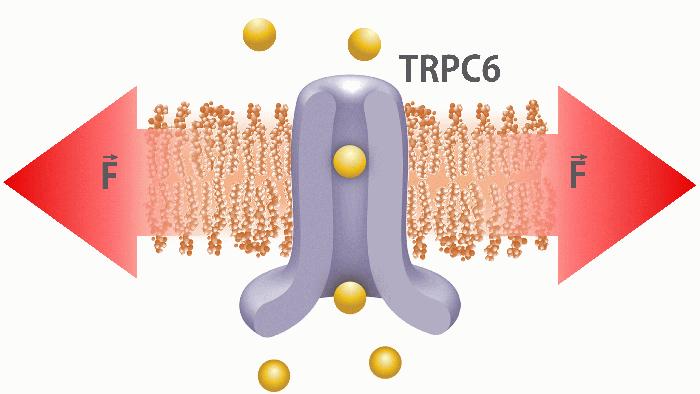
The transient receptor potential (TRP) ion channel family is a diverse group of channels gated by various physical and chemical stimuli. One of the members, TRPC6, is a calcium permeable cation channel (PCa/PNa ~ 6) located in the T-tubules of ventricular cardiomyocytes (Allen & Ward, 2011). Recent studies support the role of TRPC6 ion channels as one of the players in mechanotransduction of the heart. Abnormal TRPC6 activity has been implicated in cardiac hypertrophy, a maladaptive process likely triggered by increased mechanical stress (Seo et al., 2014). TRPC6 is gated via G-protein-coupled receptors (GPCRs) pathway which leads to generation of diacylglycerol (DAG) which ultimately activates the ion channel (Bush et al., 2006). TRPC6 can also be activated by mechanical force (Spassova et al., 2006; Wilson & Dryer, 2014). However, it is still unclear if TRPC6 is activated directly by membrane tension (bilayer mechanism; see Figure) or whether activation requires additional components including cytoskeleton and/or extracellular matrix (tether mechanism) for its activation.

In this study, we investigated the mechanosensitivity of hTRPC6 overexpressed in HEK293 cells. We have transfected C-terminal fusion hTRPC6 construct into HEK293 cells and demonstrated by confocal image it’s membrane localisation. We revealed the impact of TRPC6 channels on Ca2+ transients in stretched HEK293 cells. Our home-made stretch device is able to produce isotropic planar stretch. By applying 15% stretch to the cells we recorded stretch-activated Ca2+ entry via TRPC6 ion channel. Increase in Ca2+ fluorescence in these cells was monitored using Rhod-2 AM red calcium indicator. By applying TRPC6 agonist hyperforin (St. John's wort) it was possible to activate the channel and record Ca2+ influx without any stretch.
Moreover, by using patch clamp technique, we recorded TRPC6 single channel activity in cell-attached and whole-cell configurations. By applying negative pressure (suction) of -50 mmHg to the patch pipette we were able to record single channel activity of TRPC6 channels.Furthermore, by applying specific activators/inhibitors we were able to increase/decrease the channel activity on a single channel level.
We purified TRPC6 protein and investigated the mechanosensitivity of TRPC6 reconstituted into the liposomes. Purification were performed using GFP nanobodies. To assess the integrity of the TRPC6 protein, cell lysate was analysed by fluorescence sized exclusion chromatography (FSEC) and SDS/PAGE gel. GFP fluorescence peak using FSEC showed that TRPC6 channel is found mostly in tetramer state. Fluorescent scan of the gels demonstrated a single fluorescent band at the correct TRPC6 - GFP size. The final TRPC6 product coomassie staining gel revealed single band at the right monomer size. Using isolated protein, liposome reconstitution was carried out using DR method. We recorded TRPC6 single channel activity in liposomes by patch clamping. TRPC6 ion channel activity demonstrated outward rectification and calculated conductance was ∼22 pS. By applying negative pressure to the patch pipette, we did observe channel activity modulation. TRPC6 was spontaneously active without any negative pressure applied to the patch pipette, which suggests that the membrane tension resulting from the gigaohm seal formation was sufficient to activate the TRPC6 channels. All together these results confirm the activation mechanism of TRPC6 ion channel by membrane stretch.
Allen DG & Ward ML. (2011) In Cardiac Mechano-Electric Coupling and Arrhythmias 2nd ed. Kohl P, Sachs F, Franz M. (eds) pp. 435-441. Oxford University Press, Oxford UK. DOI: 10.1093/med/9780199570164.003.0061
Bush EW, Hood DB, Papst PJ, Chapo JA, Minobe W, Bristow MR, Olson EN, McKinsey TA. (2006) J Biol Chem 281, 33487-33496.
Seo K, Rainer PP, Shalkey Hahn V, Lee DI, Jo SH, Andersen A, Liu T, Xu X, Willette RN, Lepore JJ, Marino JP Jr, Birnbaumer L, Schnackenberg CG, Kass DA. (2014) Proc Natl Acad Sci USA 111, 1551-56.
Spassova MA, Hewavitharana T, Xu W, Soboloff J, Gill DL. (2006) Proc Natl Acad Sci USA 44, 16586-91.
Wilson C, Dryer SE. (2014) Am J Physiol Renal Physiol 306, F1018-25.