Non-steroidal anti-inflammatory drugs (NSAIDs) are under investigation for use as chemopreventives for colorectal cancer (Hawksworth et al., 2014). This work stems from their ability to inhibit cyclooxygenase (COX), and the finding that COX-2 overexpression has been linked to many cancers, particularly colon cancer (Hawksworth et al., 2014). Unfortunately, adverse side-effects, predominantly gastrointestinal ulceration, limit the chemopreventive use of NSAIDs. Since bismuth complexes, e.g. colloidal bismuth subcitrate, are marketed for the treatment of gastrointestinal disorders (including ulcers), we have been investigating BiNSAIDs as chemopreventive agents whereby we have hypothesized that the gastric damage caused by the NSAIDs could be combated by the gastrointestinal protective properties of bismuth (for review see Hawksworth et al., 2014). Recent preliminary in vivo studies in our group support this claim (in part) whereby reduced small intestinal ulceration has been observed following BiNSAID-administration vs NSAID administration.
However, many questions remain unanswered. For instance, are BiNSAIDs and NSAIDs taken up by cancer cells to the same extent? How do they traverse/interact with the cell membrane? Do they both interact with the monocarboxylate transporter (responsible for NSAID uptake) and COX (the target enzyme of NSAIDs)? This presentation will describe our work on model membrane and cell systems to begin to attempt to answer some of these questions. Studies on Bi uptake have been performed in bowel cancer cells. This presentation will focus primarily on our work via Quartz Crystal Microbalance studies and Neutron Reflectometry (NR) on the interaction of a range of NSAIDs and the bismuth analogues interacting with POPC and POPC/Cholesterol model bilayer systems.
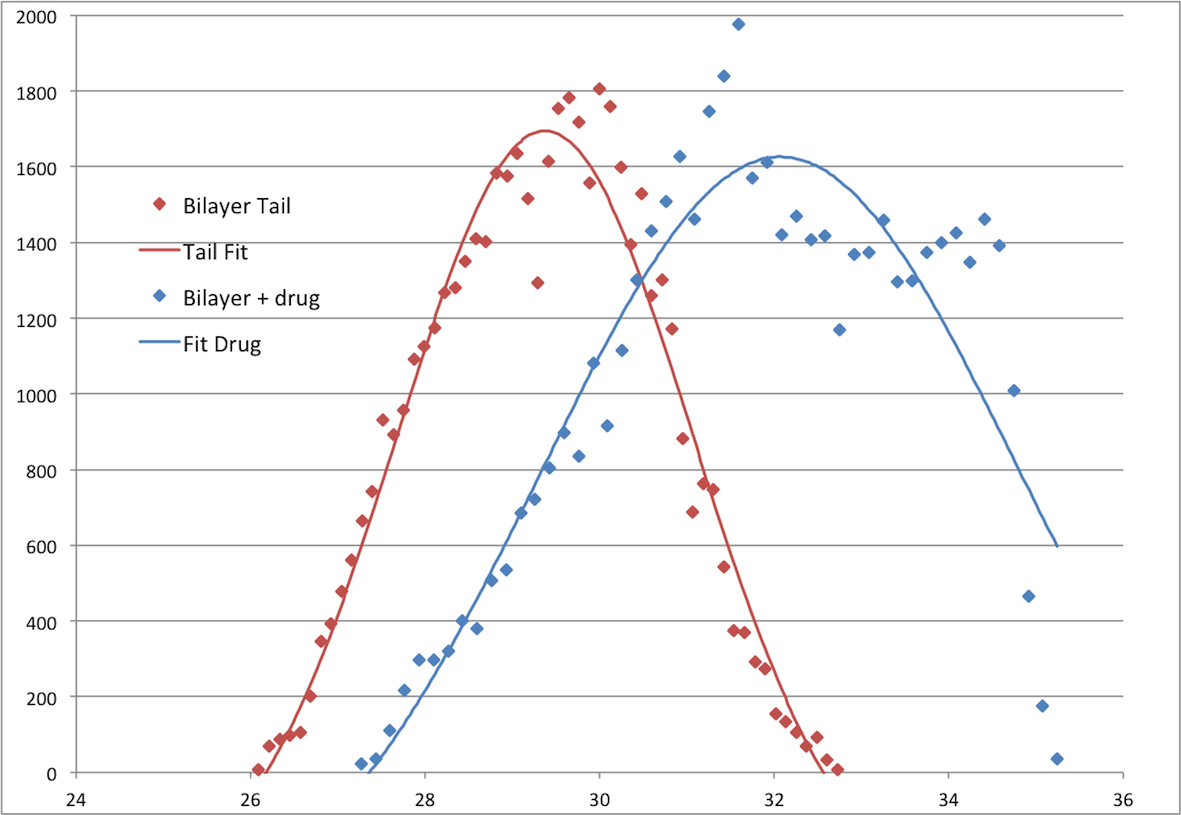
We see clear evidence of the incorporation of and washout of the BiNSAIDS and NSAIDS into bilayers. The figure shows thickness (x–axis) versus frequency of result (y-axis) for the tail region of a POPC bilayer as formed (red) and after interaction with Bi(indomethacinate)3. Data points (blue) show the distribution of the fitted NR thickness values for the hydrophobic region of the tails before and after the introduction of Bi(indomethacinate)3 solution to the bilayer. It is clear that the BiNSAID has both perturbed the thickness of the bilayer and increased the disorder in the system as evidenced by the greater width of the Gaussian fit to the parameter value.
Hawksworth EL, Andrews PC, Lie W, Lai B, Dillon CT. (2014) J Inorg Biochem 135, 28-39.
