1. There is a reversible decline in the force production by muscles when contracting at or near their maximum capacity. The task dependent nature of fatigue means that the mechanisms of fatigue may differ between different types of contractions. This paper examines how fatigue manifests during whole-body, intermittent-sprint exercise, and discusses the potential muscular and neural mechanisms that underpin this fatigue.
2. Fatigue is defined as a reversible, exercise-induced reduction in maximal power output (e.g. during cycling exercise) or speed (e.g. during running exercise), even though the task can be continued.
3. The small changes in surface EMG, along with a lack of change in voluntary muscle activation (estimated from both percutaneous motor nerve stimulations and trans-cranial magnetic stimulation), indicate that there is little change in neural drive to the muscles following intermittent-sprint exercise. This, along with the observation that the decrease in EMG is much less than that which would be predicted from the decrease in power output, suggests that peripheral mechanisms are the predominant cause of fatigue during intermittent-sprint exercise.
4. At the muscle level, limitations in energy supply, which include phosphocreatine hydrolysis and the degree of reliance on anaerobic glycolysis and oxidative metabolism, and the intramuscular accumulation of metabolic by-products, such as hydrogen ions, emerge as key factors responsible for fatigue.
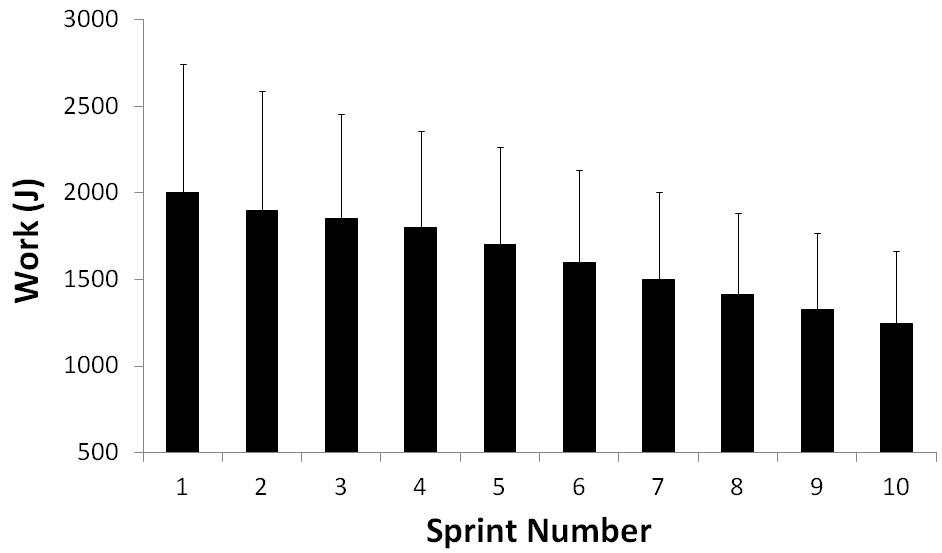
There is a reversible decline in force production by muscles when contracting at or near their maximum capacity. This has been classically demonstrated in isolated muscle cells stimulated to give maximal isometric tetani.1 Such experiments have provided valuable insights regarding potential determinants of fatigue.2 Nonetheless, such conditions are obviously artificial and the application of such findings to dynamic exercise has been questioned. While a similar pattern for the decline in muscle performance is observed when athletes are asked to perform intermittent-sprint exercise (Figure 1),3 the task-dependent nature of fatigue means that the mechanisms of fatigue may differ. In particular, there may be a role for neural/brain factors on the fatigue process during intermittent-sprint exercise (factors obviously omitted from isolated-muscle preparations). This paper examines how fatigue manifests during whole-body, intermittent-sprint exercise, and discusses the potential muscular and neural mechanisms that underpin this fatigue.

Figure 1. Typical mechanical-work profile during intermittent-sprint exercise. The data are for "all-out" sprints, interspersed with brief recovery periods, performed on a cycle ergometer.
There are many disciplines in which athletes are required to repeatedly produce maximal or near maximal efforts (i.e. sprints), interspersed with brief recovery intervals (consisting of complete rest or low- to moderate-intensity activity), over an extended period (1 to 4 hours). While some authors have used the word “sprint” to describe exercise lasting 30s or more; for the purposes of this paper, “sprint” activities will be constrained to brief exercise, in general ≤ 10s.6 Longer-duration, maximal-intensity exercise, in which there is a considerable decrease in performance, will be referred to as “all-out” exercise, but will not be discussed in this paper.
For the purpose of this paper, fatigue is defined as a reversible, exercise-induced, reduction in maximal power output (e.g. during cycling exercise) or speed (e.g. during running exercise), even though the task can be continued. Fatigue during intermittent-sprint exercise typically develops rapidly after the first sprint (Figure 1).7 It is now accepted that exercise-induced fatigue can be caused by a variety of factors, ranging from the generation of an inadequate motor command in the motor cortex (i.e. neural factors) to factors related to metabolite accumulation or energy supply (i.e. muscular factors). The aim of this paper is to discuss evidence for the contribution of these mechansims to fatigue during intermittent-sprint exercise.
An important factor influencing fatigue during intermittent-sprint exercise is the initial (i.e. first sprint) mechanical score, which has been reported to be correlated with the decrement in performance over subsequent sprints.7-9 This is probably because subjects with a greater initial sprint performance will produce greater changes in muscle metabolites, arising secondary to a higher anaerobic contribution, which have been related to larger performance decrements.10 In support of this, individuals with lower anaerobic power reserves, implying less reliance on anaerobic metabolism, show a higher fatigue resistance during intermittent-sprint exercise.7 This suggests that the metabolic pathways supporting force production, and not the absolute force generated per se, might explain power decrements during intermittent-sprint exercise. Therefore, initial sprint mechanical output per se cannot solely account for performance decrements during intermittent-sprint exercise. Indeed, previous fatiguing muscle contractions (i.e. a prior set of intermittent sprints) exacerbate the rate of fatigue during subsequent sprints, despite being matched for initial sprint power.11 Similarly, there is greater fatigability (i.e. larger work decrement) across five 6s cycling sprints repeated every 30s in low versus moderately aerobically-trained females matched for single-sprint performance.12
The mechanisms of fatigue vary with intensity, duration and type of contraction. This task-dependent nature of fatigue is also apparent with different types of intermittent-sprint exercise. For example, the exercise mode (e.g. cycling versus running) has been reported to affect the development of fatigue;13 decrement scores during intermittent-sprint cycling protocols (10-25%) are generally greater than those for running protocols (5-15%). The type of resistive load (e.g. mechanically-, wind- or electromagnetically-resisted) also appears to affect fatigue development during intermittent-sprint exercise. Moreover, fatigue resistance during intermittent-sprint exercise depends on the distribution (e.g. number of repetitions) and duration of the work periods,16 and the recovery pattern; i.e. the nature,17-19 duration,20-23 and intensity24 of the recovery between sprints. While there is some conflict within the literature,25 performing active versus passive recovery is generally associated with a higher degree of fatigue development. Compared with a passive recovery, low- and moderate-intensity active recoveries (∼20 and 35% maximal oxygen uptake (VO2max), respectively) have similar effects on intermittent-sprint exercise performance and muscle metabolism.24
As described above, there is a reversible decline in power output when athletes are asked to repeat short-duration sprints (< 10s), interspersed with brief recoveries.3 This is typically accompanied by a decrease in maximal, isometric contraction (MVC) force.26 While a concern with most studies is the time taken to transfer from the cycle ergometer to the isometric ergometer (to perform the MVC), using a specially-constructed cycle ergometer that allows a MVC to be performed directly on the cycle ergometer, we have shown that there is little influence of the short time that it takes to transfer from one ergometer to the other. This decrease in the ability of muscle to produce force has typically been attributed to the generation of an inadequate motor command in the motor cortex (i.e. neural factors) and/or factors related to metabolite accumulation or energy supply (i.e. muscular factors).
As maximal sprint exercise demands high levels of neural drive,27 failure to fully activate the contracting musculature should decrease force production and reduce intermittent-sprint performance. Potential changes in muscle activation during, and following, intermittent-sprint exercise have traditionally been assessed by surface electromyogram (EMG), percutaneous motor nerve (MN) stimulations, and, more recently, trans-cranial magnetic stimulation (TMS).
In conjunction with the decrease in maximum force/power production, a concurrent decline in the amplitude of EMG signals (integrated EMG values and root mean square; RMS) has been reported in several, although not all, studies. While further research is required, the changes in EMG appear to depend on the magnitude of fatigue reported. With mild fatigue (power decrement score < 10%), a steady level of neural activation during intermittent-sprint exercise has usually been reported. However, when there is greater fatigue (> 10%), a concurrent decline in mechanical performance and the amplitude of the EMG signals has consistently been reported across sprint repetitions. These changes in surface EMG activity suggest a suboptimal motor unit activity (i.e. a decrease in recruitment, firing rate, or both).
Although many studies have used changes in EMG as a proxy for changes in neural drive, difficulties in interpreting EMG data need to be acknowledged (e.g. amplitude cancellation phenomena, excessive sweat, changes in fibre membrane and motor unit properties).34 Another confounding factor when interpreting changes in EMG during intermittent-sprint exercise is the concurrent reduction in power output which makes it difficult to determine if the decrease in EMG activity is the consequence, or the cause, of the reduced power output. In an effort to resolve this issue, we recently used the EMG collected during three warm-up sprints to establish the power-EMG relationship and to determine whether the subsequent decrease in EMG previously observed during intermittent-sprint exercise was consistent with the decrease in power output (Girard, Racinais & Bishop; unpublished findings). Interestingly, there was only a 5.5% decrease in EMG, which was significantly less than that estimated from the power-EMG relationship established during the warm up (∼-23%) (Figure 2). These changes suggest the predominant cause of fatigue during intermittent-sprint exercise is not a decrease in neural drive (as inferred from changes in surface EMG).
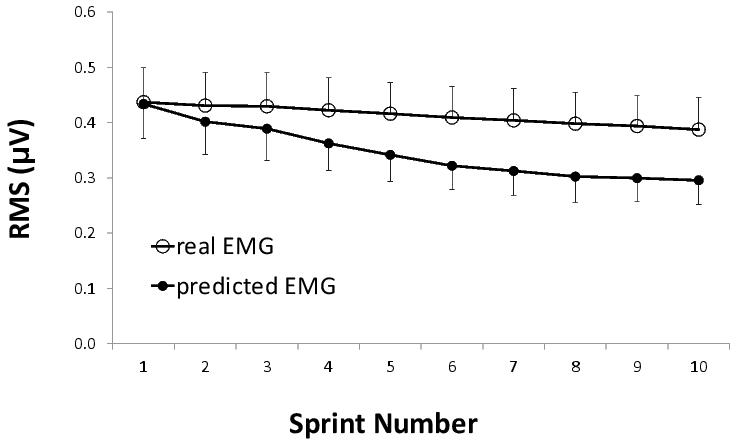
Figure 2. Changes in real and predicted (estimated from the pre-exercise power-EMG relationship) surface EMG during an intermittent-sprint test. The data are from 10 × 6s "all-out" sprints on a cycle ergometer interspersed with 30s of passive recovery. RMS = root mean square of the EMG signal.
Traditionally, MN stimulations applied mainly during isometric contractions (i.e. the twitch interpolation method) have been used to measure how much of the muscle’s possible force is produced by a voluntary contraction. Using this approach, we and others have established that there is only a small decrease in voluntary activation from the pre- to post-exercise MVC. This suggests that under conditions of considerable fatigue, failure to fully activate the contracting musculature may only make a small contribution to fatigue during intermittent-sprint exercise. This is consistent with the small changes in muscle activation inferred from changes in surface EMG activity.
A decrease in voluntary activation can theoretically arise from changes at the spinal level and/or supra-spinal factors. Potential supra-spinal factors include disturbances in brain electrical activity, cortical excitability and/or brain neurotransmitter (e.g. serotonin, dopamine, and acetylcholine) concentration. By applying TMS stimulations (together with conventional MN stimulation) during MVCs pre and post intermittent-sprint exercise it is possible to assess the completeness of cortically- and peripherally-derived estimates of voluntary activation. Using this method, we have recently reported that there is no change in voluntary activation (as estimated from TMS) following intermittent-sprint exercise.
The small changes in surface EMG, along with the lack of change in voluntary muscle activation (estimated from both percutaneous motor nerve stimulations and trans-cranial magnetic stimulation), indicate that there is little change in neural drive to the muscles following intermittent-sprint exercise. This, along with the observation that the decrease in EMG is much less than that which would be predicted from the decrease in power output, suggests that peripheral mechanisms are the predominant cause of fatigue during intermittent-sprint exercise.
Following intense dynamic contractions, there are marked ionic disturbances at the skeletal muscle level, arising secondary to decreases in Na+-K+-ATPase activity. In such cases, the Na+-K+ pump is not able to readily re-accumulate the potassium (K+) efflux from the muscle's cells, resulting in at least a doubling of the muscle extra-cellular [K+].39 While these modifications impair cell membrane excitability and depress force development, probably by slow inactivation of Na+ channels,40 unpublished observations have shown that plasma [K+], when corrected for changes in plasma volume, does not change following intermittent-sprint exercise. However, further research is required since: i) interstitial [K+] is considerably higher than venous plasma [K+] at similar work intensities; and ii) venous [K+] values may not reflect the concentration in the interstitium (i.e. the site where K+ may have its effects).39
An indirect measure of muscle excitability can be obtained by applying an electrical stimulus to peripheral nerves. Decreased muscle compound action potential (M-wave) amplitude, but not duration, has been reported after an intermittent-sprint running protocol, suggesting that action potential synaptic transmission, rather than propagation (i.e. impulse conduction velocity along the sarcolemma), may be impaired during such exercise.31 However, a potentiation of the M-wave response has also been reported following intermittent-sprint exercise.26 Thus, further research is therefore needed to determine if impairments in muscle excitability contribute to muscle fatigue induced by intermittent sprints.
Total intramuscular PCr stores are approximately 80 mmol·kg dm-1. As maximal rates of PCr breakdown can approach 9 mmol·kg dm-1·s-1,41 maximal sprinting therefore results in a severe reduction in intramuscular PCr content. For example, PCr concentration after a 6s sprint has been reported to be approximately 35–55 % of resting values.42-45 As recovery times during intermittent-sprint exercise generally do not exceed 60s, the ATP/PCr stores are likely to be only partially restored at the onset of each subsequent sprint. Coupled with the fact that the resynthesis of PCr and the recovery of power output follow similar time courses, it has been proposed that performance during intermittent-sprint exercise may become increasingly limited by PCr availability. Consistent with this hypothesis, significant correlations have been reported between the resynthesis of PCr and the recovery of power output in the first 10s of a second 30s sprint (r = 0.84; P < 0.05)5 and the partial restoration of intermittent-sprint performance (i.e. total work done) (r = 0.67, P < 0.05) (Unpublished observations). Furthermore, most research indicates that short-term creatine supplementation can improve intermittent-sprint performance.49 These results collectively suggest that PCr availability is an important determinant of intermittent-sprint performance.
Anaerobic glycolysis supplies approximately 40% of the total energy to a single 6s sprint, with a progressive decrease in glycolysis as sprints are repeated. As a consequence, there is an 8-fold decrease in the absolute ATP production from glycolysis from the first to the last sprint of 10 × 6s maximal sprints interspersed with 30s of recovery.10 While the reduction in the rate of ATP production by anaerobic processes is greater than the decrements in power output, these results nonetheless suggest that reduction in anaerobic glycolysis contributes to fatigue during intermittent-sprint exercise.
There is a perception that the capacity for oxidative metabolism is not an important determinant of intermittent-sprint performance as the contribution of oxidative phosphorylation to total energy expenditure during a single short sprint is quite small (< 10%). However, as sprints are repeated, the contribution of aerobic metabolism progressively increases such that aerobic metabolism may contribute as much as 40% of the total energy supply during the final sprints of an intermittent-sprint exercise.51 Surprisingly, subjects may even reach their VO2max during the latter sprints. This suggests that the contribution of aerobic metabolism during intermittent-sprint exercise may be limited by VO2max and that increasing VO2max may allow for a greater aerobic contribution during the latter sprints, potentially minimising fatigue. This hypothesis is supported by the observation that subjects with a greater VO2max are better able to maintain power output/sprint times during intermittent-sprint exercise, and is supported by significant correlations (r = −0.45 to −0.75) between VO2max and fatigue indices . However, it should be noted that not all studies have reported significant correlations between VO2max and fatigue indices during intermittent-sprint exercise (r = −0.20 to 0.30). The absence of stronger correlations between VO2max and intermittent-sprint performance may be related to the belief that the primary factor limiting VO2max is the ability of the cardio-respiratory system to deliver O2 to the contracting muscles, whereas intermittent-sprint performance may be primarily limited by peripheral muscle factors.66 This is supported by the observation that fatigue during intermittent-sprint exercise has been reported to be correlated with maximal ADP-stimulated mitochondrial respiration measured directly in muscle fibres.67 Thus, while perhaps not the most important factor, oxidative capacity does appear to be a determinant of fatigue during intermittent-sprint exercise.
There are a number of findings that suggest the considerable increases in muscle and blood hydrogen ion (H+) accumulation that occur during intermittent-sprint exercise may affect sprinting performance. For example, correlations have been observed between the level of fatigue during intermittent-sprint exercise and both muscle buffer capacity (βm) and changes in blood pH. Furthermore, the content of skeletal muscle monocarboxylate transporters (i.e. MCT1), which facilitate the intramuscular lactate and H+ removal process, has been inversely correlated with fatigue during intermittent-sprint exercise.71 While the mechanisms remain controversial, it is possible that H+ accumulation affects intermittent-sprint performance via adverse effects on the contractile machinery and/or through the inhibition of ATP derived from glycolysis, possibly via negative effects on phosphofructokinase and glycogen phosphorylase.72
In contrast to the above hypothesis, acidification as a direct cause of muscle fatigue has been challenged on at least three accounts: i) the time course of the recovery of force/power is much faster than that of pH; ii) high power outputs have been obtained under acidic conditions; iii) the ingestion of sodium bicarbonate (known to increase extra-cellular buffering capacity) has, in some cases, been reported not to affect intermittent-sprint performance. Furthermore, researchers have failed to observe a relationship between the recovery of muscle pH and the recovery of either 30s “all out” performance47 or intermittent-sprint performance (Unpublished observation). Further research is therefore needed to clarify whether H+ accumulation is an important determinant of fatigue during intermittent-sprint exercise.
Other factors, such as age,74 training status,59 and sex28 have also been reported to influence intermittent-sprint performance. In general, being young, female, or aerobically-trained has typically been associated with a smaller fatigue score. However, further research is required to establish whether these differences can be attributed to differences in fatigability or can largely be explained by differences in initial sprint performance.
During intermittent-sprint exercise, the inability to reproduce performance in subsequent sprints (fatigue) is manifested by a decline in sprint speed (running) or peak/mean power output (cycling). Although not extensively studied, failure to fully activate the contracting muscle does not appear to be a major determinant of fatigue during intermittent-sprint exercise. To date, the principal factors proposed to be responsible for fatigue include limitations in energy supply (e.g. PCr content and oxygen consumption) and the accumulation of by-products of metabolism (e.g. H+).
The author would like to acknowledge Prof Mark Hargreaves and Prof. Michael McKenna for inviting me to present this paper at the Australian Physiological Society meeting. I would also like to acknowledge and thank all of my past and present collaborators whose discussions infuse this manuscript. The author declares no conflict of interest.
1. Lannergren J, Westerblad H. Force decline due to fatigue and intracellular acidification in isolated fibres from mouse skeletal muscle. J. Physiol. (Lond.) 1991; 434:307-22.
2. Allen DG, Lamb GD, Westerblad H. Skeletal muscle fatigue: cellular mechanisms. Physiol. Rev. 2008; 88:287-332.
3. Bishop D, Edge J, Davis C, Goodman C. Induced metabolic alkalosis affects muscle metabolism and repeated-sprint ability. Med. Sci. Sports Exerc. 2004; 36:807-13.
4. Ball D, Burrows C, Sargeant AJ. Human power output during repeated sprint cycle exercise: the influence of thermal stress. Eur. J. Appl. Physiol. Occup. Physiol. 1999; 79:360-6.
5. Bogdanis GC, Nevill ME, Boobis LH, Lakomy HK. Contribution of phosphocreatine and aerobic metabolism to energy supply during repeated sprint exercise. J. Appl. Physiol. 1996; 80:876-84.
6. Girard O, Mendez-Villanueva A, Bishop D. Repeated-sprint ability - part I: factors contributing to fatigue. Sports Med. 2011; 41:673-94.
7. Mendez-Villanueva A, Hamer P, Bishop D. Fatigue in repeated-sprint exercise is related to muscle power factors and reduced neuromuscular activity. Eur. J. Appl. Physiol. 2008; 103:411–9.
8. Bishop D, Lawrence S, Spencer M. Predictors of repeated-sprint ability in elite female hockey players. J. Sci. Med. Sport 2003; 6:199-209.
9. Racinais S, Connes P, Bishop D, Blonc S, Hue O. Morning versus evening power output and repeated-sprint ability. Chronobiol. Int. 2005; 22:1029-39.
10. Gaitanos GC, Williams C, Boobis LH, Brooks S. Human muscle metabolism during intermittent maximal exercise. J. Appl. Physiol. 1993; 75:712-9.
11. Mendez-Villanueva A, Hamer P, Bishop D. Fatigue responses during repeated sprints matched for initial mechanical output. Med. Sci. Sports Exerc. 2007; 39:2219-25.
12. Bishop D, Edge J. Determinants of repeated-sprint ability in females matched for single-sprint performance. Eur. J. Appl. Physiol. 2006; 97:373-9.
13. Fitzsimons M, Dawson B, Ward D, Wilkinson A. Cycling and running tests of repeated sprint ability. Aus. J. Sci. Med. Sport 1993; 25:82-7.
14. Falgairette G, Billaut F, Giacomoni M, Ramdani S, Boyadjian A. Effect of inertia on performance and fatigue pattern during repeated cycle sprints in males and females. Int. J. Sports Med. 2004; 25:235-40.
15. Matsuura R, Arimitsu T, Yunoki T, Yano T. Effects of resistive load on performance and surface EMG activity during repeated cycling sprints on a non-isokinetic cycle ergometer. Br. J. Sports Med. 2009.
16. Little T, Williams AG. Effects of sprint duration and exercise: rest ratio on repeated sprint performance and physiological responses in professional soccer players. J. Strength Cond. Res. 2007; 21:646-8.
17. Buchheit M, Cormie P, Abbiss CR, Ahmaidi S, Nosaka KK, Laursen PB. Muscle deoxygenation during repeated sprint running: Effect of active vs. passive recovery. Int. J. Sports Med. 2009; 30:418-25.
18. Castagna C, Abt G, Manzi V, Annino G, Padua E, D'Ottavio S. Effect of recovery mode on repeated sprint ability in young basketball players. J. Strength Cond. Res. 2008; 22:923-9.
19. Spencer M, Bishop D, Dawson B, Goodman C, Duffield R. Metabolism and performance in repeated cycle sprints: active versus passive recovery. Med. Sci. Sports Exerc. 2006; 38:1492-9.
20. Balsom PD, Seger JY, Sjodin B, Ekblom B. Maximal-intensity intermittent exercise: effect of recovery duration. Int. J. Sports Med. 1992; 13:528-33.
21. Billaut F, Basset FA. Effect of different recovery patterns on repeated-sprint ability and neuromuscular responses. J. Sports Sci. 2007; 25:905-13.
22. Glaister M, Witmer C, Clarke DW, Guers JJ, Heller JL, Moir GL. Familiarization, reliability, and evaluation of a multiple sprint running test using self-selected recovery periods. J. Strength Cond. Res. 2009.
23. Ratel S, Williams CA, Oliver J, Armstrong N. Effects of age and recovery duration on performance during multiple treadmill sprints. Int. J. Sports Med. 2006; 27:1-8.
24. Spencer M, Dawson B, Goodman C, Dascombe B, Bishop D. Performance and metabolism in repeated sprint exercise: effect of recovery intensity. Eur. J. Appl. Physiol. 2008; 103:545-52.
25. Signorile JF, Ingalls C, Tremblay LM. The effects of active and passive recovery on short-term, high intensity power output. Can. J. Appl. Physiol. 1993; 18:31-42.
26. Racinais S, Bishop D, Denis R, Lattier G, Mendez-Villaneuva A, Perrey S. Muscle deoxygenation and neural drive to the muscle during repeated sprint cycling. Med. Sci. Sports Exerc. 2007; 39:268-74.
27. Ross A, Leveritt M, Riek S. Neural influences on sprint running: training adaptations and acute responses. Sports Med. 2001; 31:409-25.
28. Billaut F, Smith K. Prolonged repeated-sprint ability is related to arterial O2 desaturation in men. Eur. J. Appl. Physiol. 2010; 5:197-209.
29. Billaut F, Davis JM, Smith KJ, Marino FE, Noakes TD. Cerebral oxygenation decreases but does not impair performance during self-paced, strenuous exercise. Acta Physiol. 2010; 198:477-86.
30. Billaut F, Buchheit M. Hypoxia lowers muscle reoxygenation during repeated sprints. Med. Sci. Sport Exerc. 2011; 43(5), :152.
31. Perrey S, Racinais S, Saimouaa K, Girard O. Neural and muscular adjustments following repeated running sprints. Eur. J. Appl. Physiol. 2010; 109:1027-36.
32. Hautier CA, Arsac LM, Deghdegh K, Souquet J, Belli A, Lacour JR. Influence of fatigue on EMG/force ratio and cocontraction in cycling. Med. Sci. Sports Exerc. 2000; 32:839-43.
33. Matsuura R, Arimitsu T, Kimura T, Yunoki T, Yano T. Effect of oral administration of sodium bicarbonate on surface EMG activity during repeated cycling sprints. Eur. J. Appl. Physiol. 2007; 101:409-17.
34. Farina D, Merletti R, Enoka RM. The extraction of neural strategies from the surface EMG. J. Appl. Physiol. 2004; 96:1486-95.
35. Gandevia SC. Spinal and supraspinal factors in human muscle fatigue. Physiol. Rev. 2001; 81:1725-89.
36. Meeusen R, Watson P, Hasegawa H, Roelands B, Piacentini MF. Central fatigue: the serotonin hypothesis and beyond. Sports Med. 2006; 36:881-909.
37. Fraser SF, Li JL, Carey MF, Wang XN, Sangkabutra T, Sostaric S, Selig SE, Kjeldsen K, McKenna MJ. Fatigue depresses maximal in vitro skeletal muscle Na+-K+-ATPase activity in untrained and trained individuals. J. Appl. Physiol. 2002; 93:1650-9.
38. Clausen T, Nielsen OB, Harrison AP, Flatman JA, Overgaard K. The Na+,K+ pump and muscle excitability. Acta Physiol. Scand. 1998; 162:183-90.
39. Juel C, Pilegaard H, Nielsen JJ, Bangsbo J. Interstitial K(+) in human skeletal muscle during and after dynamic graded exercise determined by microdialysis. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2000; 278:R400-6.
40. Ruff RL, Simoncini L, Stuhmer W. Slow sodium channel inactivation in mammalian muscle: a possible role in regulating excitability. Muscle Nerve 1988; 11:502-10.
41. Hultman E, Sjoholm H. Energy metabolism and contraction force of human skeletal muscle in situ during electrical stimulation. J. Physiol. (Lond.) 1983; 345:525-32.
42. Boobis L, Williams C, Wootton S. Human muscle metabolism during brief maximal exercise. J. Physiol. (Lond.) 1982; 338:22P.
43. Dawson B, Goodman C, Lawrence S, Preen D, Polglaze T, Fitzsimons M, Fournier P. Muscle phosphocreatine repletion following single and repeated short sprint efforts. Scand. J. Med. Sci. Sports 1997; 7:206-13.
44. Parra J, Cadefau J, Rodas G, Amigo N, Cusso R. The distribution of rest periods affects performance and adaptations of energy metabolism induced by high-intensity training in human muscle. Acta Physiol. Scand. 2000; 169:157-65.
45. Gaitanos GC, Williams C, Boobis LH, Brooks S. Human muscle metabolism during intermittent maximal exercise. J. Appl. Physiol. 1993; 75(2):712-9.
46. Dawson B, Goodman C, Lawrence S, Preen D, Polglaze T, Fitzsimons M, Fournier P. Muscle phosphocreatine repletion following single and repeated short sprint efforts. Scand. J. Med. Sci. Sports 1997; 7:206-13.
47. Bogdanis GC, Nevill ME, Boobis LH, Lakomy HK, Nevill AM. Recovery of power output and muscle metabolites following 30s of maximal sprint cycling in man. J. Physiol. 1995; 482 ( Pt 2):467-80.
48. Sahlin K, Ren JM. Relationship of contraction capacity to metabolic changes during recovery from a fatiguing contraction. J. Appl. Physiol. 1989; 67:648-54.
49. Bishop D. Dietary supplements and team-sport performance. Sports Med. 2010; 40:995-1017.
50. Boobis L, Williams C, Wootton SA. Human muscle metabolism during brief maximal exercise. J. Physiol. 1982; 338:21P-2P.
51. McGawley K, Bishop D. Anaerobic and aerobic contribution to two, 5 × 6s repeated-sprint bouts. Coaching Sport Sci. J. 2008; 3:52.
52. Parolin ML, Chesley A, Matsos MP, Spriet LL, Jones NL, Heigenhauser GJ. Regulation of skeletal muscle glycogen phosphorylase and PDH during maximal intermittent exercise. Am. J. Physiol. 1999; 277:E890-900.
53. Girard O, Millet GP. Neuromuscular fatigue in racquet sports. Neurol. Clin. 2008; 26:181-94; x.
54. Stolen T, Chamari K, Castagna C, Wisloff U. Physiology of soccer: an update. Sports Med. 2005; 35:501-36.
55. Dupont G, Millet GP, Guinhouya C, Berthoin S. Relationship between oxygen uptake kinetics and performance in repeated running sprints. Eur. J. Appl. Physiol. 2005; 95:27-34.
56. Bishop D, Spencer M. Determinants of repeated-sprint ability in well-trained team-sport athletes and endurance-trained athletes. J. Sports Med. Phys. Fitness 2004; 44:1-7.
57. Brown PI, Hughes MG, Tong RJ. Relationship between VO2max and repeated sprint ability using non-motorised treadmill ergometry. J. Sports Med. Phys. Fitness 2007; 47:186-90.
58. Dawson B, Fitzsimons M, Ward D. The relationship of repeated sprinting ability to aerobic power and performance measures of anaerobic capacity and power. Aus. J. Sci. Med. Sport 1993; 25:88-93.
59. Rampinini E, Sassi A, Morelli A, Mazzoni S, Fanchini M, Coutts AJ. Repeated-sprint ability in professional and amateur soccer players. Appl. Physiol. Nutr. Metab. 2009; 34:1048-54.
60. Aziz AR, Mukherjee S, Chia MY, Teh KC. Relationship between measured maximal oxygen uptake and aerobic endurance performance with running repeated sprint ability in young elite soccer players. J. Sports Med. Phys. Fitness 2007; 47:401-7.
61. Castagna C, Manzi V, D'Ottavio S, Annino G, Padua E, Bishop D. Relation between maximal aerobic power and the ability to repeat sprints in young basketball players. J. Strength Cond. Res. 2007; 21:1172-6.
62. Lane KN, Wenger HA, Blair C. Relationship between maximal aerobic power and the ability to recover from repeated, high intensity, on ice sprints in male ice hockey players. Can. J. Appl. Physiol. 1997; 22:35.
63. McMahon S, Wenger HA. The relationship between aerobic fitness and both power output and subsequent recovery during maximal intermittent exercise. J. Sci. Med. Sport 1998; 1:219-27.
64. Wadley G, Le Rossignol P. The relationship between repeated sprint ability and the aerobic and anaerobic energy systems. J. Sci. Med. Sport 1998; 1:100-10.
65. Aziz AR, Chia M, Teh KC. The relationship between maximal oxygen uptake and repeated sprint performance indices in field hockey and soccer players. J. Sports Med. Phys. Fitness 2000; 40:195-200.
66. Bassett DR, Jr., Howley ET. Limiting factors for maximum oxygen uptake and determinants of endurance performance. Med. Sci. Sports Exerc. 2000; 32:70-84.
67. Thomas C, Sirvent P, Perrey S, Raynaud E, Mercier J. Relationships between maximal muscle oxidative capacity and blood lactate removal after supramaximal exercise and fatigue indexes in humans. J. Appl. Physiol. 2004; 97:2132-8.
68. Edge J, Bishop D, Hill-Haas S, Dawson B, Goodman C. Comparison of muscle buffer capacity and repeated-sprint ability of untrained, endurance-trained and team-sport athletes. Eur. J. Appl. Physiol. 2006; 96:225-34.
69. Bishop D, Claudius B. Effects of induced metabolic alkalosis on prolonged intermittent-sprint performance. Med. Sci. Sports Exerc. 2005; 37:759-67.
70. Bishop D, Edge J, Goodman C. Muscle buffer capacity and aerobic fitness are associated with repeated-sprint ability in women. Eur. J. Appl. Physiol. 2004; 92:540-7.
71. Thomas C, Perrey S, Lambert K, Hugon G, Mornet D, Mercier J. Monocarboxylate transporters, blood lactate removal after supramaximal exercise, and fatigue indexes in humans. J. Appl. Physiol. 2005; 98:804-9.
72. Spriet LL, Lindinger MI, McKelvie RS, Heigenhauser GJ, Jones NL. Muscle glycogenolysis and H+ concentration during maximal intermittent cycling. J. Appl. Physiol. 1989; 66:8-13.
73. Gaitanos GC, Nevill ME, Brooks S, Williams C. Repeated bouts of sprint running after induced alkalosis. J. Sports Sci. 1991; 9:355-70.
74. Mujika I, Spencer M, Santisteban J, Goiriena JJ, Bishop D. Age-related differences in repeated-sprint ability in highly trained youth football players. J. Sports Sci. 2009; 27:1581-90.