Background: The adult mammalian heart has very little capacity to repair after damage because adult cardiomyocytes lack the ability to proliferate. The expression of some microRNAs (miRNA), which have roles in regulating cell cycle entry and proliferation, change at the time when proliferation ceases in both rodents and sheep. We therefore investigated the expression of miRNAs and their predicated target genes after myocardial infarction in sheep hearts before and after birth when the response to injury is different.
Methods: We used sheep, which have a similar pattern of cardiomyocyte development relative to birth compared to humans, as a model to investigate the effect of age on the response to cardiac damage, by ligating the second diagonal of the left anterior descending (LAD) coronary artery. Surgery was performed on fetuses (MI, n=5; Sham, n=5; 102 days gestation; cardiomyocytes are proliferative) and six month old sheep (MI, n=4; Sham, n=4; cardiomyocytes contribute to heart growth by hypertrophy). Ewes and six month old sheep underwent surgery under aseptic conditions with general anesthesia induced with diazepam (0.3mg/kg; intravenous) and ketamine (7mg/kg; intravenous) and maintained with inhalation of isoflurane (1-2%) in oxygen. Lignocaine was administered intravenously to all fetuses (0.2 ml bolus) and lambs (100 mg/500mL) prior to thoracotomy procedure. When the sheep were recovered from anesthesia, they were given analgesia (20 μg/kg, Xylazil). Three days after surgery, ewes and six month old sheep were humanely killed with an overdose of sodium pentobarbitone (8 g). Heart tissue from the infarct, border and remote zone of the left ventricle was dissected and flash frozen in liquid nitrogen or fixed in paraformaldehyde three days after surgery. Total RNA was extracted and analysed using a custom designed miRNA microarray (LC Sciences, USA) and validated with qRT-PCR and histology.
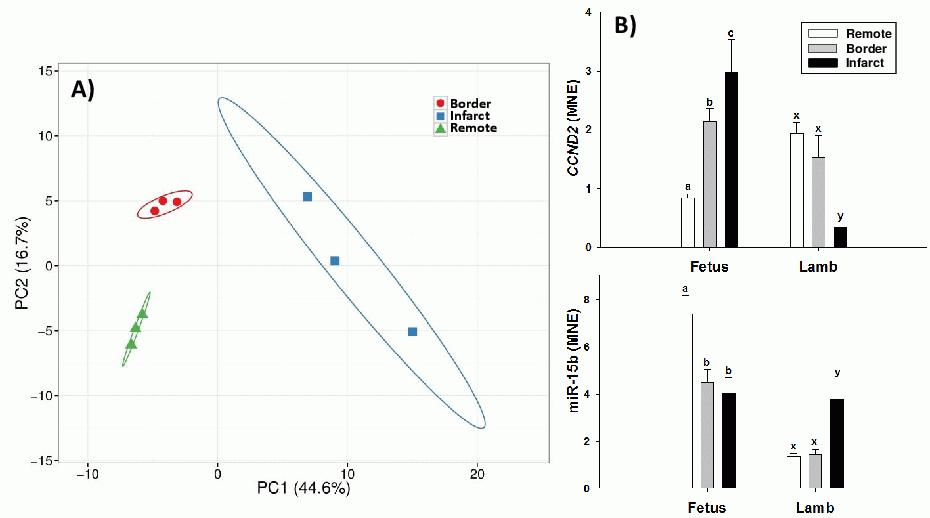
Results: The figure shows (A) principal component analysis (PCA) of fetal miRNA microarray data and (B) normalized cardiac miR-15b and CCND2 mRNA expression in infarct, border and remote zone in fetal and six month old sheep after ligation of the LAD. The differentially expressed probes from the microarray (Figure 1A) were subjected to k-means clustering in CLCBIO (k=10 revealed profile stability). The miRNA in each cluster were used as input into MiRWalk for mRNA target prediction. Predicted targets were assessed for Gene Ontology (GO) term enrichments using DAVID. Target genes of clustered miRNAs that were downregulated in the infarct area in the fetus showed enrichment for apoptosis and regulation of development. In contrast, target genes of clustered miRNAs in the six month old sheep showed enrichment for negative regulation of apoptosis and cell migration. Supporting this finding, miRNA and gene expression using qRT-PCR showed the fetus and six month old sheep had opposite expression profiles (Figure 1B).

Conclusions: miRNAs that are downregulated in the fetus following myocardial infarction may have significant roles in regulating cardiomyocyte proliferation in sheep. Low expression of these miRNAs in the fetus allows cardiac repair after damage with high expression after birth resulting in limited capacity for repair.