To understand function and regulation of proteins it is important to have knowledge about their properties in the physiological environment. In this study, we have used small segments of single skeletal muscle fibres dissected from rat skeletal muscle to examine diffusibility and fibre type expression of a number of glycogen related proteins as well as AMPK isoforms.
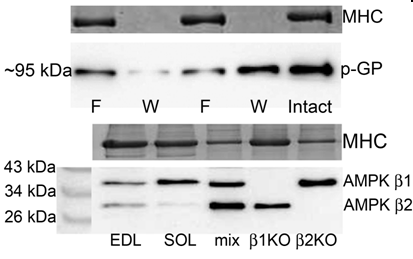
Male Long-Evans hooded rats (6-8 months old) were sacrificed using a lethal overdose of fluothane in accordance with the La Trobe University Animal Ethics Committee and the extensor digitorum longus (EDL) and soleus (SOL) muscles were excised. To compare fibre type differences EDL (exclusively type II) and SOL (predominantly type I) muscle fibres were analyzed for AMPK β1 and β2, glycogen branching enzyme (GBE), glycogen debranching enzyme (GDE), glycogen phosphorylase (GP), phospho-GP and glycogen synthase (GS). To measure protein diffusibility, individual fibres were dissected from muscles that had been immersed in paraffin oil and then mechanically-skinned and exposed to physiological K+-based solution (pCa < 10) for 1 and 10 min. The wash solution (W) and their matched fibres (F) were analyzed side by side using Western blotting (Murphy et al., 2006). For fibre type comparisons, the amount of each protein was normalized to the amount of an abundant muscle protein (i.e. actin or myosin) and then expressed relative to the amount of the given protein present in the SOL fibres. Identity of AMPK β isoforms was confirmed by their absence in muscle homogenates from respective knock-out mice.
Proteins related to glycogen breakdown (GDE, GP, p-GP) were present in higher amounts in fast- compared with slow-twitch muscle (Table). Proteins related to glycogen synthesis were similar (GS) or lower (GBE) in slow- compared with fast-twitch muscle (Table). The AMPK β isoforms had the opposite abundances in fast- and slow-twitch muscle. Some proteins were freely diffusible (GBE, AMPK β1 and β2), some appeared to be weakly bound (GP, p-GP), whilst GDE was much more tightly associated with a muscle structure and likely bound with glycogen which reportedly washes out <40% in 10 min (Goodman et al., 2005). Given that ∼80% of the GP appeared in the wash, it is likely that there is considerable excess of this enzyme compared with the amount of glycogen present .These findings strongly suggest that when glycogen related proteins or AMPK are examined in skeletal muscle, the fibre type dependence must be taken into account. This is particularly important in human skeletal muscle which is typically heterogeneous with respect to fibre types present and so these proteins should be examined in individual fibres.
| Protein / enzyme |
Approx amount in EDL compared with |
Amount appearing in wash after 10 min |
|
| SOL | EDL | SOL | |
| AMPK β1 | 0.4 | 70% | 70% |
| AMPK β2 | 4 | 70% | 70% |
| GBE | 0.2 | 80% | 85% |
| GDE | 5 | 20% | 50% |
| GP | 100 | 80% | 40% |
| phospho-GP | 10 | 60% | 40% |
| GS | 1 | ND | ND |

Goodman C, Blazev R & Stephenson G. (2005). Clinical and Experimental Pharmacology and Physioliology 32, 749-756.
Murphy RM, Verburg E & Lamb GD. (2006). Journal of Physiology 576, 595-612.