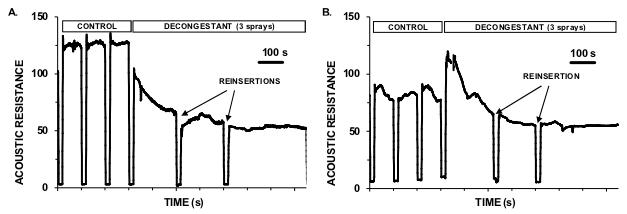
To investigate nasal congestion (DeLong & Getchell, 1987), we have developed a novel and inexpensive technique for monitoring changes in nasal air resistance (Corey, Houser & Ng, 2000; Hilberg, 2002). We monitor the small-signal acoustic input impedance of the upper airway (dominated by the nasal valve), analogous to the measurement of the middle ear impedance in audiology. The technique requires the generation of a fixed sinusoidal volume-velocity, sound-stimulus delivery tubes, nose-pieces to deliver the sound without leaks to the nasal entrances, and microphones to monitor the resultant pressure fluctuations produced there. For a fixed volume-velocity drive, the sound pressure fluctuation at the nasal opening is proportional to the nasal acoustic input impedance (at low frequencies dominated by acoustic resistance and calculated as the pressure response divided by the volume-velocity stimulus). The bilateral acoustic probe tones were generated by the laptop through an inexpensive USB external sound card (Sabrent, USB-SND8), chosen because it provided dual microphone inputs. The two earbud sound sources were separated from the nasal inserts by 300mm lengths of Silastic tubing (1mm internal diameter), so that the tubes high acoustic impedance ensured that each earbud closely approximated an AC volume-velocity source. The pressure sensors were two small, inexpensive electret microphone elements, mounted in parallel with each earbud, and also separated from each nose insert by a 300 mm length of tubing (1 mm external diameter). Software developed in LabView 7.0 (National Instruments) monitored the raw pressure waveforms and frequency spectra (particularly the probes amplitude proportional to the modulus of nasal impedance). The subjects mouth was held open at a fixed gape using a 5cm length of corrugated plastic pipe of 5cm diameter, with the tongue under the tube to minimize occlusion at the back of the throat. This kept the resistance contribution from the mouth and throat low and constant relative to nasal resistance. Our calibration resistance was a 1mL syringe (Terumo) with tip removed at the 0 ml mark. Using Pouiselle's law, its (laminar flow) resistance was ∼20 kNs/m5. A 433 Hz probe sound probe generated 104 dB SPL at the neck of the calibration resistance, so that in a normally functioning nasal passage, with a resistance 10-20% of our standard calibration tube, the sound level at the nasal entrance was between 84 to 90 dB SPL (heard as a soft tone). At 433Hz the acoustic impedance was dominated by resistance, with the phase of all measurements within 5 degrees of the phase at the opening of the calibration resistance. Two examples (separated by one week) of the drop in nasal resistance in one human subject with application (3 sprays) of over-the-counter nasal decongestants are shown in Figures A (phenylephrine as Nyal Nasal Decongestant) and B (oxymetazoline as Vicks Sinex Soother). Acoustic resistance of 100 corresponds to ∼80 kNs/m5 (40% of calibration resistance). These responses also serve as an internal control for measurement artefact. Note also the reproducibility of measurements with probe reinsertion.

DeLong RE. Getchell TV. (1987) Chemical Senses 12, 3-36.
Hilberg O. (2002) Allergy 57, 5-39.
Corey J, Houser S, Ng B. (2000) Ear, Nose & Throat Journal 79, 690-698.