While developed countries have adopted universal neonatal hearing screening (using middle-ear assessment, otoacoustic emissions and automated auditory brainstem responses or AABR), cost (particularly of AABR) is a barrier to its adoption in developing countries. Unfortunately an objective neural assay must be included in screening, because 10% of deaf babies suffer from auditory neuropathy (McMahon et al., 2008). The sound-evoked post-auricular muscle response (PAMR; O'Beirne & Patuzzi, 1999) is a promising alternative to expensive AABR: (a) it is a sound-evoked electrical muscle response obtainable near auditory threshold, and is driven from the inferior colliculus via the facial nerve (so it represents neural function equivalent to wave V of the AABR); and (b) it is 20 times larger than ABR, and so is more easily, rapidly and cheaply obtained (Purdy et al., 2005). One barrier to its adoption is an unfounded view that it is unreliable, largely because clinicians have used head rotation to potentiate it. As described here, the PAMR is actually potentiated by eye rotation, not by head rotation: the perceived unreliability of PAMR is probably due to unreliable eye rotation in subjects with no explicit instructions about what to do with their eyes. When eye rotation is controlled, the PAMR can be reliably potentiated for hearing screening, or avoided when recording smaller neurogenic responses.
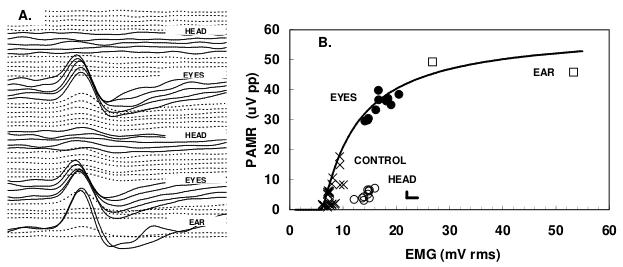
In this study, PAMR was recorded from 12 normal adults (active electrode overlying the PAM and an indifferent on the back of the pinna). PAMR and background EMG noise were amplified, software filtered (fourth-order bandpass filter from 1Hz to 300Hz) and averaged (n=300 over 16 s). Stimuli were bipolar, alternating acoustic clicks (360 μs per phase and 53 ms interval, giving a 19/s repetition rate, 50dB SL), delivered bilaterally via ear-bud headphones. Subjects first sat quietly with head and eyes facing forwards until little or no response could be observed. Subjects were then asked to turn their head hard but slowly (5 s) to the electrode side, while keeping their gaze fixed forward relative to the head, using a visual target attached to a hat. After 80s, subjects were asked to rotate their head slowly to the front control position along with their eyes. Thereafter subjects rotated their eyes alone hard to the right without moving their head, before returning their eyes slowly back to the forward-directed control gaze. One typical set of responses is shown in Figures A (waterfall plot of waveforms from top to bottom) and B (data replotted from A with CONTROL: head and eyes forward; EAR, voluntary contraction of PAM; EYES, rotation of eyes towards electrode with head facing forward; HEAD, head rotated towards electrode with maintained forward gaze relative to head). As can be seen, head rotation did little to potentiate the PAMR, while eye rotation reliably potentiated it, but by less than voluntary ear wiggling. Overall, the signal-to-noise ratio for the PAMR with eye rotation was 2.5 times greater than that with head rotation with eyes fixed relative to head, suggesting that the PAMR can be obtained about 6 times faster (2.5 × 2.5) with eye rotation than with head rotation, and it is less variable than previously reported.

O'Beirne G. Patuzzi RB. (1999) Hearing Research 138, 115-132.
McMahon CM, Patuzzi RB, Gibson WP, Sanli H. (2008) Ear and Hearing 29, 314-325.
Purdy SC, Agung KB, Hartley D, Patuzzi RB, O'Beirne GA. (2005) International Journal of Audiology 44 625-630.