1. It is apparent from the literature that there are significant differences in excitation-contraction (EC) coupling between species, particularly in the density of calcium transporting proteins in the t-system and SR Ca release channels. Unfortunately, there is a lack of information on how the principal structures that link electrical excitation to the activation of calcium-induced calcium release (CICR) are different between human and animal models (particularly rat).
2. Comparison of wheat germ agglutin and caveolin-3 labelling revealed a non-uniform distribution of surface membrane glycosylation in rat, rabbit and human and that the rat t-system appeared more complex in geometry than the latter species. Analysis of the t-system skeleton showed the t-system was highly branched in the rat compared to that of the human (0.8 ± 0.08 and 0.2 ± 0.07 branch points per µm2 respectively, p < 0.001)
3. We also compared the distribution of contractile machinery, sodium-calcium exchange, sarcoplasmic reticulum and ryanodine receptors in rat and human. f-actin and ryanodine receptor (RyR) labelling was used to estimate the area of contractile apparatus supplied by each RyR cluster. In the rat, each RyR cluster supplied an average cross sectional area of contractile machinery of 0.36 ± 0.03 µm2 compared to 0.49 ± 0.04 µm2 in human (p = 0.048). SERCA2a labelling showed that sarcoplasmic reticulum formed a tight network of loops surrounding contractile fibrils that were denser than the t-tubule network but otherwise appeared similar in both species.
4. In general, the results show a higher density in structures involved in CICR in the rat compared to human.
In ventricular myocytes, the t-tubular system provides rapid transfer of the action potential deep into the cell to enable near-synchronous activation.1 The t-tubules form a network of plasma membrane invaginations in ventricular myocytes2-4 and studies have implicated aberrant t-tubule remodelling in animal and human heart disease.5-8 This remodelling may lead to a weakening of calcium signalling in heart failure.5,6,8 In rat myocytes, the t-tubules form a complex rete and have average diameter of around ∼250 nm.9 In contrast, in rabbit,10 guinea pig11 and human myocytes6 t-tubules form spoke like structures when viewed in transverse section and are wider (∼400 nm).
The action potential within the t-tubules leads to voltage dependent L-type calcium channels (or dihydropyridine receptors - DHPRs) opening, which allows an inward movement of calcium to activate calcium-induced calcium release.12 This influx may be augmented by sodium-calcium exchange (NCX) (see references 13-15) and the ‘trigger’ calcium opens ryanodine receptors (RyR) in the sarcoplasmic reticulum (SR) which provides the majority of calcium needed to activate contraction (for review see Bers, 200216). The SR release of calcium can be seen experimentally in the form of Ca2+ sparks which undergo spatio-temporal summation to produce a cell wide Ca2+-transient.17 The close geometric organization between RyRs and t-tubules is thought to be essential for efficient CICR, however the inter-cluster spacing is smaller in rat ventricular myocytes ∼0.67 µm18 than in rabbit ∼0.9 µm10 or human ∼0.8 µm.18 Furthermore, from previous studies, the fraction of clusters that are not associated with t-tubules in the rabbit appears larger than observed for rat.10,19 Comparable data for the human do not, to our knowledge, exist.
In view of the importance of the t-system for excitation-contraction coupling, we have carried out a comparative analysis of the organization of t-system, RyR clusters and SR in rat and human. Our data show clear differences between these species that may have important implications for studies on cardiac excitation-contraction coupling.
Rat cardiac myocytes were enzymatically isolated as described previously using protocols approved by the University of Auckland Animal Ethics Committee.20 Briefly, 250 g Wistar rats were killed by a lethal injection of pentobarbitone (100 mg/kg). The heart was quickly excised, mounted onto a Langendorff perfusion system and perfused with oxygenated Ca2+-free Tyrode's solution for 5 min. The heart was then perfused with same solution containing 200 µM CaCl2, 1.0 mg/ml Type II Collagenase (Worthington Biochemical, NJ) and 0.1 mg/ml Type I protease (Sigma-Aldrich, MO) for 10-15 min before the ventricles were cut free and diced into small pieces. The ventricular tissue was further incubated in Tyrode's solution containing 200 µM CaCl2 at 37°C for another 10 min before triturating to liberate single myocytes.
The resulting cell suspension was filtered and the isolated ventricular myocytes were fixed in 2% paraformaldehyde (PFA; w/v) (pH 7.4) in phosphate buffered saline (PBS) at room temperature for 12 min and washed in PBS. Myocytes were embedded in blocks of agarose prior to antibody labelling as described previously.19 0.1% Triton X100 diluted in PBS was applied to the samples for 10 min to permeabilize membranes prior to blocking with 10% normal goat serum (Vector Laboratories, Burlingame, CA). Primary antibodies were incubated in overnight at 4°C. and secondary antibodieds were applied at room temperature for 2 h, with 3 washes between antibody incubations. ∼1mm slices of the agar blocks were cut and mounted on a No. 1.5 glass coverslip and embedded in ProLong Gold (Molecular Probes/Invitrogen, NZ).
Rat and rabbit hearts were dissected and mounted on a Langendorff perfusion system. Hearts were perfused with PBS containing 2% PFA for 10 min. Ventricles were dissected into ∼5 mm blocks and incubated in PBS with 1% PFA for a further hour at 4°C prior to cryoprotecting in 30% sucrose. Human tissue was obtained according to approval of the New Zealand Health and Disability Ethics committee (NTY/05/08/050) with the informed and written consent of the family of the donor. Tissue samples were obtained from 4 donors that could not be matched to recipients; they had a mean age of 60 (54-68), and had normal echo and ECG. One patient was receiving felodipine. Small tissue pieces were processed and fixed overnight at 4°C by immersion in PBS containing 1% PFA. Tissue blocks were frozen in 2-methylbutane chilled with liquid nitrogen and stored at −80°C until sectioning. 30 µm thick cryosections were cut on a Leica CM1900 cryostat and mounted on poly-L-lysine coated number 1.5 glass coverslips. Tissue sections were blocked in Image-iT FX signal enhancer (Invitrogen, NZ) for 1 h and subsequent antibody incubation was as described for single cells (see above).
In double labelling experiments, samples were labelled with either Alexa 647 conjugated wheat germ agglutinin (WGA) or mouse monoclonal anti-NCX1 (R3F1; SWANT, Switzerland) in conjunction with a rabbit polyclonal anti-caveolin-3 (AB2912; Abcam, Cambridge, MA) primary antibody (CAV3). These samples were then labelled with Alexa 488 goat anti-mouse IgG and Alexa 568 goat anti-rabbit IgG secondary antibodies (Invitrogen). A mouse monoclonal anti-RyR2 primary antibody (MA3916; ABR, Golden, CO), an Alexa 488 goat anti-mouse IgG1 secondary antibody and Alexa 568-linked phalloidin were used for double labelling RyR and f-actin. SERCA2a was used as a marker for the SR network. This was labelled with a mouse monoclonal anti-SERCA2a and an Alexa 488-linked goat anti-mouse IgG2a (Invitrogen)
Cardiac myocytes were imaged in both transverse and longitudinal sections as we have previously shown that CICR structures are better resolved in transverse orientation.19 3D image volumes were obtained with a z-spacing of 0.2 µm and in-plane x-y sampling was more than twice the Nyquist limit using a LSM710 laser scanning confocal microscope (Zeiss, Jena, Germany) using a 63× 1.4NA oil immersion objective or an LSM410 confocal microscope using a 63× 1.25NA oil-immersion objective. Images were deconvolved using a maximum-likelihood Richardson-Lucy algorithm as described previously and all image analysis programs were custom written in IDL v6.4 (ITT, Boulder, CO).9
The geometry of the rat and human t-tubular networks were reconstructed by skeletonizing binary images of WGA and CAV3 labelling (human) or CAV3 and NCX (rat). By combining labels, we obtained a more complete picture of the t-system. Centroids of RyR clusters were calculated using a detection algorithm described previously.18 Delaunay triangulation between these centroids were calculated and a Voronoi diagram constructed. The Voronoi diagram was then modified with a mask constructed from the half-maximal phalloidin labelling pattern to provide a measure of the area of contractile apparatus supplied by each RyR cluster.
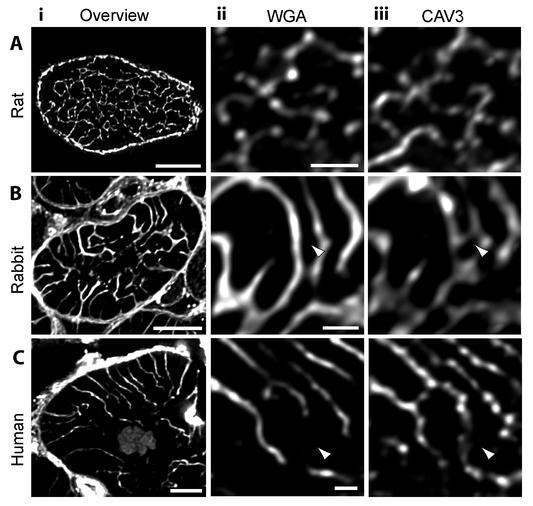
Labelling cardiac myocytes with WGA and CAV3 revealed clear differences in the structure of the t-system between species (Figure 1). In rat cells, the WGA label formed a complex rete which extensively overlapped with CAV3 labelling. In rabbit and human myocytes, the t-system appeared less complex with WGA labelling revealing a more spoke-like organization. Rat t-tubules were close to the diffraction limit (∼250-300 nm at full width at half maximum (FWHM) intensity) and smaller than rabbit or human t-tubules (∼350-500 nm FWHM). Importantly, fewer t-tubules labelled with WGA in rabbit and human as shown by CAV3 labelling. In addition, where WGA labelling was absent, the CAV3 labelled t-tubules were generally narrower (∼250-300 nm) and often formed connections between adjacent t-tubules containing both WGA and CAV3 labelling (Figure 1Bii-Biii).

Figure 1. Fluorescence imaging of t-tubules in rat, rabbit and human ventricular muscle. A-i: 1.25 µm deep maximum intensity projection of transverse confocal sections from a rat ventricular myocyte labelled for CAV3. Strong t-tubule labelling was observed in rabbit (B-i) and human (C-i) ventricular tissue, stained with WGA. The middle and right-hand columns illustrate magnified views of WGA labelling and CAV3 labelling in the interior of rat (A-ii,iii) rabbit (B-ii,iii) and human (C-ii,iii) myocytes respectively. Note the strong agreement of the t-tubular connections labelled by WGA and CAV3 in rat. In addition to the WGA labelling, CAV3 reported finer t-tubules in rabbit and human myocytes (arrowheads). Scale bars: Left column, 5 µm; Middle and right columns, 2 µm.
To quantify t-tubule structure a skeleton was constructed, and Figure 2 illustrates typical t-system skeletons. In transverse section, combination of NCX and CAV3 labels (inset Figure 2A) showed a highly branched structure in rat with branch points highlighted in white (Figure 2A). These data are very similar to the intact living t-system labelling described by Soeller & Cannell.9 Higher magnification views of rat and human tubules are shown in (Figure 2B and 2D respectively, together with their skeletons (Figures 2C and 2E). The number of t-tubule branching points was 0.80 ± 0.08 per µm2 of cell cross section in the rat compared to 0.17 ± 0.07 per µm2 in the human (respectively, p < 0.001 Mann-Witney U for df = 9). The increased branching in the rat t-system was associated with a more extensive skeleton with 1.61 ± 0.10 µm of t-tubule per µm2 of cross-section compared to 0.44 ± 0.07 µm per µm2 in the human (p < 0.001 Mann-Witney U for df = 9). In both species, the t-tubular skeleton formed complete loops as well as elements that appeared to terminate within the plane of section. However, these terminations usually connected with tubules from adjacent Z disk through an axial connection. The density of t-system was quantified by measuring the Euclidean minimum distance of interior pixels from the cell surface or t-tubule. As shown in Figure 2F, 64% of the cell interior was within 250 nm of the surface/t-system membrane (a distance at which µM Ca levels are reached in the sub-millisecond timescale and also the limit of optical resolution) in human compared 94% in the rat (χ2 = 26.25, df = 30, p < 0.001). In addition, the human appeared to have greater variability in t-tubule density as shown by the increased width of the distance histogram.
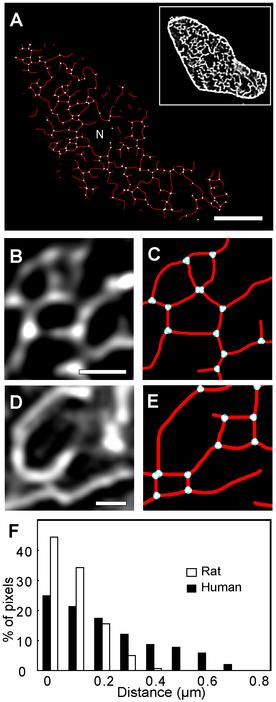
Figure 2. Skeletonization of t-tubules in rat and human ventricular myocytes. A: The t-tubular skeleton (red connecting lines) constructed from transverse confocal z-stacks of a rat myocyte labelled for CAV3 and NCX (shown in inset). Branching points in the skeleton are indicated in cyan. Note that nuclear regions (N) lacked CAV3 or NCX labelling, hence reported no t-tubules in the skeleton. B: A magnified view of a region stained for NCX and the skeleton corresponding to this region (C) in a rat myocyte. D: The magnified transverse view of NCX labelling in a similar region of a human myocyte and the corresponding skeleton (E) also reported branching points. F: The histogram shows the percentage of cytoplasmic pixels (excluding nuclear regions) as a function of the distance to either the cell surface or the nearest t-tubule skeleton in rat (white bars) and human (black) ventricular myocytes. Scale bars: A: 5 µm B&D: 2 µm.
RyR clusters in the form of ‘couplons’ are key elements that supply calcium to activate the contractile machinery.21 To analyze the relationship between RyRs and myofilaments, RyR and f-actin double labelling experiments were carried out. As shown in Figure 3, RyRs formed punctate labelling as described previously.18,22,23 These puncta were arranged around areas of strong phalloidin labelling in both rat and human (Figure 3A and 3B). From the Voronoi diagram based on RyR centroids (see Methods) we calculated the area of contractile apparatus supplied by each couplon release site and exemplar data are shown in Figure 3C and 3D. Figure 3E shows the distribution of RyR supplied areas, which appeared broadly similar in rat and human. However analysis showed that the mean cross-sectional area supplied by each couplon was slightly smaller in rat than in human (0.36 ± 0.03 µm2 compared to 0.49 ± 0.04 µm2, p = 0.048 unpaired t-test df = 8). This difference was not related to a differences in the amount of contractile machinery as measured by the phalloidin label (Figure 3F); in rat 56% ± 4% of cell cross section was labelled compared to 58% ± 4% in human (p = 0.46 df = 9).
Triple labelling was used to clarify the relationships among the SR, RyR clusters and t-system in rat and human cells (Figure 4A). In rat, most RyR clusters were closely aligned to the t-tubules (Figure 4B) as described previously,19 although isolated RyRs could be seen occasionally (arrow in Figure 4B). The organization was similar in human (Figure 4C) with some isolated RyR labelling more than 250 nm from t-tubules. The SR network was labelled with an antibody against SERCA2a (Figure 4C) and overlapped strongly with the t-system labelled by CAV3 and RyR labelling. However, there were areas where SERCA2a labelling that did not overlap with that of the surface membrane, implying that the t-system does not surround each myofibril (arrowhead in Figure 4C). A similar organization was seen in human (Figure 4D). This point can be seen more clearly in Figures 4F and 4G, where t-system labelling tends to follow the outline of areas of phalloidin labelling while the SR clearly crosses them. These data are most simply explained by the t-system following large spaces between groups of closely spaced myofibrils, while the SR surrounds each myofibril as described for EM data.4
The uniformity of Ca release will depend on the spacing between couplons, which are, in turn, dependent on the presence of an extensive t-tubular system. The rat t-system follows a highly branched network forming a complex rete2,9 whereas the human myocytes appear to have a coarser arrangement of wider t-tubules. Our data show this difference clearly, and the reduction in t-tubular complexity is reflected in a slightly larger distance between RyR clusters. This in turn, causes an small increase the area of myofilaments supplied by each couplon.
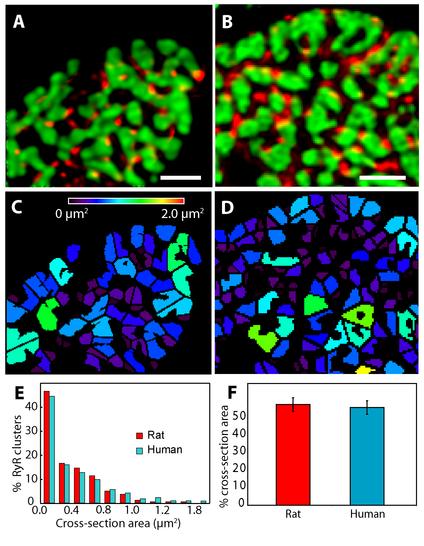
Figure 3. Analysis of the RyR clusters and local contractile regions served in rat and human myocytes. Punctate RyR labelling (red) and the cross-sectional view of the contractile machinery (labelled with Phalloidin; coloured in green) in rat (A) and human (B) ventricular myocytes are shown in overlays. Maps of the local contractile regions served by each RyR cluster, colour-coded for the cross-sectional area are shown for rat (C) and human (D) datasets. Note that most polygons are smaller than 1 µm2. E: Histogram shows the percentage of RyR clusters in rat myocytes as a function of the cross-sectional area of the local contractile machinery. Inset illustrates the equivalent histogram for human data. The mean cross-sectional area of contractile machinery served by an RyR clusters was 0.36 ± 0.03 µm2 (n = 5 cells, mean ± SEM) for rat and 0.49 ± 0.04 µm2 (n = 6 cells) for human cells. F: Bar plot illustrates that there is no difference between the percentage of the cross-sectional cytoplasmic area occupied by phalloidion labelling in rat (56.1 ± 3.8 %) and human myocytes (57.8 ± 3.9 %) analysed. Scale bars: A- 2 µm and B- 2 µm.
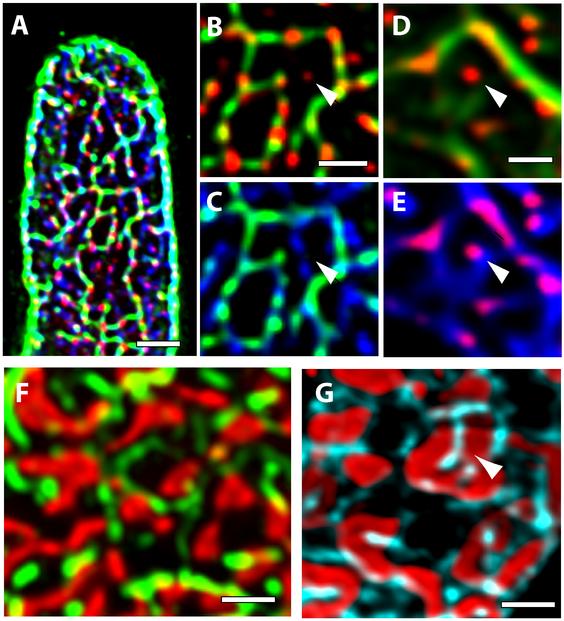
Figure 4. Relationship between t-tubules, SR network, RyR clusters and myofibrils. A: A tri-colour overlay of RyR (red), CAV3 (green) and SERCA2a (blue) labelling in a rat ventricular myocyte shown in transverse view. B: Magnified view of the overlay between CAV3 and RyR in a rat myocyte shows some RyR clusters lacking overlap with t-tubules (example shown with arrowhead). C: The overlay between SERCA2a and CAV3 image in the same region shows additional SR tubules present in the indicated regions lacking CAV3 labelling. D: The overlay between WGA (green) and RyR labelling (red) in a similar region in a human myocyte also showed RyR clusters far from any t-tubular staining (arrowhead). E: The WGA (green) and SERCA2a (blue) overlay in the same region illustrates a finer network of the SR in comparison to the t-tubules. F: The overlay between Phalloidin (red) and CAV3 staining (green) in a rat ventricular myocyte illustrates the poor overlap between the two labels. G: The SERCA2a (cyan) and Phalloidin staining (red) in a similar region shows how the elements of the SR network (arrowhead) appear to bisect the irregular shapes reported by the phalloidin. Scale bars: A- 2 µm; B,D, F & G: 1 µm.
The widths of many rat tubules are at the limit of resolution that is achievable through confocal microscopy.9 While the radial disposition of t-tubules in rabbit and human appears similar, we did not detect the pronounced varicosities described for rabbit,24 although the tubule diameter seemed similar (i.e. 250 to 500 nm). However, we point out that WGA does not report all t-tubules but seems to preferentially label the larger t-tubules in rabbit and human. WGA binds to sugar residues such as sialic acid and provides a marker for glycosylated proteins present on the cell surface.25,26 This difference in labelling pattern between rat and human suggests some difference in the distribution and extent of glycosylated protein, but we cannot ascribe such changes to any particular protein at this time. In both rat and human, t-tubules are preferentially located in larger gaps between myofibrils and in the human the myofibril bundles tend to form more plate-like structures (in cross section) than the rat. We suggest that it is this fundamental difference in contractile machinery that gives rise to the more radial organization of the t-tubular system in rabbit and human compared to rat.
The whole cell calcium current density in human has been reported to be twice that of rat which may have implications for the activation of CICR.27 However our data suggest that the t-tubular membrane area of rat may be half that of human (from a mean tubule diameter of 250 nm (rat) compared to 350 nm (human) and 0.82 µm/µm3 t-tubule (rat) compared to 0.23 µm/µm3 (human)). This difference in membrane area suggests that the local calcium current density available to trigger CICR may not be so different in these species. On the other hand, if the calcium current was restricted almost exclusively to the couplon as suggested by recent calcium channel antibody labelling,28 the rat would have only half the trigger for CICR compared to human. A solution to this discrepancy may reside in our recent data showing that only ∼51% of DHPRs are within the area of the dyad junction in human.29
We estimated the area of contractile apparatus (as measured by f-actin labelling) served by each RyR cluster. This is an extension of a previous analysis which only measured the cell area served by each RyR cluster.18 The spaces between f-actin bundles are likely to be occupied by mitochondria and t-tubules (e.g. references 30,31) and would not contribute to the diffusible space for intracellular Ca2+ and were therefore excluded from this analysis. Not surprisingly, the analysis resulted in smaller area estimates, however a similar difference between human and rat was observed with the volume of cytoplasm served by each cluster ∼36% larger on average in human myocytes.18 This difference was entirely due to the difference in RyR cluster density between the species, as we detected no significant difference in the volume occupied by f-actin labelling. The amount of RyR labelling per cluster is also down by ∼30% in human myocytes18 which, together with the reduced RyR cluster density is in reasonable agreement with reduced ryanodine binding to human heart homogenates compared to of rat (∼100 compared to 200 fmol/mg protein respectively).32 Taken together, these data could explain a slower rate of rise of the human Ca2+-transient compared to rat.33,34
Most RyR clusters showed strong alignment with t-tubules as reported by CAV3 in rat ventricular myocytes. However, a small fraction of clusters did not coincide with CAV3 labelling, as reported in a previous study.19 Human ventricular tissue samples that were labelled with WGA showed similarly ‘orphaned’ RyR clusters although we cannot be certain that WGA and CAV3 have captured all t-tubule elements. On the other hand, our estimate of t-tubule density in human with a mean t-tubule diameter of 350 nm and 1.9 µm sarcomere spacing would correspond to a t-tubule area of 0.29 µm2/µm3 which is very similar to that of rabbit (0.25 µm2/µm3).35 In addition we can estimate the volume fraction with the assumption of circular t-tubules in human as ∼3% which is similar to that of guinea pig (2.5-3.2%36) and is slightly lower than the direct volume measurement of 3.6% in rat.9 In connection with the latter figure, assuming a mean tubule diameter of 250 nm, the data presented here suggest a volume fraction of 4% for the rat (with the assumption of circular tubules).
In both species, the SERCA2a labelling was more extensive than that of the t-tubular system and the SR linked RyR clusters in regions lacking t-tubules. The loops present in the SR labelling appeared to encircle discrete regions labelled by phalloidin, suggesting that myofibrils are not completely enclosed by the t-tubular system. Such grouping of myofibrillar bundles to sandwich the SR would enhance the SERCA2a-mediated removal of Ca2+ from the contractile machinery, although we note that there is less SERCA2a protein in human myocardium compared to that of the rat.37
This work was supported by the Health Research Council of New Zealand, the Marsden Fund (NZ), the Wellcome Trust (UK), the Lotteries Grant Board (NZ), and the Auckland Medical Research Foundation. We thank Dr. Carolyn Barrett for supplying rabbit tissue.
1. Cheng H, Cannell MB, Lederer WJ. Propagation of excitation-contraction coupling into ventricular myocytes. Pflügers Arch. Eur. J. Physiol. 1994; 428: 415-7.
2. Forssmann WG, Girardier L. A study of the T system in rat heart. J. Cell Biol. 1970; 44: 1-19.
3. Forbes MS, Hawkey LA, Sperelakis N. The transverse-axial tubular system (TATS) of mouse myocardium: its morphology in the developing and adult animal. Am. J. Anat. 1984; 170: 143-162.
4. Fawcett DW, McNutt NS. The ultrastructure of the cat myocardium I: Ventricular papillary muscle. J. Cell Biol. 1969; 42: 1-45.
5. Song LS, Sobie EA, McCulle S, Lederer WJ, Balke CW, Cheng H. Orphaned ryanodine receptors in the failing heart. Proc. Natl. Acad. Sci. USA. 2006; 103: 4305-10.
6. Cannell MB, Crossman DJ, Soeller C. Effect of changes in action potential spike configuration, junctional sarcoplasmic reticulum micro-architecture and altered t-tubule structure in human heart failure. J. Muscle Res. Cell Motil. 2006; 27: 297-306.
7. Louch WE, Mørk HK, Sexton J, Strømme TA, Laake P, Sjaastad I, Sejersted OM. T-tubule disorganization and reduced synchrony of Ca2+ release in murine cardiomyocytes following myocardial infarction. J. Physiol. 2006; 574: 519-33.
8. Lyon AR, MacLeod KT, Zhang Y, Garcia E, Kanda GK, Lab MJ, Korchev YE, Harding SE, Gorelik J. Loss of T-tubules and other changes to surface topography in ventricular myocytes from failing human and rat heart. Proc. Natl. Acad. Sci. USA. 2009; 106: 6854-9.
9. Soeller C, Cannell MB. Examination of the transverse-tubular system in living cardiac rat myocytes by 2-photon microscopy and digital image-processing techniques. Circ. Res. 1999; 84: 266-75.
10. Sachse FB, Savio-Galimberti E, Goldhaber JI, Bridge JH. Towards computational modeling of excitation-contraction coupling in cardiac myocytes: reconstruction of structures and proteins from confocal imaging. Pac. Symp. Biocomput. 2009; 328-39.
11. Amsellem J, Delorme R, Souchier C, Ojeda C. Transverse-axial tubular system in guinea pig ventricular cardiomyocyte: 3D reconstruction, quantification and its possible role in K+ accumulation-depletion phenomenon in single cells. Biol. Cell 1995; 85: 43-54.
12. Fabiato A. Simulated calcium current can both cause calcium loading in and trigger calcium release from the sarcoplasmic reticulum of a skinned canine cardiac Purkinje cell. J. Gen. Physiol. 1985; 85: 291-320.
13. Grantham CJ, Cannell MB. Ca2+ influx during the cardiac action potential in guinea pig ventricular myocytes. Circ. Res. 1996; 79: 194-200.
14. Sipido KR, Maes M, Van de Werf F. Low efficiency of Ca2+ entry through the Na+-Ca2+ exchanger as trigger for Ca2+ release from the sarcoplasmic reticulum. A comparison between L-type Ca2+ current and reverse-mode Na+-Ca2+ exchange. Circ. Res. 1997; 81: 1034-44.
15. Sobie EA, Cannell MB, Bridge JH. Allosteric activation of Na+-Ca2+ exchange by L-type Ca2+ current augments the trigger flux for SR Ca2+ release in ventricular myocytes. Biophys. J. 2008; 94: L54-6.
16. Bers DM. Cardiac excitation-contraction coupling. Nature 2002; 415: 198-205.
17. Cheng H, Lederer WJ, Cannell MB. Calcium sparks: elementary events underlying excitation-contraction coupling in heart muscle. Science 1993; 262: 740-4.
18. Soeller C, Crossman D, Gilbert R, Cannell MB. Analysis of ryanodine receptor clusters in rat and human cardiac myocytes. Proc. Natl. Acad. Sci. USA. 2007; 104: 14958-63.
19. Jayasinghe ID, Cannell MB, Soeller C. Organization of ryanodine receptors, transverse tubules and sodium-calcium exchanger in rat myocytes. Biophys. J. 2009; 97: 2664-73.
20. Evans AM, Cannell MB. The role of L-type Ca2+ current and Na+ current-stimulated Na/Ca exchange in triggering SR calcium release in guinea-pig cardiac ventricular myocytes. Cardiovasc. Res. 1997; 35: 294-302.
21. Franzini-Armstrong C, Protasi F, Ramesh V. Shape, size, and distribution of Ca2+ release units and couplons in skeletal and cardiac muscle. Biophys. J. 1999. 77: 1528-39.
22. Scriven DR, Dan P, Moore ED. Distribution of proteins implicated in excitation-contraction coupling in rat ventricular myocytes. Biophys. J. 2000; 79: 2682-91.
23. Chen-Izu Y, McCulle SL, Ward CW, Soeller C, Allen BM, Rabang C, Cannell MB, Balke CW, Izu LT. Three-dimensional distribution of ryanodine receptor clusters in cardiac myocytes. Biophys. J. 2006; 91: 1-13.
24. Savio-Galimberti E, Frank J, Inoue M, Goldhaber JI, Cannell MB, Bridge JH, Sachse FB. Novel features of the rabbit transverse tubular system revealed by quantitative analysis of three-dimensional reconstructions from confocal images. Biophys. J. 2008; 95: 2053-62.
25. Bhavanandan VP, Umemoto J, Banks JR, Davidson EA. Isolation and partial characterization of sialoglycopeptides produced by a murine melanoma. Biochemistry 1977; 16: 4426-37.
26. Peters BP, Ebisu S, Goldstein IJ, Flashner M. Interaction of wheat germ agglutinin with sialic acid. Biochemistry 1979. 18: 5505-11.
27. Horiuchi-Hirose M, Kashihara T, Nakada T, Kurebayashi N, Shimojo H, Shibazaki T, Sheng X, Yano S, Hirose M, Hongo M, Sakurai T, Moriizumi T, Ueda H, Yamada M. Decrease in the density of t-tubular L-type Ca2+ channel currents in failing ventricular myocytes. Am. J. Physiol. Heart Circ. Physiol. 2010; 300: H978-88.
28. Scriven DR, Asghari P, Schulson MN, Moore ED. Analysis of Cav1.2 and ryanodine receptor clusters in rat ventricular myocytes. Biophys J. 2010; 99: 3923-9.
29. Crossman DJ, Ruygrok PR, Soeller C, Cannell MB. Changes in the organization of excitation-contraction coupling structures in failing human heart. PLoS One 2011; 6: e17901.
30. Sommer JR, Johnson EA. Cardiac muscle. A comparative study of Purkinje fibers and ventricular fibers. J. Cell Biol. 1968; 36:(3) 497-526.
31. Soeller C, Jayasinghe ID, Li P, Holden AV, Cannell MB. Three-dimensional high-resolution imaging of cardiac proteins to construct models of intracellular Ca2+ signalling in rat ventricular myocytes. Exp. Physiol. 2009; 94: 496-508.
32. Schumacher C, Konigs B, Sigmund M, Kohne B, Schondube F, Vob M, Stein B, Weil J, Hanrath P. The ryanodine binding sarcoplasmic reticulum calcium release channel in nonfailing and in failing human myocardium. Naunyn-Schmiedeberg's Arch. Pharmacol. 1995; 353: 80-5.
33. Cannell MB, Berlin JR, Lederer WJ. Effect of membrane potential changes on the calcium transient in single rat cardiac muscle cells. Science 1987; 238: 1419-23.
34. Beuckelmann DJ, Näbauer M, Erdmann E. Intracellular calcium handling in isolated ventricular myocytes from patients with terminal heart failure. Circulation 1992; 85: 1046-55.
35. Page E, Surdyk-Droske M. Distribution, surface density, and membrane area of diadic junctional contacts between plasma membrane and terminal cisterns in mammalian ventricle. Circ. Res. 1979; 45: 260-7.
36. Forbes MS, van Neil EE. Membrane systems of guinea pig myocardium: ultrastructure and morphometric studies. Anat. Rec. 1988; 222: 362-79.
37. Su Z, Li F, Spitzer KW, Yao A, Ritter M, Barry WH. Comparison of sarcoplasmic reticulum Ca2+-ATPase function in human, dog, rabbit, and mouse ventricular myocytes. J. Mol. Cell. Cardiol. 2003; 35: 761-67.