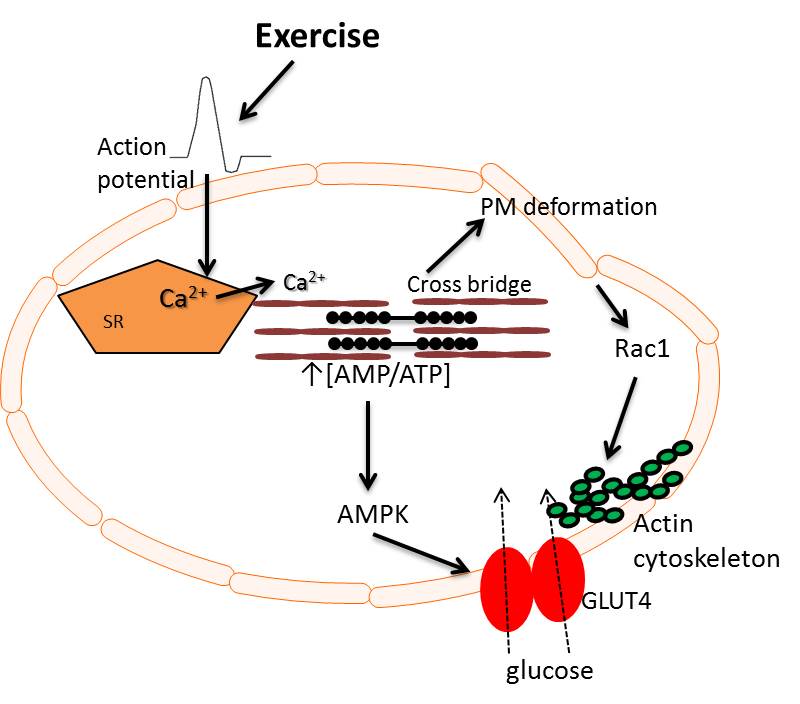
Exercise bypasses insulin resistance to increase glucose uptake in skeletal muscle and therefore represents an important alternative to insulin-stimulated glucose uptake. Nevertheless, the signaling mechanisms that mediate glucose uptake during exercise are not well understood. Recent data from our laboratory indicate that both Rac1 and AMPK, but not sarcoplasmatic reticulum Ca2+, regulate contraction-stimulated glucose transport in muscle. However, it is unknown whether Rac1 regulates exercise-induced muscle glucose uptake in vivo and furthermore whether Rac1 and AMPK alone can account for glucose uptake during muscle contraction.
Exercise-induced uptake of labelled 2-deoxyglucose (2-DG) was blocked in soleus and decreased by 80 and 60 % in gastrocnemius and tibialis anterior muscles, respectively in muscle-specific inducible Rac1 knockout (mKO) compared to wildtype (WT) littermate mice. In addition, GLUT4 translocation to the sarcolemma during exercise was blocked in Rac1 mKO muscle. Furthermore, whereas muscle specific Rac1 mKO or overexpression of a dead α2 AMPK kinase (KD) each partially decreased contraction-induced 2-DG transport, the effect was additive in Rac1 mKO mice crossed with AMPK KD mice and glucose transport was blocked in soleus and reduced by 80% in double mKO mice. In agreement, pharmacological Rac1 inhibition of incubated muscles from either AMPK β1β2 knockout or AMPK KD mice nearly abolished contraction-stimulated glucose transport (P<0.01). In conclusion, Rac1 is a regulator of exercise-induced GLUT4 translocation and glucose uptake in skeletal muscle in mice, and together with AMPK signalling Rac1 can account for most of the signal to glucose transport during muscle contraction as shown in the Figure.
