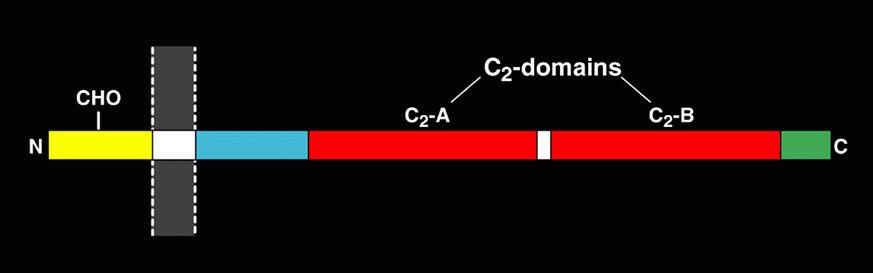
Neurotransmitters, neuropeptides and hormones are released through regulated exocytosis of synaptic vesicles (SVs) and large dense core vesicles (LDCVs), a process that is controlled by Ca2+. Synaptotagmins are a family membrane proteins that share a common domain structure (Figure 1).

Figure 1. Schematic drawing of domain structure for synaptotagmin-1. Other synaptotagmins share a similar domain structure with synaptotagmin-1, which is composed of a short N-terminal sequence, a single transmembrane domain, and two functional C2 domains. The C2 domains bind to Ca2+ and are critical in regulating neurotransmitter release and hormone secretion.
Most synaptotagmins are expressed in brain and endocrine cells, and some of these synaptotagmins bind to phospholipids and Ca2+ at the levels that trigger regulated exocytosis of SVs and LDCVs. A major research interest in my lab is to understand the molecular regulation of insulin secretion. The cellular process of insulin secretion is well established (Figure 2), and numerous molecular players involved in insulin granule biogenesis, trafficking and exocytosis have been documented and analyzed. Although it is well known that incretins, such as GLP-1 can potentiate insulin secretion (Figure 2), its underlying molecular mechanisms are poorly defined. We have used mouse genetic and physiology approaches, along with biochemistry and molecular biology to analyze the effects of depleting synaptotagmins on insulin secretion, and whether synaptotagmins are involved in the incretin potentiation of insulin secretion. Depletion of synaptotagmin-7 in mice results in impaired glucose tolerance and reduced insulin secretion, along with defective response to GLP-1 stimulation. Furthermore, synaptotagmin-7 undergoes post-translational modification in response to signaling pathways that potentiate insulin secretion. As synaptotagmin-7 regulates insulin granule exocytosis only in elevated intracellular calcium levels, downstream of high glucose-induced metabolic and membrane events, synaptotagmin-7 may represent an ideal target in enhancing glucose-sensitive insulin secretion in diabetes treatment.
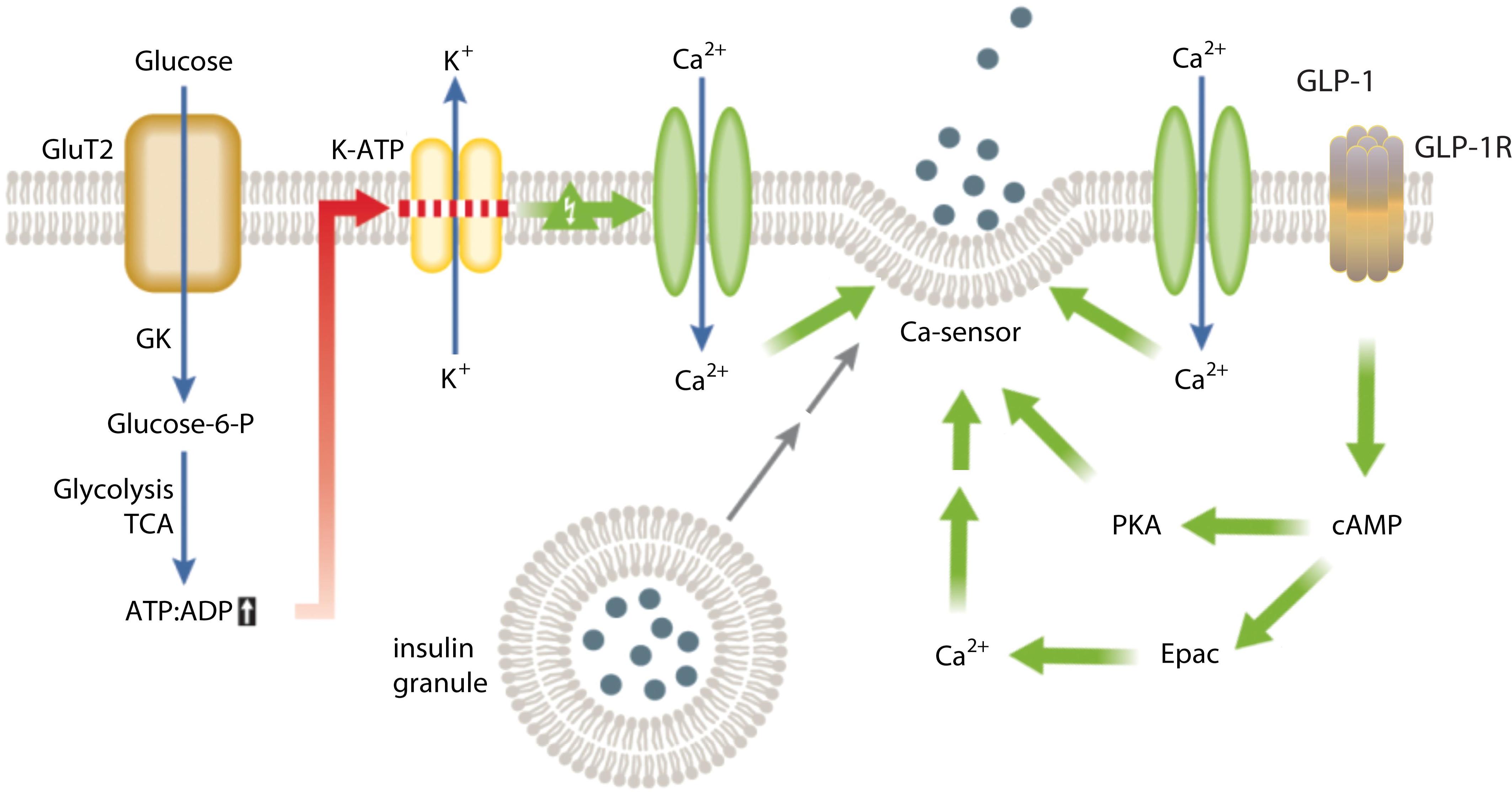
Figure 2. Molecular regulation of insulin secretion and potentiation by GLP-1 dependent signaling. Insulin granule exocytosis is triggered by elevated intracellular Ca2+ levels, which is sensed by Ca2+-sensors and the signal is relayed to SNARE proteins to execute the fusion process. GLP-1 binding to its receptor evokes activation of PKA and increased Ca2+ levels, which act on intracellular targets to potentiate insulin secretion.