1. Preterm newborns, particularly very low birth weight newborns, frequently experience intermittent hypotension and/or hypoperfusion.
2. Organ perfusion is largely distinct from systemic hypotension, suggesting that changes in underlying vascular tone are the major determinants of perfusion.
3. Preterm fetuses have a remarkable anaerobic tolerance and ability to survive major insults with no or limited injury, balanced by relative immaturity of key autonomic responses.
4. Exposure to hypoxia-ischaemia and infection trigger complex changes in vascular tone that evolve over many days, and there is evidence that these are centrally controlled, and in part linked with underlying organ metabolism.
5. Hypoperfusion frequently occurs after hypoxia-ischaemia without organ injury occurring.
6. Hypoxia-ischaemia, infection and many clinical interventions such as steroid therapy and ventilation can interact to increase or decrease the risk of brain injury.
Globally, preterm birth rates are rising, reaching 13% of live births in the USA.1 Overall, preterm infants are 70% more likely to die and 75% more likely to suffer illness and injury than term infants.2 Understandably, most complications occur in the smallest, most premature infants,3 but recent studies show that the risks of injury and disability are increased seven fold even in “late preterm” infants. Since delivery is much more common at later gestations and mortality is low, this group may contribute as much disability in absolute numbers as more preterm births.4,5 The high level of neurodevelopmental disability associated with prematurity poses a considerable burden on individuals and families, and on healthcare and education resources.2 Thus, the importance of finding ways to improve the outcomes of preterm infants cannot be overstated, for the individuals, their families and the wider community.
Improving the outcome of premature birth will require both the development of new interventions, and more detailed understanding of the complications associated with preterm birth. Cardiovascular instability in early neonatal period in preterm infants is highly associated with adverse outcomes.6 At present, even in intensive care, there is relatively limited physiological monitoring of babies, considerable variance in the treatment of cardiovascular instability between clinicians and institutions, and unresolved uncertainty over optimal treatment strategies, all of which ultimately contribute to failure to effectively improve outcomes. To further improve the outcomes after premature birth we need a greater understanding of the multifactorial pathogenic mechanisms underlying injury and illness. This includes developing a much fuller appreciation of how adverse events and clinical interventions before birth affect the cardiovascular adaptation of the preterm newborn to life.
Hypotension and hypoperfusion at birth, particularly during the first days of life, and notably in lower birth weight preterm infants, are widely believed to initiate or exacerbate injury.6,7 Most current management is based on the apparently obvious concept that poor blood flow must be related to inadequate perfusion pressure. In practice, recent studies suggest that for most infants, particularly in the first few days of life, changes in vascular resistance rather than blood pressure are the primary determinant of blood flow.8,9 Pathologically low systemic blood flow occurs in one third of infants born before 30 weeks gestation.10 In 80% of cases it was lowest at 5 to 12 hours of age, and progressively improved with time; less than 5% of infants had low flows by 48 hours.11 Although some preterm infants did appear to be in ‘compensated shock’, with hypotension and global hypoperfusion, in the majority hypoperfusion was associated with normal blood pressure.6,7,9 It is possible that the definition of ‘normal blood pressure’ might not be appropriate. Nevertheless, there is increasing evidence that blood pressure support with volume or inotropic agents does not generally improve either total blood flow or neonatal outcomes.8,12
In the first 24 hours of life, superior vena cava (SVC) blood flow, a measure of perfusion of the upper body, is a better prognostic marker for mortality and adverse neurodevelopmental outcome than blood pressure.13 Similarly, the recent ELGANS study showed that mean blood pressure and labile blood pressure in the first 24 hours did not correlate with neurological outcomes at 24 months.12 These findings are highly consistent with the poor correlation between mean arterial blood pressure and systemic perfusion as measured by SVC flow or with left ventricular output.8
Cardiovascular instability is undoubtedly multifactorial.6 The key elements that need to be considered include the baby’s stage of maturation, adaptation to neonatal life, and the potential for exposure to injury, and to clinical interventions such as maternal corticosteroids, to trigger evolving changes. Likely because of these factors, the newborn’s cardiovascular state changes rapidly over the first few days.9 As previously reviewed, factors such as poor myocardial function secondary to immature cardiac structure and patent ductus arteriosus (PDA), with arterial duct “steal”, may contribute to relatively poor systemic perfusion.14 However, the extent to which conditions such as PDA contribute to injury remains highly debated.15 The focus of the present review is the how maturation of fetal reflex responses, and fetal adaptation to adverse events such as asphyxia and infection, may shape the adaptation of the premature infant to life after birth.
In adults, the arterial baroreflex is critical for short-term maintenance of blood pressure around a set mean. The main efferent pathways of the baroreflex are the branches of the autonomic nervous system, which alter heart rate (HR) through sympathetic and parasympathetic nervous system activity, and peripheral vascular resistance through the sympathetic nerves. Potentially, reduced baroreflex sensitivity in preterm infants could increase short-term cardiovascular instability and thus increase risk of neural injury. Supporting this hypothesis, in preterm (0.7 gestation) fetal sheep, the cardiac baroreflex is strikingly asymmetrical, with a significantly slower increase in HR in response to a fall in blood pressure than the reciprocal fall in HR following a rapid rise in pressure.16 Further, although background renal sympathetic nerve activity (RSNA) was present even in these very immature fetal lambs, it was not under baroreflex control despite rapid changes in arterial blood pressure, strongly inferring that vasomotor responses to hypotension are immature.
In contrast to preterm fetuses, near-term fetal sheep have relatively mature baroreflexes as shown by effective suppression of RSNA during hypertension.17 Nevertheless, some aspects remain immature near-term, as although HR increased during low voltage sleep (active or rapid eye movement sleep), there were no changes during high voltage sleep (quiet or non-rapid eye movement sleep), and there were no significant changes in RSNA during fetal hypotension in either sleep state.17 These findings support human data showing that baroreflex sensitivity was lower in preterm neonates.18 Thus, the ‘late’ preterm newborn appears to have an impaired ability to respond rapidly to periods of hypotension, for example during fluid imbalance and systemic complications after preterm birth.19
Because the fetus relies entirely on the uteroplacental exchange to obtain oxygen, the risk of hypoxia to the fetus is high. Indeed labour is consistently associated with recurrent fetal hypoxia.20 Whereas the baroreflex is triggered mainly by short-term changes in pressure, the chemoreflex is the primary early response to an acute fall in oxygen tension. Preterm fetal sheep at 0.6 or 0.7 gestation show very different and apparently blunted responses to moderate inhalational hypoxia, haemorrhagic hypotension, and partial umbilical cord occlusion compared with term.21-25 These dramatic differences are largely related to the far greater anaerobic reserves and ability of the near-midgestation fetus to survive severe asphyxia without neural injury compared with term.26 When preterm fetuses are exposed to a more profound challenge, such as asphyxia induced by complete occlusion of the umbilical cord, they show a robust initial chemoreflex-mediated bradycardia and peripheral vasoconstriction that are highly similar to the responses at term.27,28
There is evidence of subtle maturation of the response to such supramaximal insults over the last third of gestation in the sheep. For example, the rate of femoral vasoconstriction at the start of severe asphyxia is significantly slower at 0.6 than at 0.85 of gestation.28 Consistent with this, pharmacological blockade studies suggest that resting sympathetic nerve activity (SNA) is much lower in the preterm fetus than at term.29,30 Direct recordings of RSNA confirm that the preterm sheep fetus has bursts of RSNA that are coordinated with the cardiac cycle, but at a much lower frequency than in adults.31 We speculate that this relative immaturity is part of an overall reduced sensitivity to homeostatic challenges, such as severe hypoxic stress, during or after birth. However, there are few direct data, and further research is essential to determine whether this is a substantial problem, or is compensated for by a much greater neural tolerance to asphyxial injury in the preterm brain.32
Just as the fetus transitions to become a newborn, the responses triggered by exposure to hypoxia or infection before birth can still evolve after birth. Thus, it is important to consider whether injury before birth might be exacerbated by ongoing illness or treatments after birth (the ‘multiple hits’ hypothesis), or be confounded with neonatal complications. Perinatal hypoxia is more common in infants born prematurely than at term,33 and frequently occurs before the onset of labour.2,33 Consistent with this, there is evidence that neural injury occurs in two thirds of infants before birth and or in the immediate neonatal period.34
In the preterm fetal sheep, severe asphyxia is associated with delayed (secondary) onset of vasoconstriction in both central and peripheral beds that lasts for many days.35 This may, at least in part, help support impaired myocardial function.19 Similarly, vasoconstriction after perinatal hypoxia has been postulated to promote redistribution of cardiac output to vital organs.6 The mechanisms of this delayed hypoperfusion remain controversial. Post-natally, there is evidence that changes in endothelial function after ischaemia may play a role in mediating delayed hypoperfusion.36 For example, cytokine and chemokine release in response to inflammation can facilitate release of adhesion molecules, leading to impaired blood flow.36 However, numerous studies now strongly suggest that secondary cerebral hypoperfusion, particularly during the early recovery phase, actually reflects reduced neural metabolism.37 In older fetuses and newborn lambs after hypoxia, although oxygen delivery to the brain was reduced during post-hypoxic secondary hypoperfusion,38 there was no significant change in arterio-venous oxygen extraction.38,39 Further, following severe hypoxia in near-term fetal sheep, delayed post-hypoxic cerebral hypoperfusion was associated with suppression of cerebral metabolism and increased cortical tissue oxygenation, again consistent with the hypothesis that the reduced level of perfusion is appropriate for demand.37
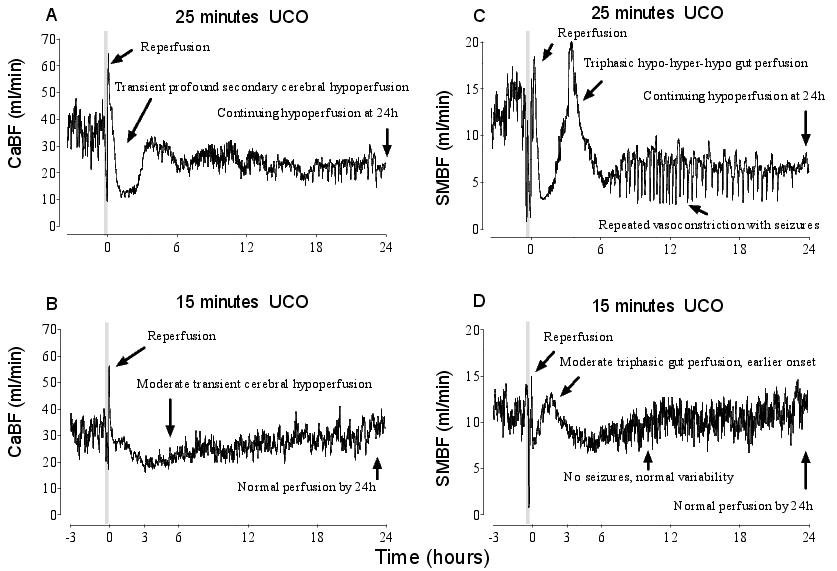
In adults, the duration and speed of onset of cerebral hypoperfusion, and to a lesser extent its degree, are broadly related to the severity of the insult.40,41 Consistent with this, in preterm fetal sheep the patterns of hypoperfusion are related to the duration of severe asphyxia (Figure 1). Further, there may be secondary episodes of superimposed vasoconstriction during seizures (Figure 1, panel C). For example, we observed that the gut is particularly susceptible to seizure mediated flow changes and hypoperfusion was less marked after a shorter insult, with faster resolution of hypoperfusion. These data, and those shown in Figure 2, highlight both marked differences in the patterns of blood flow between the organ beds after asphyxia, and the dynamic changes over time, particularly in the first hours of recovery. These changes occur despite normal or elevated blood pressure (Figure 3).

Figure 1. Carotid blood flow (CaBF) and superior mesenteric artery blood flow (SMBF) from preterm fetal sheep. The data are at 0.7 gestation after 15 minutes (panels A and C) or 25 minutes (panels B and D) of asphyxia induced by complete umbilical cord occlusion (UCO). 15 minutes of asphyxia caused either no or trivial brain injury, in contrast with severe subcortical and white matter injury after 25 min of occlusion. Neither insult caused gut or kidney injury. These data demonstrate that hypoperfusion is observed in central and peripheral beds regardless of morbidity, and that the patterns differ as a function of the duration of the insult (and thus severity) and the specific vascular bed. Data are one minute averages from individual fetuses. The end of the occlusion period is time zero.
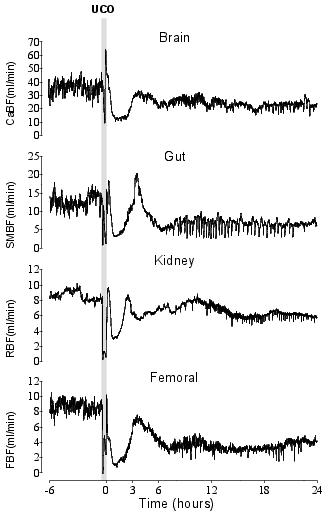
Figure 2. Carotid blood flow (CaBF), superior mesenteric artery blood flow (SMBF), renal blood flow (RBF) and femoral blood flow (FBF) responses to 25 minutes of umbilical cord occlusion. These demonstrate the temporal changes in different vascular beds during the first 24 hours of recovery from asphyxia. Data are one minute averages from a single fetus. The end of the occlusion period is time zero, occlusion period denoted by grey shaded box.
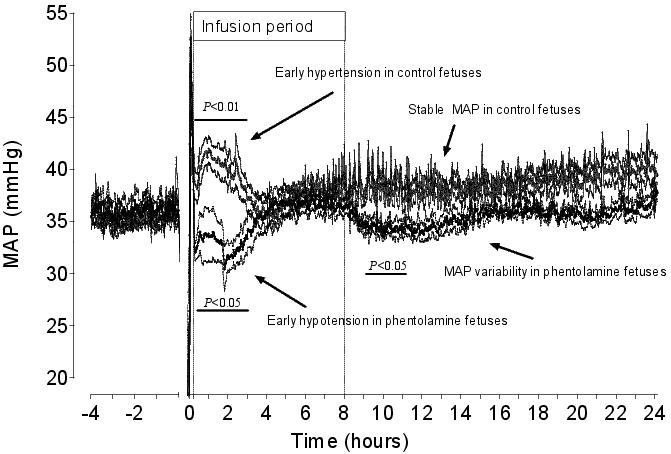
Figure 3. Mean arterial pressure in vehicle control fetuses and phentolamine fetuses. Data are one minute mean±SEM from four hours before asphyxia induced by 25 minutes of umbilical cord occlusion (UCO, occlusion period not shown) until 24 hours after UCO. Saline or the α-adrenergic antagonist phentolamine was infused intravenously to the fetus for 8 hours starting 15 minutes after the end of UCO. In the control group, there is an initial moderate but significant increase in blood pressure, which resolves to baseline values around 3 hours, remaining stable thereafter. In contrast, while phentolamine restored gut blood flow (see Figure 4) it caused hypotension during the first 3 hours, with variability in pressure observed thereafter.
There is increasing evidence that the sympathetic nervous system plays a central role in mediating post-asphyxial changes in central and peripheral blood flow and contributes to neuroinhibition.42,43 We previously reported that post-asphyxial hypoperfusion of the preterm fetal gut,44 was prevented by infusion of the mixed α-adrenergic antagonist phentolamine.42 In control fetuses hypoperfusion was not associated with hypotension, regardless of the vascular bed being measured (Figure 3). Rather, there was a brief period of mild hypertension during the first 3 hours, followed by stabilization of blood pressure around baseline values (Figure 3). By contrast, hypotension occurred when gut hypoperfusion was prevented by phentolamine, followed by variable pressure fluctuations. Notably, the period of hypotension was most pronounced during the corresponding period of hypertension in control fetuses. These data highlight the need to appreciate the temporal nature of perfusion and blood pressure changes, and the close relationships with the cessation of an adverse event like hypoxia. Interestingly, there was no increase in HR (the primary determinant of combined ventricular output in the fetus) to compensate for this fall in blood pressure.42 This may reflect the sensitivity of the baroreflex at this age, as discussed above. However, these data also suggest that vasoconstriction is necessary to support blood pressure, particularly in the early hours of recovery from asphyxia.
The heart of the preterm fetal sheep is very resistant to injury.45 Although overt cardiac damage may not develop, reversible myocardial injury and cardiac dysfunction have been reported during recovery from perinatal asphyxia in both the sheep and the human.46,47 In adults exposed to decreased cardiac output resulting from cardiogenic or hypovolaemic shock, selective vasoconstriction of the afferent mesenteric arterioles is reported to be crucial in sustaining total systemic vascular resistance, thereby maintaining systemic arterial pressure.48 Under these conditions, while there is some degree of vasoconstriction in other peripheral systems, it is disproportionately greater in the mesenteric circulation. Thus perfusion of non-mesenteric organs can be maintained at the ‘expense’ of the gut.48 Of interest, in the preterm sheep fetus, the gut and femoral artery beds appear to develop greater vasoconstriction than the renal bed (Figure 2), suggesting that these beds are also more important contributors to increased peripheral resistance in fetuses as well as adults.
In contrast with the data above supporting an active role for central SNS-mediated hypoperfusion, in preliminary studies we found that even very early infusions of the nitric oxide donor L-arginine, or the endothelial antagonist bosentan, after asphyxia in preterm fetuses did not prevent hypoperfusion (Figure 4). These data coupled with the rapidity of onset of hypoperfusion provide further support for the hypothesis that post-asphyxial peripheral vasoconstriction is centrally controlled. There is also evidence that secondary hypoperfusion of the brain is both actively controlled and associated with neuroprotection. For example, phentolamine infusion restored cerebral perfusion to ‘normal’, but was associated with earlier onset of post-asphyxial seizures.42 Noradrenaline is an important endogenous neuroinhibitor, and it is likely that the sympathetic activation after asphyxia contributed to central protective suppression of brain activity.
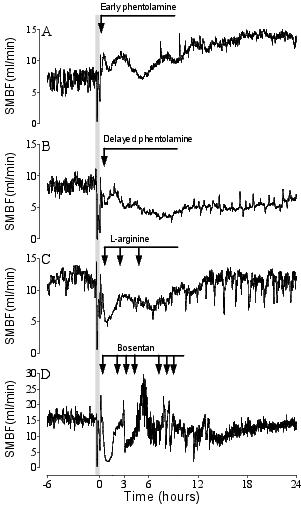
Figure 4. The effects of the α-adrenergic blocker phentolamine. The block was started at 15 minutes (panel A) and 30 minutes (panel B) after the end of asphyxia, L-arginine (panel C) and bosentan (panel D). The first arrow denotes the start of infusions of each agent, and subsequent arrows repeated boluses given to attempt to improve blood flow. Data are one minute averages from individual fetuses. The end of the occlusion period is time zero.
Consistent with this, selective α2-adrenergic receptor blockade was associated with increased epileptiform transient activity in the latent (early recovery phase) and increased brain injury after 3 days recovery.43 Conversely, exogenous infusion of an α2-adrenergic receptor agonist was associated with improved outcomes.49 It is likely that epileptiform transients are a manifestation of post-hypoxic hyperexcitability of the glutamate receptor, as occurs in immature rodents.50 In adult rodents, peri-infarct depolarising waves (spreading depression waves) develop after cerebral ischaemia, and can contribute to expansion of injury by increasing the work load of stressed cells.51 This increasing energy imbalance, in turn, hastens the energy failure in sick cells, promoting impaired cellular homeostasis, ATP production, and initiation of cell death processes (secondary energy failure).
Collectively these data further support the concept that reduced blood flow is coupled to reduced metabolism during recovery from hypoxic-ischemic insults and that this is beneficial for recovery from asphyxia. These data are consistent with the lack of efficacy of many clinical interventions such as a volume expansion, administration of inotropes, and closure of the ductus arteriosus after birth.13,52 Thus, despite the differences between fetal and postnatal life, there is a considerable need for further research to determine whether hypoperfusion is a cause of morbidity or merely a consequence.15
There is now considerable evidence that perinatal exposure to infection and inflammation such as clinical chorioamnionitis (uterine infection), vasculitis (inflammation of blood vessels) in the chorionic plate of the placenta and/or umbilical cord, and high levels of pro-inflammatory cytokines in amniotic and umbilical blood, are associated with increased mortality and morbidity.53 Some studies have found no association between histological chorioamnionitis and risk of white matter injury, whereas neonatal sepsis has been consistently associated with increased risk of brain injury.54,55
There is relatively limited information on the haemodynamic effects of sepsis in preterm newborns,56 and at present there are no agreed treatment guidelines for the management of severe sepsis and septic shock for preterm neonates. This is complicated by the lack of normative cardiovascular data for preterm infants.57 It is also clear that the responses of the newborn are very variable between infants, dynamic over time, and may differ to that of the adult in response to treatment.56,57 In this setting, the immediate effect of early onset neonatal sepsis appears to be mainly vasodilation, leading to hyperperfusion and often hypotension.54,55 Other studies have reported hypotension with peripheral vasoconstriction and increased cardiac output, or “normal” blood pressure with either vasodilatation or vasoconstriction or hypotension with either state.56,57 This may reflect a continuum from chronic low key inflammation, secondary to chorioamnionitis, through to acute severe infection.57
Nevertheless, persistent vasodilatation is often observed after exposure to inflammation in adult humans and animal models.58 Indeed, much of the acute morbidity associated with significant infection or inflammation in adults is associated with endothelial dysfunction, with altered release and sensitivity to vasodilators and vasoconstrictors, which in turn leads to haemodynamic instability and organ compromise.58 In fetal experimental studies, exposure to gram negative lipopolysaccharide (LPS) has been associated with variable haemodynamic effects, likely reflecting differences in dose and timing of exposure between studies. Some studies observed no change in blood pressure or blood flow in fetal sheep,59-61 whereas others reported acute falls in blood pressure,62-65 or a transient fall, followed by increased arterial blood pressure by 48 to 72 hours of recovery.63
Typically, early fetal hypotension in responses to LPS has been associated with cerebral vasodilatation that maintained perfusion and oxygen delivery.65,66 By contrast, a study in the late gestation sheep fetus found acute transient cerebral vasoconstriction after injection of LPS, followed by vasodilatation up to 24 hours after three repeated injections.66 Vasodilatation was associated with evidence of transiently increased nitric oxide (NO) production,66 consistent with endotoxic shock in adults.67 Similar findings were observed when LPS was given to term newborn lambs, with chronic loss of sensitivity to the vasodilator bradykinin, possibly secondary to LPS-induced loss of endothelial cells.68
In 0.7 gestation preterm fetal sheep, exposure to killed gram positive bacteria (OK-432 or Picabinil), derived from low virulence heat killed Su-strain of type 3 Group A streptococcus pyogenes,69,70 was associated with acute, transient peripheral and central vasoconstriction without hypotension.69 This phase was coupled with reduced brain activity and suppression of fetal body and breathing movements.70 These data support the hypothesis that infection was associated with reduced brain metabolism and an appropriate reduction of blood flow, rather than loss of regulatory control.
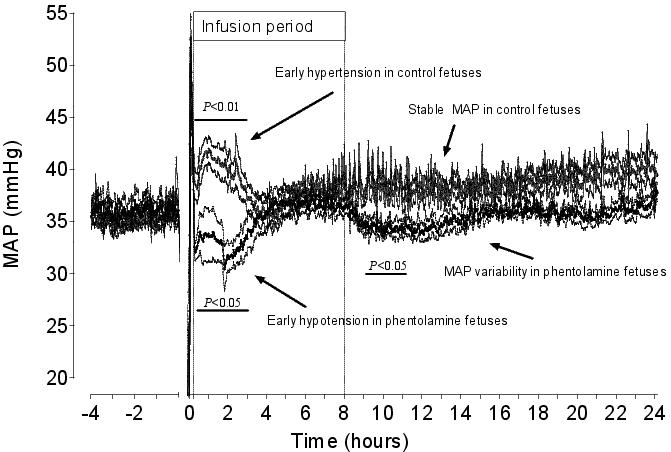
Figure 5. Time course changes in mean arterial blood pressure (MAP) and femoral blood flow (FBF) Data are from 0.7 gestation fetal sheep who received a bolus intrapleural injection of either saline (open circles) or OK-432 (0.1 mg, closed circles). The arrow denotes the point of injection. Data are mean±SEM. Note the failure of blood pressure to increase over time in the OK-432 group compared to the vehicle control group.
Over the week after exposure to OK-432, there was a progressive central and peripheral vasodilatation with increased blood flow (Figure 5).69 Although hypotension did not occur, there was loss of the normal secular increase in arterial blood pressure over this time (Figure 5). Fetal growth was not affected, suggesting that this relatively modest vasodilatation reflects a resetting of the baroreceptor threshold.16 In contrast with previous studies of LPS,66 there were no significant changes in circulating nitrite levels, suggesting that these changes were not related to altered NO production.69 However, it remains possible that there may have been altered responsiveness of the vasculature to vasodilator stimuli, or increased induction of other vasodilators. Alternatively, there are strong data to suggest that inflammatory mediators can alter release of, or sensitivity to, vasoconstrictors without changes in vasodilator activity.71,72
Notably, although OK-432 was associated with acute and chronic changes in cerebral blood flow in all fetuses, only one fetus sustained neural injury; a bilateral infarct of the hippocampus. This injury occurred despite normal blood gases and a time course of blood flow and blood pressure changes which was similar to those of the other fetuses. The early development of seizures suggests that injury occurred shortly after exposure. Thus it is unlikely that injury was directly related to changes in perfusion. This further supports previous data that neural damage associated with infection is primarily associated with neurotoxicity, likely mediated by the cytokine/microglial response.73 The fetal hemodynamic changes after exposure to gram positive bacteria, while persistent, were relatively modest, and it remains unclear if they represent a permanent change in the trajectory of blood pressure, or persist into neonatal life. Nonetheless, it is reasonable to speculate that changes in autonomic or endothelial function might compromise the ability of the fetus and newborn to adapt to further events such as hypoxia.
Profound hypoxia or septic shock causing acute injury around the time of birth are only seen in a minority of infants, and are insufficient to account for the majority of long-term neurodevelopmental disability in preterm infants. However, infants may frequently be exposed to both insults. Intriguingly, experimental data in rodents suggest that exposure to mild infection or inflammation can sensitise the brain, so that short or milder periods of hypoxia-ischaemia, which do not normally injure the developing brain, can trigger severe damage.73 However, the effect is complex and time dependent. A low dose of LPS given either shortly (four or six hours) or well before (72 hours or more) hypoxia in rat pups was associated with increased injury (‘sensitisation’).73 In mice, fetal exposure to LPS affected the responses to hypoxia even in adulthood, with both reduced and increased injury, in different regions.74 In contrast, when given at an intermediate time (24 hours) before hypoxia/ischaemia, LPS actually reduced injury (‘tolerance’).73
No large animal studies have been undertaken to evaluate key mechanisms or to confirm whether these relationships hold up in more complex species. Nevertheless, recent data suggest that prenatal exposure to inflammation exacerbates ventilation-mediated brain injury, supporting the concept that multiple insults can contribute in complex ways to perinatal brain injury.75 Intriguingly, there is now evidence that exposure of preterm fetal sheep to a clinical course of maternal glucocorticoids is associated with acute changes in EEG transient activity and epileptiform events, followed by evidence of neural maturation.76 Given that seizures in early life in the rat can induce neuroprotection through preconditioning mechanisms,77 it is plausible that steroids may also trigger a sensitization/preconditioning sequence independently of pathological events.
The preterm fetus exhibits a remarkable anaerobic tolerance and ability to survive major insults with no or limited injury.32 At the same time, it is clear that autonomic function is still developing over late gestation, and that responses to acute arterial hypotension are particularly immature. Premature newborns are commonly exposed to perinatal hypoxia and infection around the time of birth, which trigger long-lasting changes in central and peripheral tone, which in combination can sensitise the brain to injury. Finally, there is increasing evidence that ‘standard’ clinical interventions such as antenatal maternal steroids and neonatal ventilation can further modulate the risk of damage. The authors believe that to disentangle these multiple factors and provide a solid evidence-base to test innovative paradigms and improve long-term outcomes, we need to develop an approach of comprehensive monitoring and neurointensive care.
The authors’ studies in the present review were supported by grants from the Health Research Council of New Zealand, USPHS grant RO1 HD-32752, March of Dimes, the Auckland Medical Research Foundation, and the Lottery Grants Board of New Zealand. Mr P. Drury was supported by the Neurological Foundation.
1. Allen MC, Cristofalo EA, Kim C. Outcomes of preterm infants: morbidity replaces mortality. Clin. Perinatol. 2011; 38:441-54.
2. Committee on understanding premature birth and assuring healthy outcomes. In: Behrman RE, Butler AS, (eds.). Preterm Birth: Causes, Consequences, and Prevention. Institute of Medicine of the National Academies: Washington DC, USA. 2007.
3. Lemons JA, Bauer CR, Oh W, Korones SB, Papile LA, Stoll BJ, Verter J, Temprosa M, Wright LL, Ehrenkranz RA, Fanaroff AA, Stark A, Carlo W, Tyson JE, Donovan EF, Shankaran S, Stevenson DK. Very low birth weight outcomes of the National Institute of Child health and human development neonatal research network, January 1995 through December 1996. NICHD Neonatal Research Network. Pediatrics 2001; 107:E1.
4. Petrou S, Eddama O, Mangham L. A structured review of the recent literature on the economic consequences of preterm birth. Arch. Dis. Child. Fetal Neonatal Ed. 2011; 96:F225-32.
5. MacKay DF, Smith GC, Dobbie R, Pell JP. Gestational age at delivery and special educational need: retrospective cohort study of 407,503 schoolchildren. PLoS Med. 2010; 7:e1000289.
6. Soleymani S, Borzage M, Seri I. Hemodynamic monitoring in neonates: advances and challenges. J. Perinatol. 2010; 30 Suppl:S38-45.
7. Dempsey EM, Barrington KJ. Evaluation and treatment of hypotension in the preterm infant. Clin. Perinatol. 2009; 36:75-85.
8. Kluckow M. Functional echocardiography in assessment of the cardiovascular system in asphyxiated neonates. J. Pediatr. 2011; 158:e13-8.
9. Groves AM, Kuschel CA, Knight DB, Skinner JR. Relationship between blood pressure and blood flow in newborn preterm infants. Arch. Dis. Child. Fetal Neonatal Ed. 2008; 93:F29-32.
10. Osborn DA, Evans N, Kluckow M. Hemodynamic and antecedent risk factors of early and late periventricular/intraventricular hemorrhage in premature infants. Pediatrics 2003; 112:33-9.
11. Evans N, Osborn D, Kluckow M. Preterm circulatory support is more complex than just blood pressure. Pediatrics 2005; 115:1114-5.
12. Logan JW, O'Shea TM, Allred EN, Laughon MM, Bose CL, Dammann O, Batton DG, Engelke SC, Leviton A. Early postnatal hypotension and developmental delay at 24 months of age among extremely low gestational age newborns. Arch. Dis. Child. Fetal Neonatal Ed. 2011; 96:F321-8.
13. Hunt RW, Evans N, Rieger I, Kluckow M. Low superior vena cava flow and neurodevelopment at 3 years in very preterm infants. J. Pediatr. 2004; 145:588-92.
14. Capozzi G, Santoro G. Patent ductus arteriosus: patho-physiology, hemodynamic effects and clinical complications. J. Matern. Fetal Neonatal Med. 2011; 24 Suppl 1:15-6.
15. Sehgal A. Haemodynamically unstable preterm infant: an unresolved management conundrum. Eur. J. Pediatr. 2011.
16. Booth LC, Malpas SC, Barrett CJ, Guild SJ, Gunn AJ, Bennet L. Is baroreflex control of sympathetic activity and heart rate active in the preterm fetal sheep? Am. J. Physiol. Regul. Integr. Comp. Physiol. 2009; 296:R603-R9.
17. Booth LC, Gunn AJ, Malpas SC, Barrett CJ, Davidson JO, Guild SJ, Bennet L. Baroreflex control of renal sympathetic nerve activity and heart rate in near-term fetal sheep. Exp. Physiol. 2011; 96:736-44.
18. Andriessen P, Oetomo SB, Peters C, Vermeulen B, Wijn PF, Blanco CE. Baroreceptor reflex sensitivity in human neonates: the effect of postmenstrual age. J. Physiol. 2005; 568:333-41.
19. Bennet L, Booth L, Malpas SC, Quaedackers JS, Jensen E, Dean J, Gunn AJ. Acute systemic complications in the preterm fetus after asphyxia: the role of cardiovascular and blood flow responses. Clin. Exp. Pharmacol. Physiol. 2006; 33:291-9.
20. Westgate JA, Wibbens B, Bennet L, Wassink G, Parer JT, Gunn AJ. The intrapartum deceleration in center stage: a physiological approach to interpretation of fetal heart rate changes in labor. Am. J. Obstet. Gynecol. 2007; 197:e1-e11.236.
21. Boddy K, Dawes GS, Fisher R, Pinter S, Robinson JS. Foetal respiratory movements, electrocortical and cardiovascular responses to hypoxaemia and hypercapnia in sheep. J. Physiol. 1974; 243:599-618.
22. Iwamoto HS, Stucky E, Roman CM. Effect of graded umbilical cord compression in fetal sheep at 0.6-0.7 gestation. Am. J. Physiol. Heart Circ. Physiol. 1991; 261:H1268-H74.
23. Szymonowicz W, Walker AM, Yu VY, Stewart ML, Cannata J, Cussen L. Regional cerebral blood flow after hemorrhagic hypotension in the preterm, near-term, and newborn lamb. Pediatr. Res. 1990; 28:361-6.
24. Gleason CA, Hamm C, Jones MD, Jr. Effect of acute hypoxemia on brain blood flow and oxygen metabolism in immature fetal sheep. Am. J. Physiol. Heart Circ. Physiol. 1990; 258:H1064-H9.
25. Iwamoto HS, Kaufman T, Keil LC, Rudolph AM. Responses to acute hypoxemia in fetal sheep at 0.6-0.7 gestation. Am. J. Physiol. Heart Circ. Physiol. 1989; 256:H613-H20.
26. Shelley HJ. Glycogen reserves and their changes at birth and in anoxia. Br. Med. Bull. 1961; 17:137-43.
27. Bennet L, Rossenrode S, Gunning MI, Gluckman PD, Gunn AJ. The cardiovascular and cerebrovascular responses of the immature fetal sheep to acute umbilical cord occlusion. J. Physiol. 1999; 517:247-57.
28. Wassink G, Bennet L, Booth LC, Jensen EC, Wibbens B, Dean JM, Gunn AJ. The ontogeny of hemodynamic responses to prolonged umbilical cord occlusion in fetal sheep. J. Appl. Physiol. 2007; 103:1311-7.
29. Assali NS, Brinkman CR, Woods JR, Jr, Dandavino A, Nuwayhid B. Development of neurohumoral control of fetal, neonatal, and adult cardiovascular functions. Am. J. Obstet. Gynecol. 1977; 129:748-59.
30. Nuwayhid B, Brinkman CR, Su C, Bevan JA, Assali NS. Development of autonomic control of fetal circulation. Am. J. Physiol. 1975; 228:337-44.
31. Booth LC, Bennet L, Guild SJ, Barrett CJ, May CN, Gunn AJ, Malpas SC. Maturation-related changes in the pattern of renal sympathetic nerve activity from in utero to adulthood. Exp. Physiol. 2011; 96:85-93.
32. Gunn AJ, Quaedackers JS, Guan J, Heineman E, Bennet L. The premature fetus: not as defenseless as we thought, but still paradoxically vulnerable? Dev. Neurosci. 2001; 23:175-9.
33. Low JA. Determining the contribution of asphyxia to brain damage in the neonate. J. Obstet. Gynaecol. Res. 2004; 30:276-86.
34. Dyet LE, Kennea N, Counsell SJ, Maalouf EF, Ajayi-Obe M, Duggan PJ, Harrison M, Allsop JM, Hajnal J, Herlihy AH, Edwards B, Laroche S, Cowan FM, Rutherford MA, Edwards AD. Natural history of brain lesions in extremely preterm infants studied with serial magnetic resonance imaging from birth and neurodevelopmental assessment. Pediatrics 2006; 118:536-48.
35. Bennet L, Dean JM, Gunn AJ. The pathogenesis of preterm brain injury. In: Stevenson DK, Benitz WE, Sunshine P, Druzin ML, (eds.). Fetal and Neonatal Brain Injury: Mechanisms, Management, and the Risks of Practice. 4th edn. Cambridge University Press: Cambridge, UK. 2009.
36. Amantea D, Nappi G, Bernardi G, Bagetta G, Corasaniti MT. Post-ischemic brain damage: pathophysiology and role of inflammatory mediators. FEBS J. 2009; 276:13-26.
37. Jensen EC, Bennet L, Hunter CJ, Power GG, Gunn AJ. Post-hypoxic hypoperfusion is associated with suppression of cerebral metabolism and increased tissue oxygenation in near-term fetal sheep. J. Physiol. 2006; 572:131-9.
38. Rosenberg AA. Regulation of cerebral blood flow after asphyxia in neonatal lambs. Stroke 1988; 19:239-44.
39. Gunn AJ, Gunn TR, de Haan HH, Williams CE, Gluckman PD. Dramatic neuronal rescue with prolonged selective head cooling after ischemia in fetal lambs. J. Clin. Invest. 1997; 99:248-56.
40. Huang J, Kim LJ, Poisik A, Pinsky DJ, Connolly ES, Jr. Titration of postischemic cerebral hypoperfusion by variation of ischemic severity in a murine model of stroke. Neurosurgery 1999; 45:328-33.
41. Karlsson BR, Grogaard B, Gerdin B, Steen PA. The severity of postischemic hypoperfusion increases with duration of cerebral ischemia in rats. Acta Anaesthesiol. Scand. 1994; 38:248-53.
42. Quaedackers JS, Roelfsema V, Heineman E, Gunn AJ, Bennet L. The role of the sympathetic nervous system in post-asphyxial intestinal hypoperfusion in the preterm sheep fetus. J. Physiol. 2004; 557:1033-44.
43. Dean JM, Gunn AJ, Wassink G, George S, Bennet L. Endogenous α2-adrenergic receptor-mediated neuroprotection after severe hypoxia in preterm fetal sheep. Neuroscience 2006; 142:615-28.
44. Bennet L, Quaedackers JS, Gunn AJ, Rossenrode S, Heineman E. The effect of asphyxia on superior mesenteric artery blood flow in the premature sheep fetus. J. Pediatr. Surg. 2000; 35:34-40.
45. Bennet L, Kozuma S, McGarrigle HHG, Hanson MA. Temporal changes in fetal cardiovascular, behavioural, metabolic and endocrine responses to maternally administered dexamethasone in the late gestation fetal sheep. Br. J. Obstet. Gynaecol. 1999; 106:331-9.
46. Gunn AJ, Maxwell L, De Haan HH, Bennet L, Williams CE, Gluckman PD, Gunn TR. Delayed hypotension and subendocardial injury after repeated umbilical cord occlusion in near-term fetal lambs. Am. J. Obstet. Gynecol. 2000; 183:1564-72.
47. Lumbers ER, Gunn AJ, Zhang DY, Wu JJ, Maxwell L, Bennet L. Nonimmune hydrops fetalis and activation of the renin-angiotensin system after asphyxia in preterm fetal sheep. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2001; 280:R1045-R51.
48. Reilly PM, Wilkins KB, Fuh KC, Haglund U, Bulkley GB. The mesenteric hemodynamic response to circulatory shock: an overview. Shock 2001; 15:329-43.
49. Dean JM, George SA, Wassink G, Gunn AJ, Bennet L. Suppression of post hypoxic-ischemic EEG transients with dizocilpine is associated with partial striatal protection in the preterm fetal sheep. Neuropharmacology 2006; 50:491-503.
50. Jensen FE, Wang C, Stafstrom CE, Liu Z, Geary C, Stevens MC. Acute and chronic increases in excitability in rat hippocampal slices after perinatal hypoxia in vivo. J. Neurophysiol. 1998; 79:73-81.
51. Somjen GG. Mechanisms of spreading depression and hypoxic spreading depression-like depolarization. Physiol. Rev. 2001; 81:1065-96.
52. Hunt R, Osborn D. Dopamine for prevention of morbidity and mortality in term newborn infants with suspected perinatal asphyxia. Cochrane Database Syst. Rev. 2002:CD003484.
53. Mwaniki MK, Atieno M, Lawn JE, Newton CR. Long-term neurodevelopmental outcomes after intrauterine and neonatal insults: a systematic review. Lancet 2012; 379:445-52.
54. Basu S, Dewangan S, Shukla RC, Anupurva S, Kumar A. Cerebral blood flow velocity in early-onset neonatal sepsis and its clinical significance. Eur. J. Pediatr. 2012; 171:901-9.
55. Chau V, Poskitt KJ, McFadden DE, Bowen-Roberts T, Synnes A, Brant R, Sargent MA, Soulikias W, Miller SP. Effect of chorioamnionitis on brain development and injury in premature newborns. Ann. Neurol. 2009; 66:155-64.
56. de Waal K, Evans N. Hemodynamics in preterm infants with late-onset sepsis. J. Pediatr. 2010; 156:918-22, 22 e1.
57. Wynn JL, Wong HR. Pathophysiology and treatment of septic shock in neonates. Clin. Perinatol. 2010; 37:439-79.
58. Sessler CN, Shepherd W. New concepts in sepsis. Curr. Opin. Crit. Care 2002; 8:465-72.
59. Eklind S, Mallard C, Leverin AL, Gilland E, Blomgren K, Mattsby-Baltzer I, Hagberg H. Bacterial endotoxin sensitizes the immature brain to hypoxic--ischaemic injury. Eur. J. Neurosci. 2001; 13:1101-6.
60. Duncan JR, Cock ML, Suzuki K, Scheerlinck JP, Harding R, Rees SM. Chronic endotoxin exposure causes brain injury in the ovine fetus in the absence of hypoxemia. J. Soc. Gynecol. Investig. 2006; 13:87-96.
61. Duncan JR, Cock ML, Scheerlinck JP, Westcott KT, McLean C, Harding R, Rees SM. White matter injury after repeated endotoxin exposure in the preterm ovine fetus. Pediatr. Res. 2002; 52:941-9.
62. Dalitz P, Harding R, Rees SM, Cock ML. Prolonged reductions in placental blood flow and cerebral oxygen delivery in preterm fetal sheep exposed to endotoxin: possible factors in white matter injury after acute infection. J. Soc. Gynecol. Investig. 2003; 10:283-90.
63. Garnier Y, Frigiola A, Li Volti G, Florio P, Frulio R, Berger R, Alm S, von Duering MU, Coumans AB, Reis FM, Petraglia F, Hasaart TH, Abella R, Mufeed H, Gazzolo D. Increased maternal/fetal blood S100B levels following systemic endotoxin administration and periventricular white matter injury in preterm fetal sheep. Reprod. Sci. 2009; 16:758-66.
64. Blad S, Welin AK, Kjellmer I, Rosen KG, Mallard C. ECG and heart rate variability changes in preterm and near-term fetal lamb following LPS exposure. Reprod. Sci. 2008; 15:572-83.
65. Peebles DM, Miller S, Newman JP, Scott R, Hanson MA. The effect of systemic administration of lipopolysaccharide on cerebral haemodynamics and oxygenation in the 0.65 gestation ovine fetus in utero. BJOG 2003; 110:735-43.
66. Feng SY, Phillips DJ, Stockx EM, Yu VY, Walker AM. Endotoxin has acute and chronic effects on the cerebral circulation of fetal sheep. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2009; 296:R640-50.
67. Thiemermann C. Nitric oxide and septic shock. Gen. Pharmacol. 1997; 29:159-66.
68. Feng SY, Samarasinghe T, Phillips DJ, Alexiou T, Hollis JH, Yu VY, Walker AM. Acute and chronic effects of endotoxin on cerebral circulation in lambs. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010; 298:R760-6.
69. Bennet L, Cowie RV, Stone PR, Barrett R, Naylor AS, Blood AB, Gunn AJ. The neural and vascular effects of killed Su-Streptococcus pyogenes (OK-432) in preterm fetal sheep. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010; 299:R664-72.
70. Cowie RV, Stone PR, Parry E, Jensen EC, Gunn AJ, Bennet L. Acute behavioral effects of intrapleural OK-432 (Picibanil) administration in preterm fetal sheep. Fetal Diagn. Ther. 2009; 25:304-13.
71. Lange M, Bröking K, Hucklenbruch C, Ertmer C, Van Aken H, Lücke M, Bone HG, Westphal M. Hemodynamic effects of titrated norepinephrine in healthy versus endotoxemic sheep. J. Endotoxin Res. 2007; 13:53-7.
72. Pleiner J, Heere-Ress E, Langenberger H, Sieder AE, Bayerle-Eder M, Mittermayer F, Fuchsjäger-Mayrl G, Böhm J, Jansen B, Wolzt M. Adrenoceptor hyporeactivity is responsible for Escherichia coli endotoxin-induced acute vascular dysfunction in humans. Arterioscler. Thromb. Vasc. Biol. 2002; 22:95-100.
73. Wang X, Rousset CI, Hagberg H, Mallard C. Lipopolysaccharide-induced inflammation and perinatal brain injury. Semin. Fetal Neonatal Med. 2006; 11:343-53.
74. Wang X, Hagberg H, Nie C, Zhu C, Ikeda T, Mallard C. Dual role of intrauterine immune challenge on neonatal and adult brain vulnerability to hypoxia-ischemia. J. Neuropathol. Exp. Neurol. 2007; 66:552-61.
75. Polglase GR, Nitsos I, Baburamani AA, Crossley KJ, Slater MK, Gill AW, Allison BJ, Moss TJ, Pillow JJ, Hooper SB, Kluckow M. Inflammation in utero exacerbates ventilation-induced brain injury in preterm lambs. J. Appl. Physiol. 2012; 112:481-9.
76. Davidson JO, Quaedackers JS, George SA, Gunn AJ, Bennet L. Maternal dexamethasone and EEG hyperactivity in preterm fetal sheep. J. Physiol. 2011; 589:3823–35.
77. Saghyan A, LaTorre GN, Keesey R, Sharma A, Mehta V, Rudenko V, Hallas BH, Rafiuddin A, Goldstein B, Friedman LK. Glutamatergic and morphological alterations associated with early life seizure-induced preconditioning in young rats. Eur. J. Neurosci. 2010; 32:1897-911.