Background: Nano-sized therapeutics can be carefully engineered to promote targeted delivery to sites of action and thereby maximise therapeutic effects and minimise harmful side effects. For instance, following injection into tissues nano-therapeutics are typically too large to directly enter blood capillaries and instead are cleared via the more permeable lymphatic capillaries and nodes. This property has been exploited in the use of nano-therapeutics to enhance vaccination and to treat lymphatic cancer metastases. However, much is still to be learned regarding the mechanisms that direct the biodistribution of nano-therapeutics. For instance, recent studies in our lab demonstrated that even intravenously administered nano-therapeutics transfer to the lymph in substantial quantities (Kaminskas et al., 2013). The current studies aim to determine the site and mechanism by which blood-to-lymph transfer of nano-therapeutics occurs in order to advance the fundamental understanding of the mechanisms that direct the biodistribution of nano-therapeutics and inform better design of nano-therapeutics.
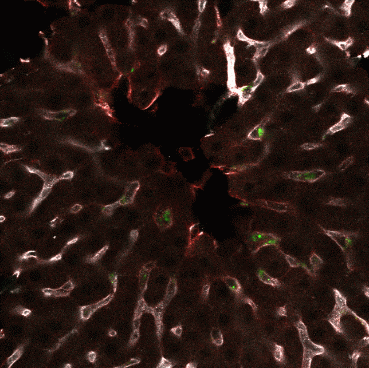
Methods: All experiments were conducted in male Sprague Dawley rats anaesthetized via SC injection of 37 mg/kg ketamine, 10 mg/kg xylazine, 0.4 mg/kg acepromazine followed by 40 mg/kg ketamine, 0.4 mg/kg acepromazine when required. To determine the body region(s) in which blood-to-lymph transfer of nano-therapeutics occurs, the jugular vein (to enable intravenous injection), carotid artery (to enable blood collection) and liver, intestine or thoracic lymphatic duct (to enable lymph collection) were cannulated. The nano-therapeutic PEGylated interferon α2a (IFN-PEG40, 60 kDa) was then administered via rapid infusion over 2 min at a dose of 5 μg/kg. Blood and lymph samples were subsequently collected at multiple time points for more than 7 h and plasma separated from blood. IFN-PEG40 concentrations in plasma and lymph was quantified using a commercially available ELISA kit for IFNα2. Two-photon microscopy was also utilised to visualise the mechanisms directing blood to lymph transfer of a model 50nm sized fluorescent nano-therapeutic in the liver and intestine.

Conclusion: A substantial proportion of blood-to-lymph transfer of nano-therapeutics occurs in the intestine and liver. Within the liver the transfer is particularly efficient and involves extravasation of nano-therapeutics from the fenestrated sinusoids followed by diffusion to the liver lymphatics. Intravenously administered nano-therapeutics therefore facilitate targeted delivery to the liver and intestinal lymphatics which may be beneficial in the treatment of diseases involving these lymphatic sites, such as chronic liver diseases, liver transplant rejection, cancer metastases, and autoimmune diseases. Future studies will assess whether the site and mechanism of blood-to-lymph transfer, and therapeutic effect, of nano-therapeutics can be controlled via modulation of the physicochemical characteristics of the nano-therapeutic.
Kaminskas LM, Ascher DB, McLeod VM, Herold MJ, Le CP, Sloan EK, Porter CJ. (2013) Journal of Controlled Release 168, 200-8.