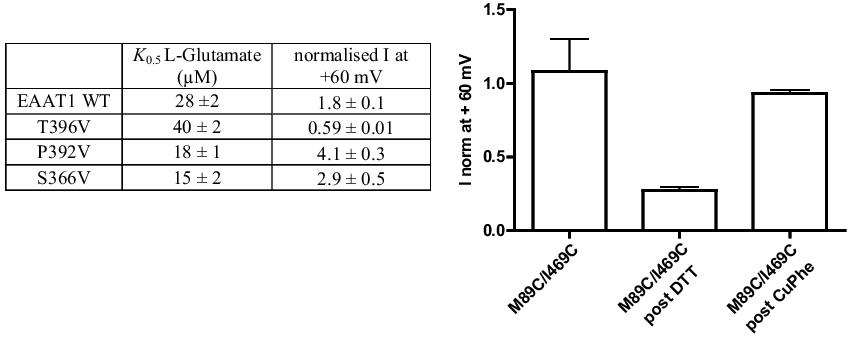
The concentration of glutamate within a glutamatergic synapse is tightly regulated by excitatory amino acid transporters (EAATs). EAATs function not only as glutamate transporters, but also as substrate activated chloride (Cl−) channels. Several crystal structures of the EAAT homologue, GltPh, at different stages of the transport cycle have been solved (Boudker et al., 2007; Reyes et al., 2009; Verdon et al., 2012; Yernool et al., 2004). In the most recent structure to be solved (Verdon et al., 2012) a small cavity lined by polarizable residues, several of which have been implicated in Cl− permeation (Ryan et al., 2004), has been identified. We hypothesize that throughout the transport cycle this cavity opens up to form the Cl− channel. In this study, site directed mutagenesis of EAAT1 and electrophysiology have been utilized to determine if this cavity forms part of the Cl− permeation pathway. Additionally, double cysteine mutants were generated in a cys-less EAAT1 background and analysed in an attempt to trap the protein in a purely Cl− conducting state to assist with further structural studies of GltPh. When WT and mutant EAAT1 transporters are expressed in Xenopus laevis oocytes, the current observed at +60 mV is primarily attributed to Cl− conductance. For this reason, current at +60 mV is indicative of Cl− channel function. When residues T396, S366 and P392 are mutated to valine, alterations in Cl− channel function occur without effecting glutamate transport (See table for K0.5 values and currents at +60 mV) thus indicating a role for these residues in the formation of the Cl− permeation pathway.

After treatment of the double cysteine mutant M89C/I469C with the reducing agent dithiothreitol (DTT), the current at +60 mV decreases. Subsequent treatment with the oxidizing reagent copper phenanthroline (CuPhe) leads to a recovery of the current at +60 mV (see Figure). These results suggest that these two cysteine residues can form a spontaneous crosslink and trap the transporter in a Cl− conducting state.
Boudker O, Ryan RM, Yernool D, Shimamoto K, Gouaux E (2007) Coupling substrate and ion binding to extracellular gate of a sodium-dependent aspartate transporter. Nature 445: 387-393.
Reyes N, Ginter C, Boudker O (2009) Transport mechanism of a bacterial homologue of glutamate transporters. Nature 462: 880-885.
Ryan RM, Mitrovic AD, Vandenberg RJ (2004) The chloride permeation pathway of a glutamate transporter and its proximity to the glutamate translocation pathway. Journal of Biological Chemistry 279: 20742-20751.
Verdon G, Boudker O (2012) Crystal structure of an asymmetric trimer of a bacterial glutamate transporter homolog. Nature Structural and Molecular Biology 19: 355-357.
Yernool D, Boudker O, Jin Y, Gouaux E (2004) Structure of a glutamate transporter homologue from Pyrococcus horikoshii. Nature 431: 811-818.