1. Prenatal alcohol exposure impairs kidney development resulting in a reduced nephron number. However, the mechanism through which alcohol acts to disrupt renal development is largely unknown. Retinoic acid is critically involved in kidney development and it has been proposed that a diminished concentration is a contributing factor to fetal alcohol syndrome.
2. In this study we proposed that the ethanol-induced inhibition of ureteric branching morphogenesis and glomerular development in the cultured rat kidney would be ameliorated by co-culture with exogenous retinoic acid, and that examining the expression profile of key genes involved in the development of the kidney would provide insights into potential molecular pathways involved.
3. Whole rat metanephroi cultured in the presence of exogenous retinoic acid without ethanol appeared larger and had significantly more ureteric branch points, tips and glomeruli than metanephroi cultured in control media. Those cultured in the presence of ethanol alone (0.2%) had 20% fewer ureteric branch points, tips and glomeruli, which was ameliorated by co-culture with retinoic acid.
4. Gene expression analysis identified changes in the expression levels of enzymes involved in the metabolism of alcohol, in conjunction with changes in key regulators of kidney development including cRET.
5. These results demonstrate that the teratogenic effects of alcohol in vitro on kidney development resulting in reduced ureteric branching morphogenesis and glomerular development can be ameliorated through co-culture with retinoic acid. These results provide the foundation for future research into the mechanism through which alcohol acts to disrupt kidney development.
Alcohol (ethanol) is commonly consumed by pregnant women in western society despite the knowledge that it is a known teratogen and the effects of chronic ethanol exposure on the developing fetus have been well established.1-3 However, the effects of acute ethanol exposure (equivalent of 3-4 standard drinks on one or two occasions) have been less well characterised. It has been demonstrated that acute ethanol exposure in animal studies results in neuroapoptosis,4,5 reduced brain growth6 and pulmonary alveolar dysfunction.7 Furthermore, we have recently shown that acute ethanol exposure in the pregnant rat results in fetal growth restriction and a 10% elevation in blood pressure at 6 months of age in conjunction with a 15% reduction in nephron number identified at postnatal day 30.8 This is important as the reduced nephron endowment is permanent and has been linked to the development of adult-onset diseases, such as hypertension.9,10 However, the number of studies investigating the mechanism through which ethanol affects the developing kidney is limited. In chronic studies of ethanol-exposed animals, adverse affects on kidney development, through a reduction in DNA and protein content were observed.11
Mammalian kidney development involves complex molecular reciprocal interactions between the ureteric tree and the surrounding metanephric mesenchyme.12,13 Branching of the ureteric tree is a critical process and plays a significant role in determining nephron number, as new nephrons only form adjacent to ureteric tips. The reduced nephron number observed following fetal ethanol exposure may be a result of reduced ureteric branching. In support of this we have recently identified that embryonic kidneys cultured in the presence of ethanol for 48 h had 15% fewer ureteric branch points and tips and 20% fewer developing glomeruli.8 However, it is largely unknown through which molecular pathways ethanol may be exerting its effects.
Retinoic acid (RA), a Vitamin A derivative, and its receptors play critical roles in kidney development.4,14 In an early study, Wilson and colleagues (1953) showed that maternal vitamin A deficiency resulted in renal hypoplasia in rats which could be prevented by vitamin A administration to pregnant animals.15 Ethanol metabolism and RA synthesis share a common enzymatic pathway, which includes alcohol dehydrogenase (ADH) and aldehyde dehydrogenase (ALDH). It has been demonstrated in animal models that following ethanol exposure there is a transient reduction in the concentration of plasma RA.16,17 In addition, it has been shown that the number of glomeruli in whole cultured rat metanephroi is increased after exogenous RA administration.18 It has been proposed that a transient reduction in RA levels is a contributing factor to the developmental presentation of fetal alcohol syndrome (FAS).17,19 In the present study, we hypothesised that ethanol-induced inhibition of ureteric branching morphogenesis in rat metanephric culture is due to decreased RA and would thus be ameliorated by co-culture with exogenous RA.
To further delineate the relationship between alcohol, RA and renal development we examined the expression levels of the key genes involved in branching morphogenesis [glial cell line-derived neurotrophic factor (GDNF), and its receptor tyrosine kinase receptor RET (cRET)], and enzymes common to the RA and alcohol metabolism pathways [alcohol dehydrogenase class 1 (ADH-1), aldehyde dehydrogenase 1 and 2 (ALDH-1a2, ALDH-2) and the retinoic acid receptor RXRα].
At the end of the culture period, metanephroi were fixed in methanol at −20°C for a minimum of 15 min. After fixation, for visualisation of the ureteric tree, metanephroi were washed briefly in 1% Tween 20 (PBST) and incubated with primary antibody (monoclonal mouse anti-pan cytokeratin (Sigma-Aldrich)) at a dilution of 1:100 at 37°C for 2 h. Metanephroi were then washed thoroughly in PBST before addition of the secondary antibody; Alexa 488 goat anti-mouse IgG (Molecular Probes, Eugene, OR) at a dilution of 1:100 at 37°C for 2 h. Metanephroi were then washed in PBST before mounting in PBS/glycerol mounting media (Sigma-Aldrich).
Following immunostaining, metanephroi were visualized using an epifluorescence microscope (Olympus) and manually skeletonised. Ureteric branch points and tips were counted. Branch points were defined as the intersection of three or more branches (lines on the skeletonised image).
Following fixation, those metanephroi to be prepared for glomerular number estimation were briefly washed in PBS. They were incubated in 50mM NH4Cl at room temperature for 1 h following which the tissue was permeabilized with 0.1% saponin in PBS for 1 h at room temperature. After digestion with 2% H2O2 in methanol at room temperature for 30 min, metanephroi were washed in 0.1% saponin in PBS for 30 min. Metanephroi were then incubated in 0.1U/ml of neuraminidase from Vibrio cholerae (Sigma-Aldrich) in 1% CaCl2 in PBS for 2 h at 37°C, before being washed twice for 30 min in 0.2% saponin in PBS. Metanephroi were then incubated in rhodamine-labelled peanut agglutinin (PNA) (50μg/ml; Sigma-Aldrich) in 0.3% Triton in PBS with 0.1M ions (MgCl2, MnCl2, CaCl2) overnight at 4°C. Metanephroi were then extensively washed in 0.1% saponin to remove excess PNA and then mounted on cavity slides with fluorescence preserving mounting media (Sigma-Aldrich).
Following lectin PNA histochemistry, metanephroi cultured for 5 days were observed under an epifluorescence microscope for direct counting of glomeruli.
Gene expression analysis was performed in metanephroi cultured for 48 h under the six conditions. Three to six pools of RNA were collected with each pool containing 10 metanephroi derived from 10 embryos and at least 5 dams. At the end of the culture period, total RNA was extracted using RNeasy extraction kits (Qiagen). One μg of each RNA sample was reverse transcribed as previously described.20
Levels were determined via real-time PCR using an Applied Biosystems One step Machine. Sequences and optimal working concentrations have previously been published.8,20 A comparative cycle of threshold fluorescence (CT) method was used with 18S as an internal control.
The CT values for 18S were subtracted from the CT value for the gene of interest to give a ΔCT for each sample. The ΔCT of the calibrator (the mean ΔCT for the control group (0% ethanol) was then subtracted from each sample to give a ΔΔCT value. This was then inserted into the equation 2-ΔΔCT to give a final expression level relative to the calibrator.
Values are means ± SEM except where otherwise indicated. All data for the 0% ethanol & 10nM RA and 0% ethanol & 20nM RA were merged as there were no significant differences between these two groups for any of the parameters examined. This was also the case for the 0.2% ethanol & 10nM RA and 0.2% ethanol & 20nM RA groups. A one-way ANOVA was utilised for analysis of ureteric branching morphogenesis, glomerular number and real-time PCR data across the four groups. A Tukey’s post-hoc test was utilised to identify differences between groups. Significance was accepted at p<0.05.
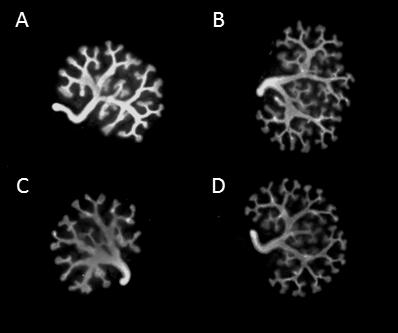
Metanephroi cultured in the presence of RA (with or without alcohol) appeared larger than metanephroi cultured without RA (Figure 1). Furthermore, culture of metanephroi in the presence of ethanol did not alter the qualitative appearance of the ureteric tree (Figure 1). However, quantitative analysis of the ureteric tree after 2 days of culture in the presence of ethanol (0.2%) identified a 20% reduction in ureteric branch points and tips compared to control metanephroi (Figure 2). Quantitative analysis revealed that addition of RA to both control and ethanol cultures significantly increased the number of ureteric branch points in comparison to metanephroi cultured in control media or media containing ethanol (Figure 2). A similar effect was observed for ureteric tips (Figure 2).

Figure 1. Immunofluorescence images of the ureteric tree of whole rat metanephroi. The panels show results after 48 h culture in media containing: A: 0% ethanol; B: 0% ethanol and RA; C: 0.2% ethanol; D: 0.2% ethanol and RA. Note: results for RA at 10nM or 20nM are combined (see text for details).
Culture in the presence of ethanol also adversely affected glomerular development. Metanephroi cultured for 5 days in the presence of ethanol contained 25% fewer glomeruli than metanephroi cultured in control media (Figure 2). The addition of exogenous RA to both control cultures and ethanol cultures significantly increased the number of glomeruli at 5 days in comparison to metanephroi cultured in control media or ethanol-supplemented media. Significantly, co-culture with ethanol and RA resulted in a similar glomerular number to that observed in control cultures (Figure 2).
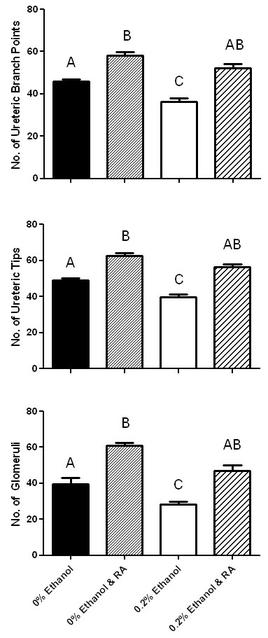
Figure 2. The effect of ethanol and RA on ureteric branch points and tips. Shown are the number of branch points (top graph) and tips (middle graph) after 48 h and glomerular number (bottom graph) after 5 days culture with 0% or 0.2% ethanol with/without exogenous RA. Data are mean ± SEM and were analysed using a one-way ANOVA with a Tukey’s post-hoc test. Groups that do not share a common letter are significantly different from each other, p<0.05. N=10-15 metanephroi per group.
Compared to vehicle, the relative mRNA levels of ADH-1 were significantly elevated in the presence of ethanol and RA. Ethanol alone caused an intermediate effect that was not different to either vehicle (p=0.09) or 2% EtOH & RA (p=0.07). However, there was no change in the relative levels for ALDH-1a2. Similarly, relative levels of ALDH-2 mRNA and GDNF mRNA were similar in the four culture groups. Interestingly, the expression level of the major GDNF receptor cRET was not significantly affected by culture in ethanol, but when cultured in the presence of RA its expression level was significantly increased. The relative expression level of the retinoic acid receptor RXRα tended to be lower in kidneys cultured in 0% ethanol supplemented with RA (p=0.07), ethanol (p=0.09) and ethanol supplemented with RA (p=0.08) compared to vehicle (Figure 3). In addition, there was no change in the expression of the apoptotic markers, BAX and Bcl-2 between groups (data not shown).
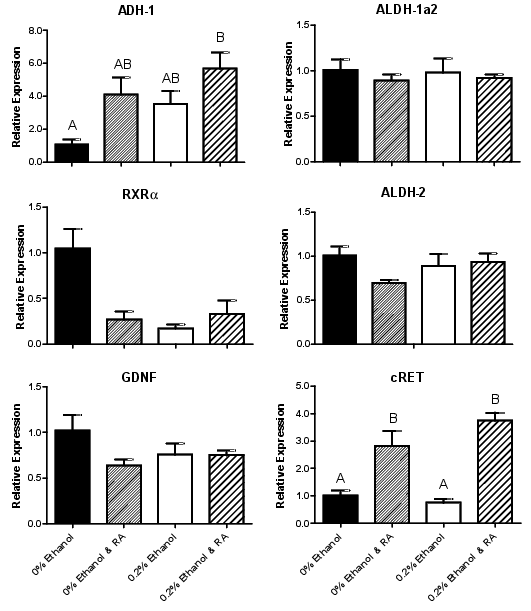
Figure 3. Real-time PCR analysis performed on E14 metanephroi cultured for 48 h, for ADH-1, ALDH-1a2, ALDH-2, RXRα, cRET and GDNF. Data are mean ± SEM, and were analysed using a one-way ANOVA with a Tukey’s post-hoc test, N=3-6 RNA pools per group. Groups that do not share a common letter are significantly different from each other, p<0.05.
This study has demonstrated that the effects of ethanol on metanephric kidney development in vitro can be ameliorated by the presence of exogenous RA. Through the use of whole metanephric culture we have demonstrated that RA can prevent the reduction in number of ureteric branch points, ureteric tips and glomeruli in kidney’s exposed to alcohol in vitro. Real-time PCR identified changes in gene expression of an enzyme involved in ethanol metabolism as well as genetic regulators of kidney development, providing further evidence for a role of RA in preventing the inhibitory effects of ethanol on kidney development. Most importantly, RA was able to increase expression levels of cRET, a target receptor for GDNF, which is a critical regulator of ureteric branching morphogenesis.
In recent years we have demonstrated that in addition to the well documented effects of prenatal ethanol exposure on the developing central nervous system, ethanol exposure can disrupt kidney development such that offspring have a reduced nephron endowment.3,8 The mechanisms through which alcohol acts to disrupt kidney development, resulting in a deficit in nephron number, are unknown. However, previous work undertaken within our laboratory has established a link between a reduction in ureteric branching morphogenesis and the observed reduction in nephron number in vivo. It was identified that culture in the presence of ethanol at the same concentration used within these current studies, was able to reduce the number of ureteric branch points, tips and glomeruli by 20%.8 RA, the active derivative of vitamin A synthesis, is a promoter of kidney development in vivo and in vitro.18,21-23 As both vitamin A synthesis and ethanol metabolism employ a common enzymatic pathway (see Figure 4), it has been proposed that diminished RA concentrations may contribute to fetal alcohol syndrome.17,19 The effect of exogenous RA on metanephric development in vitro and nephron endowment in vivo has been reported in several studies, with conflicting outcomes. Most studies have reported that culture in the presence of RA significantly increases the number of glomeruli formed.18,24,25 For example, Vilar and colleagues (1996)18 found that addition of RA at a concentration of 100nM, 10-fold higher than the concentration used in the current study, resulted in a 200% increase in nephron number after 6 days in culture. However, Weller and colleagues (1991)25 found that RA failed to induce additional glomerular development in isolated mouse metanephroi after 72 h in culture. Results from the current study have demonstrated that addition of RA, at a significantly lower concentration than that previously used, was able to stimulate kidney development, restoring the number of ureteric branch points, ureteric tips and glomeruli to levels observed in control cultured kidneys. In vivo, RA administration during pregnancy can increase nephron number in the growth restricted rat pup.26 However, administration in the prematurely delivered baboon had no effect of nephrogenesis.27 Together, these studies suggest the effects of RA are dependent upon concentrations to which the kidney is exposed and time of administration.
Gene expression studies demonstrated an effect of ethanol and RA on the expression of enzymes involved in ethanol metabolism and RA synthesis. ADH is the first enzyme responsible for the conversion of ethanol to acetaldehyde. There are a number of isoforms of ADH but ADH-1 rather than ADH-3 or ADH-4, is the predominant from in the developing epithelia of the kidney.28 It was thus not surprising that in the presence of ethanol, there tended to be elevated ADH-1 expression. Compared to vehicle, ADH-1 mRNA expression was only statistically increased in the EtOH plus RA group however ADH-1 expression in the EtOH group expression also tended to be elevated. In fact, expression levels in the EtOH group were not statistically different compared to either vehicle, or the EtOH plus RA groups. However, what was unexpected was that ADH expression was increased to a similar degree by RA alone. This could have been a product of RA self-regulating the expression of the enzymes that are needed for its synthesis.29,30
ALDH, which is the next enzyme in the metabolism of ethanol, in addition to metabolising retinal to RA, also has a number of isoforms many of which are present in only some species.31 In the rat, ALDH1a2 and ALDH-2 are the predominant forms. In this study, the expression of ALDH-1a2 was unchanged While it would be expected that ALDH would have a similar expression to that of ADH, whereby it would be increased in the presence of ethanol and RA, the lack of difference between the groups could be attributed to the many isoforms of ALDH that exist.32 In the developing kidney, at least in the mouse, it is ALDH-2 which is most highly expressed.33 Although ALDH-2 was also unchanged across the four groups, there was a tendency for decreased levels in the RA group. We could speculate that expression of this enzyme has been down-regulated in the presence of high levels of RA to prevent further increases which may lead to toxic levels of RA.
An unexpected finding was that there was a strong tendency for decreased expression of the retinoic acid receptor RXRα in the treatment groups that had RA added. While it is expected that in the presence of ethanol there would be a down-regulation of RXRα, a consequence of ethanol diminished RA concentrations, the down-regulation in the RA supplemented groups is intriguing. While it would be expected that in the presence of RA there would be an increase in the expression of RXRα, the lack of change could be an indication of RA signalling through a different receptor. It has been well established that there are many different receptors involved in the signalling of RA, and that the down-regulation of RXRα could be a product of RA promoting kidney growth through a different RA receptor.29,30,34
We have previously described reduced expression of GDNF, a key regulator of kidney development, in embryonic kidneys following in vivo ethanol exposure.8 In the current study, there were no significant changes in GDNF expression following ethanol exposure, although levels tended to be decreased in all treatment groups compared to control. While we did not identify a change in GDNF expression, we did identify a change in the expression of its receptor, cRET, whereby in the presence of RA either alone or in combination with ethanol, expression was significantly up-regulated. Under normal culture conditions the expression of cRET declines with time in culture.34 One can therefore speculate that the addition of RA to the culture is stimulating the production of cRET, preventing its decline. In turn, this enhanced production in cRET would stimulate ureteric branching morphogenesis through the cRET signalling pathway. Taken together, the reduction in nephron number due to ethanol exposure identified previously8 and in this current study, could be a product of the enzymes involved in ethanol metabolism and RA synthesis being preferentially used for the metabolism of ethanol in place of the synthesis of RA. This in turn, would result in diminished levels of RA, causing a lack of signalling of ureteric branching through the cRET GDNF signalling pathway, leading to a reduction in the number of ureteric branch points and subsequent glomerular development (Figure 4).
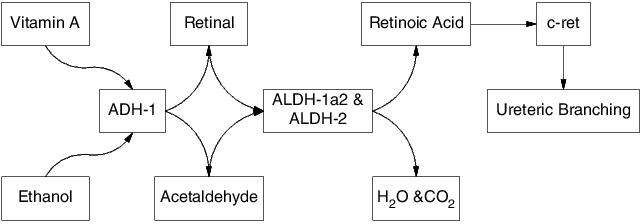
Figure 4. Schematic representation of the enzymatic pathway through which ethanol is metabolised and retinoic acid is synthesised demonstrating the similar enzymes that both pathways employ. Retinoic acid signals ureteric branching through the c-ret receptor, such that if ADH-1, ALDH-1a2 and ALDH-2 are preferentially metabolising ethanol in place of synthesising retinoic acid, there would be diminished retinoic acid production. This in turn would reduce the signals through the c-ret receptor and subsequently limit ureteric branching morphogenesis, leading to a reduction in glomerular development.
While in this study it has been demonstrated that the RA synthesis pathway contributes to the effects of ethanol on kidney development, oxidative stress has also previously been implicated as a potential mechanism.35,36 Consumption of alcohol is associated with an increase in the abundance of oxygen free radicals, resulting in increased oxidative stress within cells. Normally this enhanced state of oxidative stress is eliminated through the actions of antioxidants. However, the actions of the antioxidants are low in the developing fetus, limiting the removal of the enhanced oxidative stress caused by alcohol.37 Furthermore, it has been proposed that the increased oxidative stress within the developing fetus causes DNA damage in tissue that is undergoing rapid proliferation, such as the brain and liver, leading to apoptosis. This enhanced apoptosis would significantly reduce the number of cells available within the developing tissue, resulting in a reduced organ size. In the current study, the apoptotic markers BAX and Bcl-2 were analysed to identify if enhanced apoptosis within ethanol-exposed kidneys was contributing to the reduced ureteric branching morphogenesis and glomerular number. Relative expression of BAX and Bcl-2 were similar in the four treatment groups, indicating that the slowing of ureteric branching morphogenesis was unlikely to be a direct product of enhanced apoptosis.
Taken together, this study has demonstrated that the effects of ethanol in vitro on kidney development resulting in a significant reduction in ureteric branch points, ureteric tips and glomeruli can be ameliorated by the addition of RA. Furthermore, through the use of RT-PCR it has been identified that the augmentation in ureteric branching morphogenesis in ethanol-exposed kidneys co-cultured with RA is associated with altered expression of cRET. In addition, expression levels of genes common to both the enzymatic pathway of ethanol metabolism and RA synthesis, were altered, and are likely to be involved in the changes identified. These results provide the foundation for further research into the molecular mechanism through which alcohol disrupts, and RA rescues, kidney development.
This work was supported by a National Health & Medical Research Council grant (Grant ID 511162). The authors would like to acknowledge the technical assistance of Associate Prof. Mary Wlodek and Mr Chaminda Premaratne.
1. Abel EL. Prenatal effects of alcohol. Drug Alcohol Depend. 1984; 14: 1-10.
2. Day NL, Richardson GA. Prenatal alcohol exposure: A continuum of effects. Semin. Perinatol. 1991; 15: 271-279.
3. Gray SP, Kenna K, Bertram JF, Hoy WE, Yan EB, Bocking AD, Brien JF, Walker DW, Harding R, Moritz KM. Repeated ethanol exposure during late gestation decreases nephron endowment in fetal sheep. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008; 295: R568-R574.
4. Maier SE, Miller JA, West JR. Prenatal binge-like alcohol exposure in the rat results in region-specific deficits in brain growth. Neurotoxicol. Teratol. 1998; 21: 285-291.
5. Maier SE, West JR. Regional differences in cell loss associated with binge-like alcohol exposure during the first two trimesters equivalent in the rat. Alcohol 2001; 23: 49-57.
6. Young C, Olney JW. Neuroapoptosis in the infant mouse brain triggered by a transient small increase in blood alcohol concentration. Neurobiol. Dis. 2006; 22: 548-554.
7. Wang X, Gomutputra P, Wolgemuth DJ, Baxi L. Effects of acute alcohol intoxication in the second trimester of pregnancy on development of the murine fetal lung. Am. J. Obstet. Gynecol. 2007; 197: 269.e261-269.e264.
8. Gray SP, Denton KM, Cullen-McEwen L, Bertram JF, Moritz KM. Prenatal exposure to alcohol reduces nephron number and raises blood pressure in progeny. J. Am. Soc. Nephrol. 2010; 21: 1891-1902.
9. Brenner BM, Garcia DL, Anderson S. Glomeruli and blood pressure. Less of one, more the other? Am. J. Hypertens. 1988; 1: 335-347.
10. Hoy WE, Hughson MD, Singh GR, Douglas-Denton R, Bertram JF. Reduced nephron number and glomerulomegaly in Australian Aborigines: A group at high risk for renal disease and hypertension. Kidney Int. 2006; 70: 104-110.
11. Gallo PV, Weinberg J. Organ growth and cellular development in ethanol-exposed rats. Alcohol 1986; 3: 261-267.
12. Burrow CR. Regulatory molecules in kidney development. Pediatr. Nephrol. 2000; 14: 240-253.
13. Dressler GR. The cellular basis of kidney development. Annu. Rev. Cell. Dev. Biol. 2006; 22: 509-529.
14. Mendelsohn C, Batourina E, Fung S, Gilbert T, Dodd J. Stromal cells mediate retinoid-dependent functions essential for renal development. Development 1999; 126: 1139-1148.
15. Wilson JG, Roth CB, Warkany J. An analysis of the syndrome of malformations induced by maternal vitamin A deficiency. Effects of restoration of vitamin A at various times during gestation. Am. J. Anat. 1953; 92: 189-217,
16. Crabb DW, Pinairs J, Hasanadka R, Fang M, Leo MA, Lieber CS, Tsukamoto H, Motomura K, Miyahara T, Ohata M, Bosron W, Sanghani S, Kedishvili N, Shiraishi H, Yokoyama H, Miyagi M, Ishii H, Bergheim I, Menzl I, Parlesak A, Bode C. Alcohol and retinoids. Alcohol. Clin. Exp. Res. 2001; 25: 207S-217S.
17. Deltour L, Ang HL, Duester G. Ethanol inhibition of retinoic acid synthesis as a potential mechanism for fetal alcohol syndrome. FASEB J. 1996; 10: 1050-1057.
18. Vilar J, Gilbert T, Moreau E, Merlet-Bénichou C. Metanephros organogenesis is highly stimulated by vitamin A derivatives in organ culture. Kidney Int. 1996; 49: 1478-1487.
19. Goodyer P, Kurpad A, Rekha S, Muthayya S, Dwarkanath P, Iyengar A, Philip B, Mhaskar A, Benjamin A, Maharaj S, Laforte D, Raju C, Phadke K. Effects of maternal vitamin A status on kidney development: A pilot study. Pediatr. Nephrol. 2007; 22: 209-214.
20. Singh RR, Cullen-McEwen LA, Kett MM, Boon W-M, Dowling J, Bertram JF, Moritz KM. Prenatal corticosterone exposure results in altered AT1/AT2, nephron deficit and hypertension in the rat offspring. J. Physiol. 2007; 579: 503-513.
21. Batourina E, Gim S, Bello N, Shy M, Clagett-Dame M, Srinivas S, Costantini F, Mendelsohn C. Vitamin A controls epithelial/mesenchymal interactions through Ret expression. Nature Gen. 2001; 27: 74-78.
22. Gilbert T. Vitamin A and kidney development. Nephrol. Dial. Transplant. 2002; 17: 78-80.
23. Lelievre-Pegorier M, Vilar J, Ferrier ML, Moreau E, Freund N, Gilbert T, Merlet-Benichou C. Mild vitamin A deficiency leads to inborn nephron deficit in the rat. Kidney Int. 1998; 54: 1455-1462.
24. Humes HD, Cieslinski DA. Interaction between growth factors and retinoic acid in the induction of kidney tubulogenesis in tissue culture. Exp. Cell Res. 1992; 201: 8-15.
25. Weller A, Sorokin L, Illgen EM, Ekblom P. Development and growth of mouse embryonic kidney in organ culture and modulation of development by soluble growth factor. Dev. Biol. 1991; 144: 248-261.
26. Makrakis J, Zimanyi MA, Black MJ. Retinoic acid enhances nephron endowment in rats exposed to maternal protein restriction. Pediatr. Nephrol. 2007; 22: 1861-1867.
27. Sutherland MR, Gubhaju L, Yoder BA, Stahlman MT, Black MJ. The effects of postnatal retinoic acid administration on nephron endowment in the preterm baboon kidney. Pediatr. Res. 2009; 65: 397-402.
28. Ang HL, Deltour L, Zgombic-Knight M, Wagner MA, Duester G. Expression patterns of class I and class IV alcohol dehydrogenase genes in developing epithelia suggest a role for alcohol dehydrogenase in local retinoic acid synthesis. Alcohol Clin. Exp. Res. 1996; 20: 1050-1064.
29. Ruberte E, Dolle P, Chambon P, Morriss-Kay G. Retinoic acid receptors and cellular retinoid binding proteins. II. Their differential pattern of transcription during early morphogenesis in mouse embryos. Development 1991; 111: 45-60.
30. Ruberte E, Dolle P, Krust A, Zelent A, Morriss-Kay G, Chambon P. Specific spatial and temporal distribution of retinoic acid receptor gamma transcripts during mouse embryogenesis. Development 1990; 108: 213-222.
31. Duester G. Genetic dissection of retinoid dehydrogenases. Chem. Biol. Interact. 2001; 130-132: 469-480.
32. Lieber CS. Alcohol: Its metabolism and interaction with nutrients. Annu. Rev. Nutr. 2000; 20: 395-430.
33. Niederreither K, McCaffery P, Dräger UC, Chambon P, Dollé P. Restricted expression and retinoic acid-induced downregulation of the retinaldehyde dehydrogenase type 2 (RALDH-2) gene during mouse development. Mech. Dev. 1997; 62: 67-78.
34. Moreau E, Vilar J, Lelievre-Pegorier M, Merlet-Benichou C, Gilbert T. Regulation of c-ret expression by retinoic acid in rat metanephros: implication in nephron mass control. Am. J. Physiol. 1998; 275: F938-F945.
35. Cano MJ, Ayala A, Murillo ML, Carreras O. Protective effect of folic acid against oxidative stress produced in 21-day postpartum rats by maternal-ethanol chronic consumption during pregnancy and lactation period. Free Radic. Res. 2001; 34: 1-8.
36. Mitchell JJ, Paiva M, Moore DB, Walker DW, Heaton MB. A comparative study of ethanol, hypoglycemia, hypoxia and neurotrophic factor interactions with fetal rat hippocampal neurons: A multi-factor in vitro model for developmental ethanol effects. Dev. Brain Res. 1998; 105: 241-250.
37. Addolorato G, Gasbarrini A, Marcoccia S, Simoncini M, Baccarini P, Vagni G, Grieco A, Sbriccoli A, Granato A, Stefanini GF, Gasbarrini G. Prenatal exposure to ethanol in rats: Effects on liver energy level and antioxidant status in mothers, fetuses, and newborns. Alcohol 1997; 14: 569-573.