 6.541) and endocytosis.30 These pH changes might also affect epithelial cell ion channels such as the CLC-2 chloride channel42 that are activated by acid pH (enhanced at pH less than 7.443), or TASK potassium channels that are inhibited at acid pH.44
Fluid flow and fluid composition in the pancreas is principally an aqueous vehicle for pancreatic enzyme secretion. It provides the bulk fluid flow necessary to drive pancreatic output to the small intestine. However it is also likely to play a significant role in the dynamics of enzyme activation. Many enzymes are secreted as precursors that are activated at the small intestine but at the point of pancreatic outflow optimal conditions in the fluid constituents will ensure maximal enzyme activity. For example high calcium is a requirement for the functioning on many of the secreted enzymes including amylase, trypsin and lipase.17 An alkali pH is also associated with optimal activity for many enzymes.45 Calcium released as a result of granule fusion in the acinar cells and HCO3− from the activity of the duct cells will therefore act in consort to deliver the optimal fluid environment for enzyme activity in the small intestine.
Using two-photon microscopy on tissue fragments, largely consisting of secretory endpieces46 we have recently recorded dramatic luminal acidification on cell stimulation (Figure 3). We show that this acidification is coincident with granule fusion and likely arises from the release of protons that are contained within the granule and then get released when the granule fuses with the apical plasma membrane.
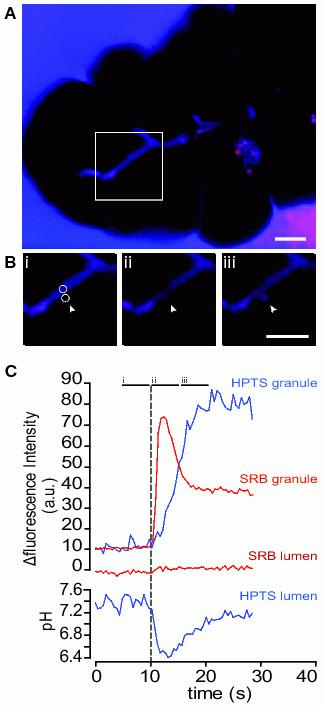
Figure 3. A single granule fusion event elicits a large transient acidification of the lumen. A shows a low magnification image, taken before stimulation, of a cluster of acinar cells at the end of a larger fragment of tissue. The tissue is bathed in a Na-rich extracellular media containing no pH buffer with the fluorescent dyes HPTS (blue) and SRB (red) added. The colour image is the resultant overlay of blue and red and shows that the dyes are largely colocalized outside the fragment and inside the lumen seen as the branching diagonal structure across the centre of the image. Scale bar 5μm. B shows an enlarged series of images taken over the times indicated on the graph in C. The two circles highlight regions of interest drawn over the lumen and over the exocytic event – also indicated by the arrow. Image (i) is before the event occurs, image (ii) is at the peak of the influx of extracellular dye into the fused granule. At this point (image ii) the granule appears red because the SRB (red) fluorescence is unaffected by granule acidity where-as the HPTS (blue) is quenched. Finally image (iii) is taken at a later time in the granule lifetime at a point where the pH is in equilibrium with the lumen. C shows the average intensity of the SRB and HPTS fluorescence plotted over time in the region of interest of the granule and in the region of interest of the lumen.
In these experiments we use extracellular fluorescent dyes to record exocytosis; the dyes enter the granule, through the open fusion pore (at the time shown by the dotted vertical line in Figure 3). Usually we employ inert extracellular dyes, like sulforhodamine B (SRB47) and have shown that these dyes enter the granule after fusion, accumulate on the granule content (giving the initial peak in the SRB granule fluorescence in Figure 3). As granule content is lost the bound dye also comes out until a point is reached where the granule is empty of its original content but still filled with aqueous solution that contains SRB (leading to the decline of fluorescence to a plateau seen in the SRB granule fluorescence in Figure 348).
In experiments exploring pH changes we add the pH-sensitive dye, 8-Hydroxypyrene-1,3,6-trisulfonic acid, trisodium salt, HPTS32 to the extracellular solution and record its fluorescent changes (SRB is also present in these experiments). In our system the wavelengths of excitation and emission light we use lead to a decrease of HPTS fluorescence on acidification. As seen in Figure 3 the HPTS granule fluorescence rises much more slowly than the SRB fluorescence. This is unlikely to reflect differences in dye binding to granule content (since MPTS, a pH-insensitive dye with a close structural similarity to HPTS shows similar fluorescence changes to SRB, data not shown). Instead the relatively slow rise in HPTS granule fluorescence is due to the initial acid environment of the granule quenching the HPTS signal. Tracking the luminal fluorescence changes in a region of interest immediately adjacent to the exocytic site, shows a small increase in SRB fluorescence (as the content-bound dye diffuses out of the granule) but shows a dramatic, transient decrease in HPTS fluorescence. Calibration of the pH-dependent changes in HPTS fluorescence, assuming a starting pH of 7.4, gives us an estimate of the actual pH change which in this example is nearly one pH unit; a ten fold increase in proton concentration.
These pH changes are summarized in Figure 4 which shows that on exocytosis the lumen acidifies. Given the dramatic pH changes we now observe (Figure 3), we expect that these are likely to be important physiologically potentially influencing the PMCA, the apical chloride channels or even the exocytotic/endocytotic process itself (Figure 4).
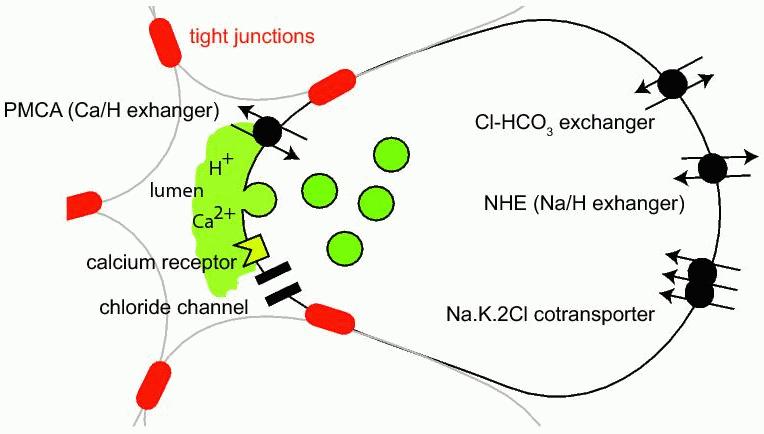
Figure 4. Illustration of some of the membrane transport mechanisms likely to be effective in pH balance in exocrine acinar cells. Here we show the acidification and likely increase in calcium that result from exocytosis. In turn these luminal changes have the possibility to act on the PMCA, chloride channels, the secretory process itself and the calcium receptor.
The composition of the luminal fluid is under physiological control and in diseases, such as cystic fibrosis and pancreatitis, even measures of total secretory output from the organ show changes in composition. Our data suggest that local dynamic changes in the luminal content may be even more dramatic and may be a critical factor in the development of these diseases.
Cystic fibrosis is a disease that arises from mutations in a chloride channel (CFTR). It is disabling and can lead to premature death. Since this chloride channel is physiologically involved in fluid secretion it is not surprising that luminal constituents are affected.5 In the lung these pathological changes increase the viscosity of the secretions lining the lung leading to higher rates of infection and impaired lung function.7 In exocrine tissues the role of the chloride channel in bicarbonate exchange impairs fluid secretion and, in the pancreas this can lead to organ damage and a reduction in enzyme secretion.49 Interestingly in cystic fibrosis patients very large shifts to an acid pH have been observed in lung breath condensate (from pH 6.15 in normal to 5.32 in cystic fibrosis50) and pancreas output (from pH 8.5 in normal to 6.6 in cystic fibrosis30). Since ion channels and the secretory process itself are sensitive to pH in this range it is probable that the disease-induced acidification of the luminal environment will have consequences for the regulation of ion channels and granule membrane recovery30 and therefore contribute to the disease phenotype.
Pancreatitis is a disease of the pancreas that is initiated by intraglandular activation of enzymes51 and leads to symptoms including severe pain, nausea, inflammation and tissue damage. Current treatments are limited to symptomatic care such as relief of pain and inflammation.52 Aberrant pancreatic enzyme secretion is thought to be a key step in disease progression with a paradoxical reduction in whole-gland secretion but an increase in acinar cell enzyme secretion.51 Not surprisingly the lumen content changes and these changes include a 3 fold rise in calcium in the pancreatic outflow53 and an acidification, measured at the duodenal outflow of the pancreas from pH 7.0 to 6.1.53 Together these changes in calcium and pH would potentially promote autoactivation of pancreatic trypsin, one of the hallmarks of the disease55,56 and indeed weak-base treatment using chloroquine has been shown to be effective in ameliorating pancreatitis.57 We would predict that acinar cell hyper-secretion would dramatically acidify and raise calcium in the lumen of the secretory endpieces exerting not only a local influence on the acinar cells but influencing the behaviour of the duct cells downstream in the gland.
There is clear evidence that the luminal environment of hollow organs is dynamically altered by the activity of the cells that line the lumen. Furthermore, studies show that the composition of this luminal fluid changes in disease states. What we now show is that there is a possibility for very local and dramatic changes in the luminal constituents that could well be a key feedback regulator of organ function.
Imaging vesicular secretion. We have published extensively on the use of the two photon microscopy for the study of single granule secretory events in pancreatic acinar cells1 and described experiments using the entry of extracellular fluorescent dyes to identify granule fusion. In these published experiments we show that the extracellular dye, under our conditions labels the secretory granules. In principle the dye would enter any endocytic compartment. But we show that the fluorescent labelling of granules is in the right place (only apical in polarized epithelia), occurs with the right time course (compared to measures of enzyme secretion) and labels structures of the right size for granules (∼0.8μm in acinar cells). Furthermore, in immunocytochemical data we show that fluorescently labelled granules also contain residual amounts of chymotrypsinogen (a specific secretory enzyme only found in zymogen granules).29 In the experiments shown in this review SRB and HPTS were both added to the extracellular solution at a concentration of 400μM.
Electron microscopy. Standard techniques of tissue preparation and transmission electron microscopy were used. Tissue fragments were fixed in glutaraldehyde using a microwave method. After dehydration tissue was mounted in epon resin and 70nm thin sections prepared for imaging.
This work was funded by an Australian Research Council Grant (DP0771481), and a Research Infrastructure Block Grant from The University of Queensland to PT.
1. Larina, O, Thorn P. Ca2+ dynamics in salivary acinar cells: distinct morphology of the acinar lumen underlies near-synchronous global Ca2+ responses J. Cell Science. 2005; 118: 4131-9.
2. Fliegauf M, Benzing T, Omran H. When cilia go bad: cilia defects and ciliopathies. Nature 2007; Rev. Mol. Cell Biol. 8: 880-93
3. Nehrke K, Arreola J, Nguyen H-V, et al. Loss of hyperpolarization-activated Cl− current in salivary acinar cells from Clcn2 knockout mice. J. Biol. Chem. 2002; 277: 23604-11.
4. Nauntofte B. Regulation of electrolyte and fluid secretion in salivary acinar cells. Am. J. Physiol. 1992; 263: G823-G37.
5. Young JA, Cook DI, Van Lennep EW, Roberts ML. Secretion by the major salivary glands. In: Johnson L, Christensen J, Jackson M, Jacobson E, J Walsh (eds). Physiology of the Gastrointestinal Tract. Raven Press. New York, 1994; 1061-117.
6. Steward MC, Ishiguro H, Case RM. Mechanisms of bicarbonate secretion in the pancreatic duct. Ann. Rev. Physiol. 2005; 67:377-409.
7. Tarran R, Grubb BR, Parsons D, et al. The CF salt controversy in vivo observations and therapeutic approaches. Mol. Cell. 2001; 8: 149-58.
8. Song Y, Tjiagarajah J, Verkman AS. Sodium and chloride concentrations, pH and depth of airway surface liquid in distal airways. J. Gen. Physiol. 2003; 122: 511-9.
9. Gorodeski GI, Hopfer U, Liu CC, Margle E. Estrogen acidifies vaginal pH by up-regulation of proton secretion via the apical membrane of vaginal ectocervical epithelial cells. Endocrinology 2004; 146: 816-24.
10. Pastor-Soler N, Beaulieu V, Litvin TN, et al. Bicarbonate-regulated adenylyl cyclase (sAC) is a sensor that regulates pH-dependent V-ATPase recycling. J. Biol. Chem. 2003; 278: 49523-9.
11. Eckardstein SV, Cooper TG, Rutsch K, et al. Seminal plasma characteristics as indicators of cystic fibrosis transmembrane conductance regulator (CFTR) gene mutations in men with obstructive azoospermia. Fertility and Sterility 2000; 73: 1226-31.
12. Martinez JR, Holzgreve H, Frick A. Micropuncture study of submaxillary glands of adult rats. Pflügers Archiv. 1966; 290: 124-33.
13. Young JA, Schogel E. Micropuncture investigation of sodium and potassium excretion in rat submaxillary saliva. Pflügers Archiv. 1966; 291: 85-98.
14. Gray, M.A. Bicarbonate secretion: it takes two to tango. Nature Cell Biol. 2004; 6: 292-4.
15. Choi JY, Muallem D, Kiselyov K, Lee MG, Thomas PJ, Muallem S. Aberrant CFTR-dependent HCO3− transport in mutation associated with cystic fibrosis. Nature 2001; 410: 94-7.
16. Argent BE, Case RM, Scracherd T. Amylase secretion by the perfused cat pancreas in relation to the secretion of calcium and other electrolytes and as influenced by the external ionic environment. J. Physiol. 1973; 230: 575-93.
17. Ceccarelli B, Clemente F, Meldolesi J. Secretion of calcium in pancreatic juice. J. Physiol, 1975; 245: 617-38.
18. Clemente, F. Meldolesi J. Calcium and pancreatic secretion. J. Cell Biol. 1975; 65: 88-102.
19. Nicaise G, Maggio K, Thirion S, Horoyan M, Keicher E. The calcium loading of secretory granules. A possible key event in stimulus-secretion coupling. Biol.Cell 1992;. 75: 89-99.
20. Belan P, Gardner J, Gerasimenko O, et al. Localization of calcium extrusion sites in pancreatic acinar cells. J. Biol. Chem. 1996; 272: 4106-11.
21. Tepikin AV, Voronina SG, Gallacher DV, Petersen OH. Pulsatile calcium extrusion from single pancreatic acinar cells during receptor activated cytosolic calcium spiking. J. Biol. Chem. 1992; 267: 14073-6.
22. Zhao X-S, Shin DM, Liu LH, Shull GE, Muallem S. Plasticity and adaptation of calcium signaling and calcium-dependent exocytosis in SERCA2+/− mice. EMBO J. 2001; 20: 2680-9.
23. Bruce JIE, Yang X, Ferguson CJ, et al. Molecular and functional identification of a calcium (polyvalent cation)-sensing receptor in rat pancreas. J. Biol. Chem. 1999; 274: 20561-8.
24. Ward DT. Calcium receptor-mediated intracellular signalling. Cell Calcium 2004; 35: 217-28.
25. Racz GZ, Kittel A, Riccardi D, Case RM, Elliot AC, Varga G. Extracellular calcium sensing receptor in human pancreatic cells. Gut 2002; 51: 705-11.
26. Conigrave A, Brown EM. Taste receptors in the gastrointestinal tract. II L-amino acid sensing by calcium-sensing receptors: implications for GI physiology. Am. J. Physiol. 2006; 291: G753-61.
27. Caroppo R, Gerbino A, Fistetto G, et al. Curci S. Extracellular calcium acts as a “third messenger” to regulate enzyme and alkaline secretion. J. Cell Biol. 2004; 166: 111-9.
28. Alés E, Tabares L, Poyato JM, Valero V, Lindau M, Alvarez de Toledo G. High calcium concentrations shift the mode of exocytosis to the kiss and run mechanism. Nature Cell Biol. 1999; 1: 40-4.
29. Larina O, Bhat P, Pickett JA, et al. Dynamic regulation of the large exocytic fusion pore in pancreatic acinar cells. Mol. Biol. Cell 2007; 18: 3502-11.
30. Freedman SD, Kern HF, Scheele GA. Pancreatic acinar cell dysfunction in CFTR−/− mice is associated with impairments in luminal pH. Gastroenterology 2001; 121: 950-7.
31. Muallem S, Loessberg PA. Intracellular pH-regulatory mechanisms in pancreatic acinar cells. I. Characterization of H+ and HCO3− transporters. J. Biol. Chem. 1990; 265: 12806-12.
32. Willoughby D. Schwiening CJ. Depolarization-induced pH microdomains and their relationship to calcium transients in isolated snail neurones. J. Physiol. 2002; 544: 487-9.
33. Schwiening CJ, Kennedy HJ, Thomas RC. Calcium-hydrogen exchange by the plasma membrane Ca-ATPase of voltage-clamped snail neurones. Proc. Roy. Soc. Lond. 1993; 253: 285-9.
34. Kidd J, Thorn P. The properties of secretagogue-evoked chloride current in mouse pancreatic acinar cells. Pflügers Archiv. 2001; 441:489-97.
35. Zhao H, Muallem S. Na+, K+, and Cl− transport in resting pancreatic acinar cells. J. Gen Physiol. 1996; 106: 1225-42.
36. Brown DA, Melvin JE, Yule DI. Critical role for NHE1 in intracellular pH regulation in pancreatic acinar cells. Am. J. Physiol. 2003; 285: G804-12.
37. Zhao H, Star RA, Muallem S, Membrane localization of H+ and HCO3− transporters in the rat pancreatic duct. J. Gen. Physiol. 104: 57-85.
38. Ko SBH, Shcheynikov N, Choi JY et al. A molecular mechanism for aberrant CFTR-dependent HCO3− transport in cystic fibrosis. EMBO J. 2002; 21: 5662-72.
39. Bardlow A, Moe D, Nyvad B, Nauntofte B. The buffer capacity and buffer systems of human whole saliva measured without loss of CO2. Archiv. Oral Biol. 2000; 45: 1-12.
40. Ahdut-Hacohen R, Duridanova D, Meiri H. Rahamimoff R. Hydrogen ions control synaptic vesicle ion channel activity in Torpedo electromotor neurones. J. Physiol. 2004; 556: 347-52.
41. Cazalis M, Dayanithi G. Nordmann JJ. Requirements for hormone release from permeabilized nerve endings isolated from the rat neurohypophysis. J. Physiol. 1987; 390: 71-91.
42. Carew MA, Thorn P. Identification of CLC-2 like chloride currents in pig pancreatic acinar cells. Pflügers Arch 1996; 433: 84-90.
43. Blaisdell CJ, Edmonds RD, Wang Z-T, Guggino S. Zeitlin PL. pH-regulated chloride secretion in fetal lung epithelia. Am. J. Physiol. 2000; 278: L1250-5.
44. Duprat F, Girard C, Jarretou G. Lazdunski M. Pancreatic two P domain K+ channels TALK-1 and TALK-2 are activated by nitric oxide and reactive oxygen species. J. Physiol. 2005; 562: 235-44.
45. Worthington Enzyme Manual 1993; Worthington Biochemical Corporation, Lakewood, New Jersey, USA.
46. Thorn P, Fogarty KE. Parker I. Zymogen granule exocytosis is characterized by long fusion pre openings and preservation of vesicle lipid identity. Proc. Nat. Acad. Sci. 2004; 101: 6774-9.
47. Nemoto T, Kimura R, Ito K, et al. Sequential-replenishment mechanism of exocytosis in pancreatic acini. Nature Cell Biol. 2001; 3: 253-8.
48. Thorn P. Parker I. Two phases of zymogen granule lifetime in mouse pancreas: ghost granules linger after exocytosis of contents. J. Physiol. 2005; 563: 433-42.
49. Gaisano HY, Lutz MP, Leser J, et al. Supramaximal cholecystokinin displaces Munc18c from the pancreatic acinar basal surface, redirecting apical exocytosis to the basal membrane. J. Clin. Invest. 2001; 108: 1597-611.
50. Tate S, MacGregor G, Davis M, Innes JA, Greening AP. Airways in cystic fibrosis are acidified: detection by exhaled breath condensate. Thorax 2002; 57: 926-9.
51. Whitcomb DC. Acute pancreatitis New Eng. J. Med. 2006; 354: 2142-50.
52. Cruciani RA, Jain S. Pancreatic pain: a mini review. Pancreatology 2008; 8: 230-5.
53. Furui T, Kondoh S, Harada T, et al. Calcium concentration and artificial precipitates in human pancreatic juice. Pancreas 2000; 21: 257-61.
54. Dutta SK, Russell RM, Iber FL. Influence of exocrine pancreatic insufficiency on the intraluminal pH of the proximal small intestine. Dig. Dis. & Sci. 1979; 24: 529-34.
55. Otani T, Chepilko SM, Grendell JH, Goerlick FS. Codistribution of TAP and the granule membrane protein GRAMP-92 in rat caerulein-induced pancreatitis. Am. J. Physiol. 1998; 275: G999-1009.
56. Waterford SD, Kolodecik TR, Thrower EC, Gorelick FS. Vacuolar ATPase regulated zymogen activation in pancreatic acini. J. Biol. Chem. 2005; 280: 5430-4.
57. Leach SD, Bilchik AJ, Karapetian O, et al. Influence of chloroquine on diet-induced pancreatitis Pancreas 1993; 8: 64-9.