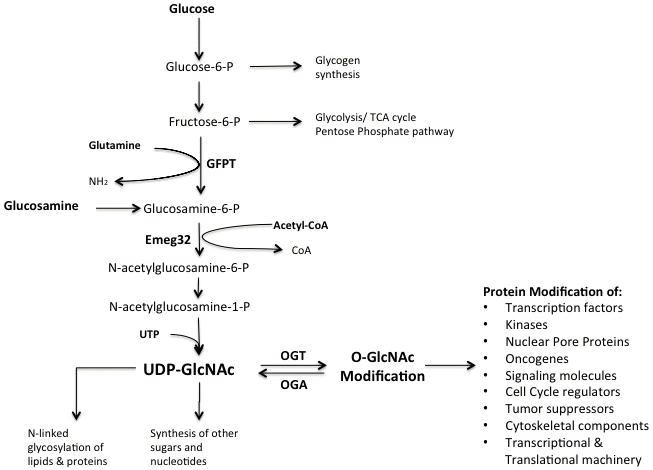
A stressful environment during development can program adult disease propensity in a sexually dimorphic manner. Female offspring appear to be more resistant to an adverse maternal environment whereas male offspring are adversely affected. Our previous studies of short-term (60h) exposure to maternal glucocorticoid during gestation suggest that male offspring susceptibility to adult disease may occur at the level of the placenta (Cuffe et al., 2012). Although the mechanisms by which the fetal/placental unit senses and responds to stressful environments are not understood, the ability of cells to respond to multiple forms of stress is associated with rapidly increased glucose uptake and metabolism through the hexosamine signalling pathway (HSP) outlined in the figure. This is an essential survival response of adaptive importance in healthy cells.

Our results demonstrate that the O-GlcNAcylation response to stress is blunted in male placentae relative to female placentae suggesting dysregulation of this signaling pathway may underlie the sexually dimorphic response to a stressful environment in utero. Increased levels of the stress-responsive, pro-survival factor Akt2, in placentae from female (P<0.05), but not male fetuses following glucoccorticoid administration further supports this notion. Given that dysregulation of O-GlcNAcylation is implicated in a growing number of adult diseases our results suggest that sex-specific alterations in placental OGT levels in response to stress may act to protect the female fetus from propensity to adult disease in later life.
Cuffe JS, O'Sullivan L, Simmons DG, Anderson ST, Moritz KM. (2012). Endocrinology 153: 5500-5511.