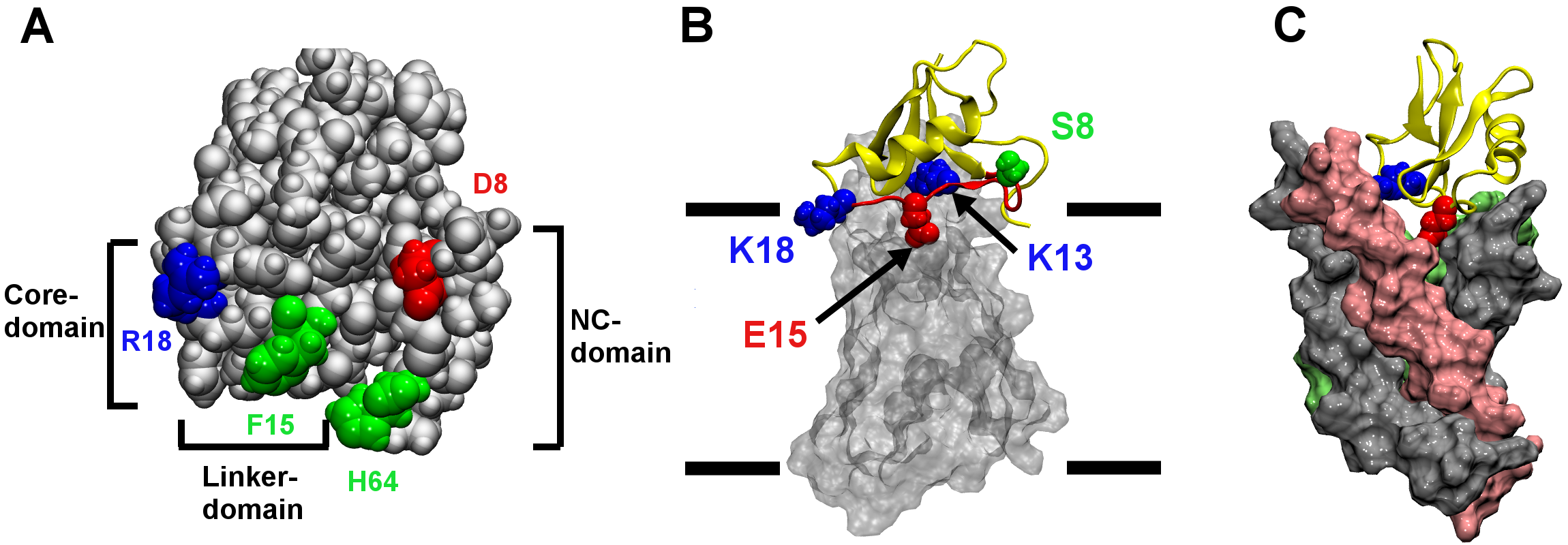
Sodium channels open in response to membrane depolarization and initiate action potentials. Two classes of polypeptide toxins extracted from scorpion venoms, known as α- and β-toxins, interfere with the gating mechanisms of sodium channels by binding to and hindering the movement of the voltage-sensing domains of the channels. Numerous experiments have demonstrated that α-toxins inhibit fast inactivation, whereas β-toxins cause channels to open at less depolarized voltages. The excellent selectivity of the toxins for different subtypes of mammalian and insect sodium channels has been demonstrated, rendering these toxins as potential scaffolds for novel drugs and insecticides. Using a combination of several computational techniques, including homology modeling, molecular docking and molecular dynamics simulations, we have examined in atomic detail the binding of two α-toxins and two β-toxins to their corresponding receptor sites (Chen & Chung, 2012a,b). The binding modes predicted with the computational methods have been found to be in good agreement with mutagenesis experiment. We demonstrated the pivotal role of the loop between positions 8 and 18, which we refer to as the Linker-domain, in toxin binding (Figure 1). Knowledge on the binding modes of the toxins we uncovered may prove useful in designing more selective and effective activators of sodium channels.
R. Chen, S.H. Chung. (2012a) Conserved functional surface of anti-mammalian scorpion β-toxins. Journal of Physical Chemistry B 116: 4796-4800.
R. Chen, S.H. Chung. (2012b) Binding modes and functional surface of anti-mammalian scorpion α-toxins to sodium channels. Biochemistry DOI:10.1021/bi300776g.
