The β1- and β2 adrenergic receptors dominate the cardiac response to catecholamines through their coupling to Gs proteins and to the production of the common second messenger cAMP (Xiang & Kobilka, 2003). Cyclic AMP controls the catecholamine-dependent changes in rate, force and speed of contraction of the heart. However, selective stimulation of these two receptor subtypes leads to clearly distinct physiological and pathophysiological responses. In healthy cardiac myocytes β1- but not β2ARs stimulate the cAMP-dependent protein kinase mediated phosphorylation of phospholamban and cardiac contractile proteins (Xiao, 2001). Heart-specific overexpression of β1- but not of β2AR in transgenic mice induces progressive cardiac hypertrophy and heart failure (Engelhardt et al., 1999). Such subtype specific differences have been attributed to distinct patterns of cAMP compartmentation from the two receptor subtypes observed using electrophysiological and fluorescent biosensors (Nikolaev et al., 2006). However, the exact distribution of the β1- and β2ARs is still elusive. Unfortunately, methods to detect such localized effects have so far been limited.
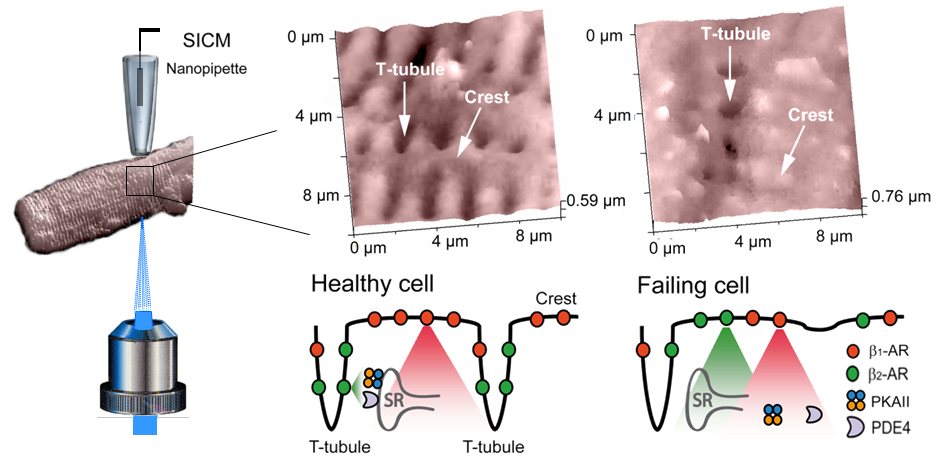
Our new functional approach combines scanning ion conductance microscopy (SICM) with measurements of cAMP production by fluorescence resonance energy transfer (FRET) after localized receptor stimulation (Nikolaev et al., 2010). SICM is a non-optical method which uses a nanopipette as a scanning probe to image the surface topography of living cells and allows to resolve the structural features of cardiac myocytes such as Z-grooves and transverse (T) tubules with a resolution equal to the pipette's inner diameter (Novak et al., 2009), typically ∼20-50 nm (the Figure). After the acquisition of the cell surface topography we precisely position the pipette onto various membrane regions of defined morphology to locally apply agonists and antagonists of the βARs (The Figure). Local stimulation is achieved by applying pressure into the pipette while constantly superfusing the cells with the buffer/antagonist solution from the side (Nikolaev et al., 2006). Upon selective stimulation of β1- and β2ARs we measure cAMP synthesis in defined areas of the cells by FRET microscopy using a sensitive cAMP sensor Epac2-camps (Nikolaev et al., 2004).
To study the localization of receptors and cAMP signals in cardiomyocytes isolated from healthy adult rat hearts, we locally stimulated β1- and β2ARs either in the T-tubule or on the crest of the cell. Selective stimulation of β1ARs in both regions resulted in a robust decrease of FRET, reflecting the stimulation of cAMP synthesis by the receptors localized in different parts of the membrane. In sharp contrast, β2AR selective stimulation led to cAMP signals only in the T-tubules, but not in the outer regions of the membrane.
In a rat model of chronic heart failure, β2ARs redistributed from the transverse tubules to the cell crest, leading to diffuse receptor-mediated cAMP signaling. We showed that surface morphology and T-tubular structure are significantly disrupted in chronic heart failure cells (Lyon et al., 2009), and that impacts on the redistribution of sarcolemmal β2AR and localized secondary messenger signaling.
Engelhardt S, Hein L, Wiesmann F, Lohse MJ. (1999) Proc Natl Acad Sci USA 96: 7059-7064.
Lyon AR, MacLeod KT, Zhang Y, Garcia E, Kanda GK, Lab MJ Korchev YE, Harding SE, Gorelik J. Proc Natl Acad Sci USA 2009; 106(16):6854-6859.
Nikolaev VO, Bunemann M, Hein L, Hannawacker A, Lohse MJ. (2004) J Biol Chem 279(36): 37215-37218.
Nikolaev VO, Bunemann M, Schmitteckert E, Lohse MJ, Engelhardt S. Circulation Research 2006; 99(10):1084-1091.
Nikolaev VO, Moshkov A, Lyon AR, Miragoli M, Novak P, Paur H, Lohse MJ, Korchev YE, Harding SE, Gorelik J. (2010) Science 327: 1653-7.
Novak P, Li C, Shevchuk AI, Stepanyan R, Caldwell M, Hughes S et al. (2009) Nature Methods 6: 279-281.
Xiang Y, Kobilka BK.Science 2003; 300(5625):1530-1532.
Xiao RP. (2001) Science Signalling 2001(104):RE15.
