Duchenne muscular dystrophy (DMD) affects approximately 1 in every 3,500 live male births making DMD the most common and lethal genetic disease experienced by humans. Those afflicted experience progressive muscle wasting and subsequent weakness with age resulting in a loss of functional mobility and ultimately a decrease in lifespan. Taurine supplementation has been found to increase force production of skeletal muscle in adult mdx mice, although in adulthood mdx mice undergo a natural reduction in the pathology that may contribute to this improvement. No studies have assessed taurine supplementation during the period of peak damage (21-28 days of age) in mdx mice. Investigating taurine supplementation during this peak damage period would more accurately reflect the potential for taurine as a therapeutic treatment for DMD.
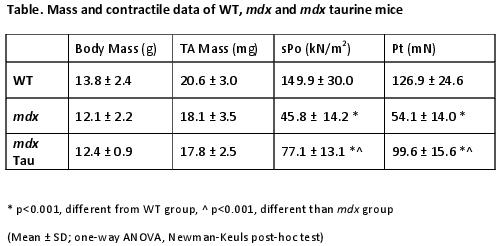
Offspring from C57BL/10ScSn (WT), mdx and mdx taurine (Tau) supplemented mice (n=10, 13, 8 respectively) were obtained and utilized for experimentation at 28 (±1) days of age. Supplemented mdx offspring received taurine (dose 2.5%), in drinking water for 28 days. At 28 days of age, mice were anaesthetised with Nembutal (6mg/ml) in accordance with the La Trobe University Animal Ethics Committee. Tibialis anterior (TA) muscles were surgically exposed, and attached to an in situ muscle apparatus to measure force characteristics. Mice were euthanized by Nembutal overdose post experimentation. The Table summarises the findings. Specifically, there was no difference in either body or TA mass between all treatment types (P > 0.05, 1-way ANOVA). Peak twitch (Pt) and specific force (sPo) were significantly lower in both mdx groups compared to WT control (P<0.001), however, both Pt and sPo were significantly higher in the mdx Tau groups compared to mdx untreated (P<0.001). In general TA twitch characteristics, optimum length (Lo), time to peak (TPT) and half-relaxation time (1/2RT), were not different between groups (all P>0.05). Visualisation of haematoxylin and eosin (H&E) staining of the contracted TA of each mouse treatment (n=4) identified extensive damage in mdx mice, as seen by centralized nuclei and highly variant fibre sizes, that were less prominent in the taurine treated group which more closely emulated the WT.
These findings suggest that taurine supplementation acts to increase muscular force production and improves muscle architecture in the peak damage 28 day mdx mice. This is an exciting prospect for the future of DMD treatment.
