Inwardly rectifying potassium (Kir) channels maintain the membrane resting potential and regulate the action potential in electrically excitable cells. We investigate the Kir1.1b isoform predominantly expressed in the kidney; Kir2.1 expressed throughout the heart and nervous system; and Kir3.2 expressed in the brain, pituitary and testis. Dysfunction of these Kir channels has been implicated in various neurological disorders, such as seizure generation for Kir3.2, Bartters syndrome for Kir1.1, and type-1 Andersen syndrome for Kir2.1. There are a number of unresolved issues regarding the conductance properties of Kir channels. Firstly, the physical basis of inward rectification is thought to occur due to a combination of blockage by intracellular cations and an unspecified intrinsic gating process. Second, the structural changes required for gating are yet to be elucidated. Third, the recognition mechanism involved in antagonist binding to the external vestibule remains largely unexplored.
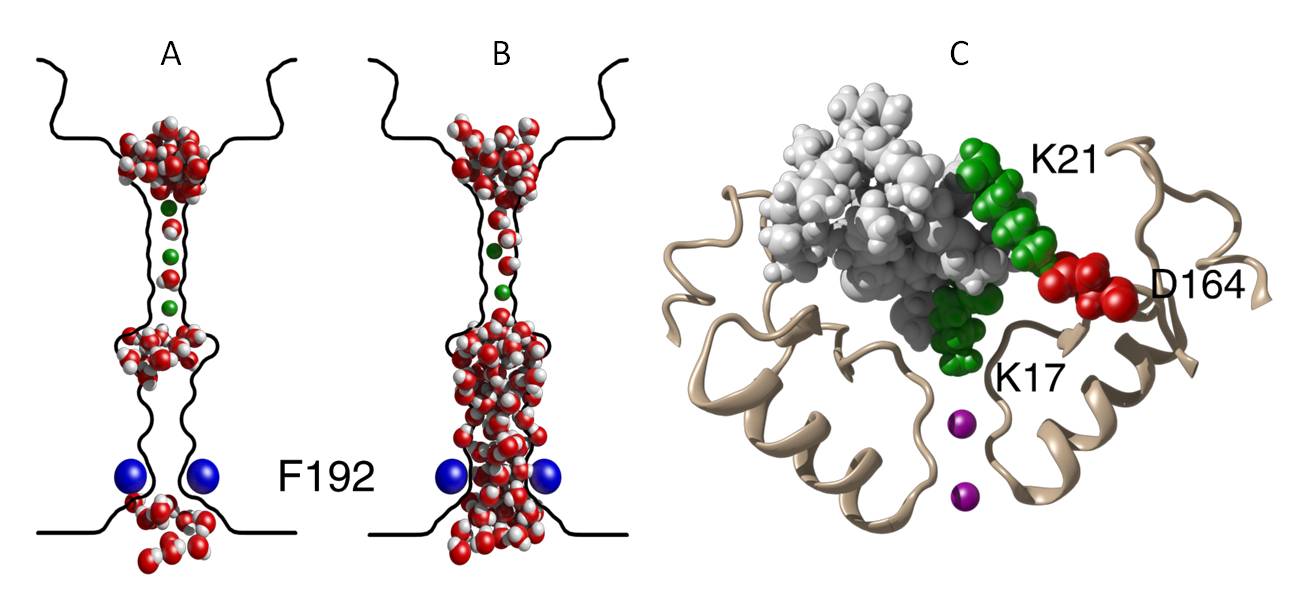
The crystal structure of Kir3.2 has recently become available, creating an opportunity to computationally investigate its conductance properties. Kir1.1b and Kir2.1 homology models are generated using the crystal structure of Kir2.2. Using molecular and Brownian dynamics simulations, we elucidate several conductance properties of these Kir channels. We show that the pore is closed by a hydrophobic gating mechanism similar to that observed in Kv1.2 (illustrated in Figure A and B). Once open, potassium ions move into, but not out of, the cell. The asymmetrical current-voltage relationship arises from the lack of negatively charged residues at the narrow intracellular mouth of the channel. When the residues guarding the intracellular gate are mutated to glutamate residues the channel no longer exhibits inward rectification. In Kir3.2, inward rectification is restored when it becomes blocked by intracellular Mg2+.

Although a number of drugs including anti-arrhythmia, anti-psychotic and anti-depressants block Kir channels, these compounds tend to be non-selective and block the pores at relatively high concentrations. Tertiapin (shown in Figure C), a polypeptide toxin isolated from the honey bee, is known to block the pores of Kir channels with nanomolar to micromolar affinity. Tertiapin binds to Kir1.1b with an affinity of 2 nM, whereas Kir2.1 is relatively insensitive with an affinity of 20 μM. We identify critical residues in the toxin and channels for the formation of a stable complex and determine binding affinities. Our binding affinities compare reasonably with experimentally determined values. By mutating the isoleucine residue at position 8 to arginine we improve the affinity to Kir2.1 to 0.7 nM. Our findings will further the understanding of Kir channels and could lead to the development of targeted pharmaceuticals.