The molecular and cellular mechanisms of pacemaker activity in the sino-atrial node (SAN) have been dissected with profound precision in several mammalian species. However, an anatomically defined mammalian SAN is only a small part of complex 3-dimensional structure known as distributed pacemaker complex (Boineau et al., 1988), which cannot be fully understood at the cellular level. We aimed to delineate the complex anatomical structure of the sinus node with the pattern of excitation under various physiological conditions in humans and test a hypothesis postulating that SAN connects to atria via specialized exit pathways (Boineau et al., 1988).
SAN has been studied for many years in the mouse, rabbit, canine and other mammalian species. However, much less is known about the function of the human SAN. We have developed a program at Washington University in St. Louis, USA, which aims to investigate normal and pathological physiology of the human heart in vitro using state-of-the-art basic physiological methods. We have obtained 146 live human hearts from (1) heart failure patients undergoing transplantation and (2) donors whose hearts could not be transplanted for various reasons. Hearts were cardioplegically arrested in the operating room immediately after removal from the chest, and within 15-20 minutes delivered to the basic electrophysiology laboratory. Using several modalities of imaging and molecular biology, we have investigated gene and protein expression, electrical activation and repolarization, and calcium handling in normal and failing human hearts in vitro. In a different group of patients, using novel non-invasive electrocardiographic imaging, we have mapped at the Bakoulev Center for Cardiovascular Surgery (Moscow, Russia) atrial excitation in vivo in order to determine excitation pattern in humans and compare that obtained in vitro.

We applied fluorescent imaging with near-infrared voltage-sensitive dye to map epicardial excitation in the human right atrium (n=4) (Fedorov et al., 2010). Imaging revealed that the human sinus node, similar to that of the canine, is electrically coupled with the atria via several specialized sino-atrial exit pathways (SAEP) and insulted elsewhere. Under normal physiological conditions, excitation originates within the anatomically defined SAN and slowly spreads via the superior SAEP to breakthrough near the base of the superior vena cava, and then rapidly spreads over the atria. Vagal or electrical stimulation shifts excitation inferiorly towards either mid- or inferior SAEPs, changing the pattern of excitation and P-wave. Histology and immunohistochemistry confirmed the cellular and anatomic basis of SAEPs.
Using a novel closed chest non-invasive electrocardiographic imaging modality (Denisov et al., 2008), we have mapped epi- and endocardial patterns of excitation in 26 patients at the Bakoulev Center for Cardiovascular Surgery in Moscow, Russia. For validation of the novel non-invasive imaging methodology we have carried out atrial mapping using the CARTO III system (J&J, USA) in 3 patients and have compared results with noninvasive mapping data. We have found a satisfactory coincidence of atrial excitation patterns acquired by both methods.
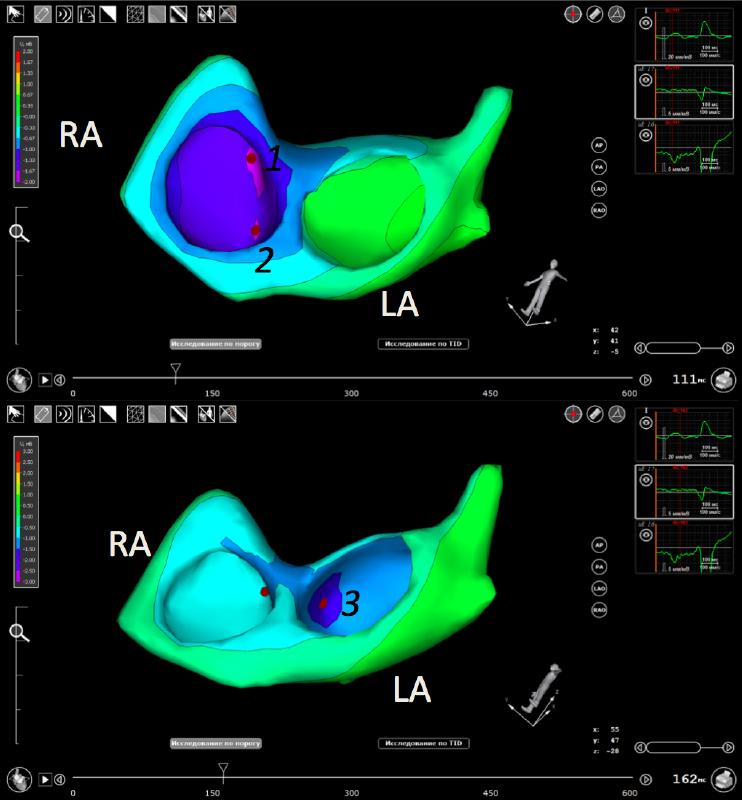
Similar to the in vitro studies, in vivo mapping revealed breakthrough sites at the superior (see 1 in figure) and inferior (2 in figure) SAEPs. Furthermore, it revealed an additional lateral SAEP (3 in figure), which connects SAN with the roof of the left atrium.
Conclusion: Our human data, obtained by three different imaging modalities both in vitro and in vivo, support existence of specialized sino-atrial exit pathways, which connect tge SAN and the atria.
Boineau JP, Canavan TE, Schuessler RB, Cain ME, Corr PB, Cox JL. (1988) Demonstration of a widely distributed atrial pacemaker complex in the human heart. Circulation 77: 1221–37.
Fedorov VV, Glukhov AV, Chang R, Kostecki G, Aferol H, Hucker WJ, Wuskell JP, Loew LM, Schuessler RB, Moazami N, Efimov IR. (2010) Optical mapping of the isolated coronary-perfused human sinus node. Journal of the American College of Cardiology 56(17): 1386-94.
Denisov AM, Zakharov EV, Kalinin AV, Kalinin VV. (2008) Numerical solution of the inverse electrocardiography problem with the use of the Tikhonov regularization method. Moscow University Computational Mathematics and Cybernetics 32(2): 61-68.