Background: Insulin resistance (IR) underpins a spectrum of prevalent and inadequately treated cardiometabolic diseases, including type 2 diabetes (T2D). Excess adipose, particularly visceral adipose tissue (VAT), increases the risk of IR. The expansion of VAT leads to pathogenic inflammatory and metabolic changes in VAT that stimulate IR. The intestinal lymphatics flow through VAT. High fat diets (HFD) promote hyperpermeability of the intestinal lymphatics and VAT expansion. VAT accumulation and expansion is accelerated in transgenic rodents with dysfunctional lymphatics. The content of inflammatory and metabolic mediators in intestinal lymph is also increased in response to HFD. Altogether this data suggests that increased exposure of VAT to intestinal lymph promotes VAT expansion and this may promote IR development.
Aim: To determine whether lymph leakage from the intestinal lymphatics stimulates changes to VAT function that promote the development of whole-body IR
Methods: 3T3-L1 cells at different stages of development into adipocytes from ‘fibroblast’ to ‘mature adipocyte’ were exposed to 2% mesenteric lymph fluid collected from rats fed with a chow fat diet (CFD) or HFD for 4-6 weeks. All experiments were conducted in male Sprague Dawley rats anaesthetised via SC injection of 37 mg/kg ketamine, 10 mg/kg xylazine, 0.4 mg/kg acepromazine followed by 40 mg/kg ketamine, 0.4 mg/kg acepromazine when required. To determine if lymph promotes changes in adipocyte function consistent with the changes that promote IR in vivo, the impact of lymph on adipocyte differentiation, lipid accumulation, inflammatory cytokine expression and fatty acid release (lipolysis) was assessed. Additionally, conditioned media experiments were completed to determine whether lymph alters the secretory profile of adipocytes to promote muscle IR. 3T3-L1 adipocytes were treated with CFD or HFD lymph for 48 h, the lymph removed and cells incubated in standard media for 24 h to produce ‘conditioned media’. L6-Glut4 myotubes were then treated with the conditioned media for 48 h then insulin sensitivity (2-deoxy-glucose uptake) assessed. Statistical differences were determined via student t-tests or two way ANOVA with α<0.05 significance level.
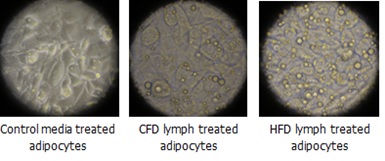
Results: Exposure of 3T3-L1 fibroblasts to lymph accelerated adipocyte differentiation with early lipid accumulation and droplet formation (Figure), and significantly increased mRNA expression of markers of differentiation (PPARγ, leptin and GAPDH) and lipid storage (ap-2). Exposure of mature adipocytes to lymph for 2 days also increased lipid accumulation. Mature adipocyte lipolysis was upregulated directly after lymph exposure but was significantly lower 2 days after lymph removal. Surprisingly, despite the changes in adipocyte function, the initial results for the conditioned media experiments showed a trend toward increased 2-deoxy-glucose uptake and thereby insulin sensitivity in the myotubes treated with conditioned media from lymph exposed adipocytes. In all cases the differences were more pronounced in HFD lymph treated cells versus CFD lymph treated.

Conclusion: Lymph exposure, particularly HFD fed lymph, promoted adipocyte functional changes consistent with the changes that promote IR in vivo. The functional changes led to the secretion of mediators from adipocytes that influenced skeletal muscle cell insulin sensitivity and glucose utilization. These studies advance the fundamental understanding of the pathogenic drivers of IR. Future in vitro and in vivo investigations will confirm the mechanism by which increased exposure to intestinal lymph induces changes in adipose function that influence whole body IR development, and will explore the potential to treat IR by modulating these processes.