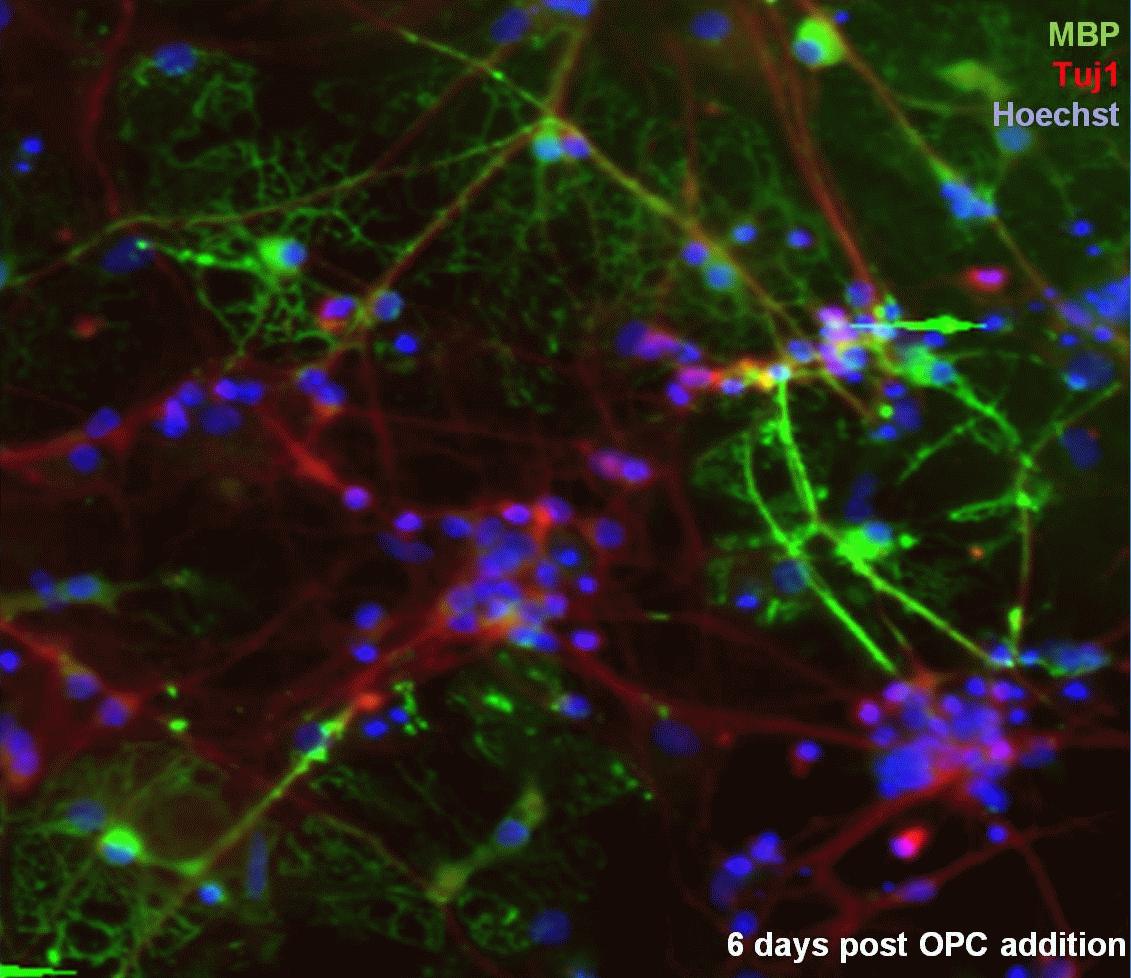
Understanding the cellular and molecular responses to CNS demyelination could provide insight into pathogenic processes that occur at the earliest stages of lesion formation in multiple sclerosis (MS). Developing an in vitro model of CNS demyelination that enables real-time monitoring and manipulation of multiple independent variables in a fully defined culture system would offer many advantages. Here we describe progress towards generating such a model, comprising co-cultures of purified CNS retinal ganglion cells (RGCs) and oligodendrocyte progenitor cells (OPCs). OPCs were isolated from transgenic mice which express diphtheria toxin receptor (DTR) under the control of the oligodendrocyte-specific myelin basic protein (MBP) promoter. In vitro differentiation of MBP-DTR+ OPCs into mature myelinating oligodendrocytes results in the specific induction of DTR expression on mature oligodendrocytes rendering them sensitive to diphtheria toxin (DT)-mediated apoptosis. To date, we have established a rapidly myelinating co-culture system using mouse RGCs and OPCs (n=6). In addition, lenti-viral transduction of OPCs using a green fluorescent protein (GFP)-expressing virus prior to seeding onto dorsal root ganglion (DRG) neurons has enabled us to assess myelination in live cultures using time-lapse confocal microscopy (n=4). We have also demonstrated that the addition of 50 ng/ml DT to a RGC/ GFP+ OPC co-culture system results in the loss of oligodendrocytes only in MBP-DTR+ co-cultures in comparison to WT co-cultures (n=1). An in vitro model of CNS demyelination will provide a novel defined system to assess the role of specific factors and cell types implicated in the formation and evolution of early MS lesions and further our understanding of responses to degeneration of the axo-glial interface.
