Introduction. More than a century has passed since Canon’s pioneering studies, and a vast literature now underpins the field of gut motility (Szurszewski 1998). However, it must be concluded that clinical translation has lagged. Many clinical disorders of gut motility are still poorly defined, and there remains a lack of reliable tools to guide diagnosis, with few therapies that effectively address root causes.
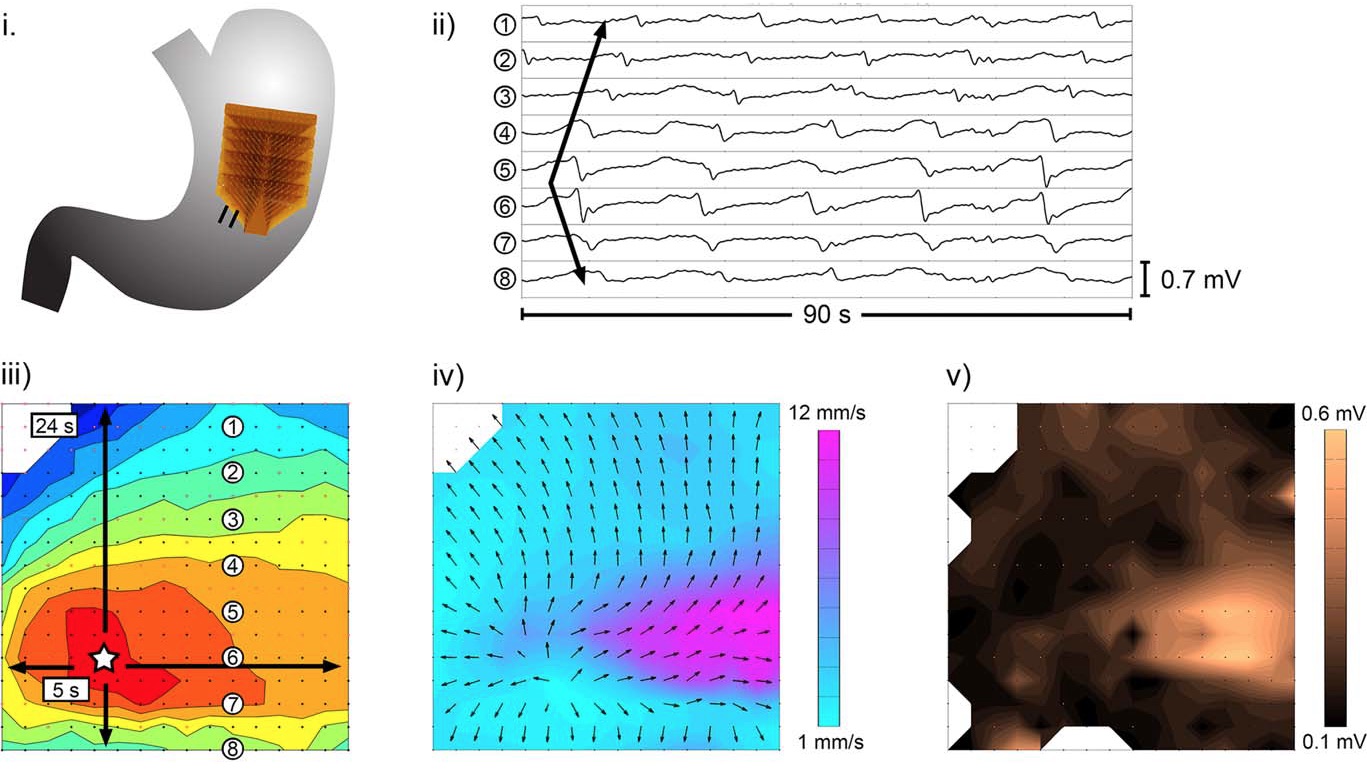
One area of success has been in oesophageal motility, where the direct, high-resolution (HR) analysis of motility patterns now allows the comprehensive classification of disease states (Kahrilas et al., 2015). However, throughout the remainder of the gut, from stomach to the rectum, clinical tools for reliably analysing motor patterns remain limited. Consequently, diagnostic classification schemes for abnormal motor patterns in the stomach, small intestine, colon and rectum and have remained embryonic. In recent years, however, translational progress in HR motility diagnostics has begun to advance (Dinning et al., 2010; Cheng et al., 2013). We describe two areas of significant current progress: HR electrical mapping and HR colonic manometry, with a specific focus on recent translational applications. HR electrical mapping is a technique adapted from modern methods in cardiac electrophysiology, involving the use of finely-spaced arrays of electrodes to track electrical propagation sequences in fine spatiotemporal detail (Du et al., 2009; O'Grady et al., 2013). This method was pioneered in the GI tract by Lammers et al., who custom-built silver wire arrays and applied them in multiple innovative studies describing the motility patterns of the GI tracts of animal models (Lammers, 2015a, 2015b).
Clinical methods for the translation of GI HR mapping are now emerging. The principle device applied to date in human studies has been a flexible printed circuit (FPC) array, employing regular arrays of gold contacts (Du et al., 2009). The advantages of these arrays are that they can be mass-produced and easily sterilised, but with a trade-off in signal quality, and they can only be applied at invasive surgery. A vast volume of data is retrieved, which is processed through semi-automated software algorithms to enable efficient processing (Yassi et al., 2012).
The FPC arrays have enabled the first reliable studies of human gastric slow wave activation, including pacemaker behaviour and regional variations in activity (O'Grady et al., 2010a). More recently, the arrays have also been applied to study the specific physiology of the terminal gastric antrum, where a sudden acceleration of slow waves occurs prior to the pylorus, contributing to effective trituration (Berry et al., 2016). In addition, the FPC device is also being applied in studies defining patterns of gastric dysrhythmia in patients with motility disorders, including studies into gastroparesis and chronic unexplained nausea and vomiting (CUNV) (O'Grady et al., 2012; Angeli et al., 2015). Results from these studies have enabled a provisional classification scheme of human gastric dysrhythmias, with distinction between ‘disorders of slow wave initiation’ and ‘disorders of slow wave conduction’ (O'Grady et al., 2014). The figure shows an example of the classification schemes for gastric dysrhythmia that are emerging, based on mechanisms and spatial patterns of slow wave activation. Aberrant initiation relates to abnormalities of intrinsic interstitial cells of Cajal frequencies and example activities include stable ectopic pacemakers and unstable regions of ectopic foci. Aberrant conduction involves disruption of the normal slow wave entrainment and examples include abnormal velocities, conduction blocks and re-entrant activities (Reproduced with permission from O'Grady et al., 2014). More work is required to validate this scheme and assess its utility in diagnosis and practice, but it is already proving useful in research.
The crucial next step for HR gastric electrical mapping will be to develop non-invasive devices to expand clinical investigations. Minimally-invasive (laparoscopic) devices have been proposed (O'Grady et al., 2009; Berry et al., 2016), but an endoscopic device is critically needed, to enable routine studies of gastric dysrhythmia during upper GI endoscopy. Significant progress towards this goal has been presented in preliminary form (Angeli et al., 2016b) and human studies are now awaited. In addition, while gastric dysrhythmias have been strongly implicated in generating nausea and vomiting (Koch 2014; Owyang & Hasler 2002), their significance in the overall symptom profile and diagnosis and management of motility disorders still needs to be defined (O'Grady et al., 2014).
Another important and emerging area of clinical interest is the application of HR electrical mapping to cases of gut dysmotility arising after surgical manipulations (Du et al., 2015). An example application is in patients after sleeve gastrectomy, where the native gastric pacemaker is resected (O'Grady et al.,. 2016). The same HR mapping techniques could also be applied to surgical manipulations in the small intestine, colon and rectum (Lammers 2015b),(Lammers 2013), although translational work in these areas has not yet significantly progressed.

New therapeutic directions are also needed for applications in conjunction with HR mapping, as has been successfully achieved for many years in cardiology (Tse et al., 2016). Electrical stimulation using the only currently-approved device (Enterra®, Medtronic) does not modify gastric dysrhythmias (Angeli et al., 2016a), and attention is returning to the use of long-pulse (high-energy) gastric pacing in conjunction with HR mapping (O'Grady et al., 2010, Lin et al., 1998). Whether this approach will be successful in patients with gastroparesis and CUNV, where ICC networks are depleted and damaged awaits to be seen. Another interesting proposal is the emerging concept of using targeted ablation therapy to eliminate aberrant sources of gastric slow wave initiation (Angeli et al., 2016c).
In summary, the evolution and continued application of HR gastric mapping holds promise to finally resolve longstanding questions about the clinical significance and therapeutic importance of gastric dysrhythmias. However, work is still needed to show the clinical importance of this new technology.
High-resolution colonic manometry is a second prominent emerging technique with potential to critically advance translational GI motility (Dinning et al., 2010). A key advance enabling HR colonic studies has been the development of fibre-optic catheters by Arkwright et al., which apply a fibre-Bragg grating method to achieve multi-point recordings at resolutions of 1 cm over long distances (Arkwright et al., 2009).
To date, the main group publishing HR colonic manometry work has been Dinning and colleagues in Australia (Bampton & Dinning 2013), although publications are now also emerging from groups in New Zealand and Europe (Vather et al., 2016; Corsetti et al., 2016). An important step has been to present a comprehensive description of normal baseline activity in the human colon (Dinning et al., 2014). A particularly interesting result of this work has been to reveal the prominence of lower-amplitude propagating patterns, which could not be adequately resolved with lower-resolution methods (Dinning et al., 2013).
In particular, a cyclic activity in the distal colon and rectum has now been shown to be the most active motility pattern in the post-prandial state, which often propagates in the retrograde direction, and which may play a role in limiting rectal filling. This pattern has potential clinical implications, as it appears to be enhanced by sacral nerve stimulation, pointing to a possible mechanism of action for this therapy in faecal incontinence (Patton et al., 2013). In addition, this pattern is diminished in slow transit constipation, with patients lacking the normal post-prandial activity increase, pointing to underlying neuromuscular dysfunction (Dinning et al., 2015).
Another emerging area of interest is again post-operative states of dysmotility. Vather et al recently performed HR colonic manometry in patients with normal bowel function following anterior resection, and showed that by one-year post-operatively, distal colonic motility had recovered (Vather et al., 2016). This recovery included restoration of the normal post-prandial increase in cyclic activity, as well as the ability of activity sequences to freely propagate across sites of healed anastomosis, implying regeneration of neuromuscular cellular elements through scar. Comparison studies are now awaited from patients with anterior resection syndrome, where bowel function has not recovered after surgery.
In summary, the continued application of HR colonic manometry holds promise to resolve longstanding questions about the role of dysmotility in common disease states such as severe constipation, irritable bowel syndrome, faecal incontinence and anterior resection syndrome. However, much work is still needed to determine if this technique will become a routine clinical tool.
Conclusions. It is clear that a key element to progress in clinical motility disorders will be through improved understanding of the underlying abnormal motility patterns. Towards this end, translational tools for HR motility analysis have emerged, in the form of HR electrical mapping and HR colonic manometry. Early progress in applying these tools has been encouraging, and as a result, the coming decade may prove a particularly productive period for the field of translational GI motility.
Angeli TR, Cheng LK, Du P, Wang TH, Bernard CE, Vannucchi MG, Faussone-Pellegrini MS, Lahr C, Vather R, Windsor JA, Farrugia G, Abell TL, O'Grady G8. (2015). Loss of interstitial cells of Cajal and patterns of gastric dysrhythmia in patients with chronic unexplained nausea and vomiting. Gastroenterology 149(1), 56–66.
Angeli TR, Du P, Midgley D, Paskaranandavadivel N, Sathar S, Lahr C, Abell TL, Cheng LK, O'Grady G. (2016a). Acute slow wave responses to high-frequency gastric electrical stimulation in patients with gastroparesis defined by high-resolution mapping. Neuromodulation In Press. doi: 10.1111/ner.12454.
Angeli TR, Paskaranandavadivel N, Du P, Asirvatham SJ, Farrugia G, Cheng LK, O'Grady G. (2016b). Mo1588 High-resolution mapping of slow wave propagation from the gastric mucosa using flexible printed circuit electrodes. Gastroenterology 150(4), S721.
Angeli TR, Quesada MJH, Du P, Paskaranandavadivel N, Amirapu S, Beyder A, Asirvatham SJ, Farrugia G, Cheng LK, O'Grady G. (2016c). Mo1587 Gastric ablation as a novel therapeutic technique for modulating gastric slow wave activity. Gastroenterology 150(4), S721.
Arkwright JW, Underhill ID, Maunder SA, Blenman N, Szczesniak MM, Wiklendt L, Cook IJ, Lubowski DZ, Dinning PG. (2009). Design of a high-sensor count fibre optic manometry catheter for in-vivo colonic diagnostics. Opt Express 17(25), 22423–22431.
Bampton PA & Dinning PG. (2013). High resolution colonic manometry--what have we learnt?--A review of the literature 2012. Curr Gastroenterol Rep 15(6), 328.
Berry R, Miyagawa T, Paskaranandavadivel N, Du P, Angeli TR, Trew ML, Windsor JA, Imai Y, O'Grady G, Cheng LK. (2016). Functional physiology of the human terminal antrum defined by high-resolution electrical mapping and computational modeling. Am J Physiol. Gastrointest Liver Physiol In Press. doi:p.ajpgi.00255.2016.
Cheng LK, Du P & O'Grady G. (2013). Mapping and modeling gastrointestinal bioelectricity: from engineering bench to bedside. Physiology 28(5), 310–317.
Corsetti M, Pagliaro G, Demedts I, Deloose E, Gevers A, Scheerens C, Rommel N, Tack J. (2016). Pan-colonic pressurizations associated with relaxation of the anal sphincter in health and disease: A new colonic motor pattern identified using high-resolution manometry. Am J Gastroenterol In Press. doi:10.1038/ajg.2016.341.
Dinning PG, Wiklendt L, Maslen L, Patton V, Lewis H, Arkwright JW, Wattchow DA, Lubowski DZ, Costa M, Bampton PA. (2015). Colonic motor abnormalities in slow transit constipation defined by high resolution, fibre-optic manometry. Neurogastroenterol Motil 27(3), 379–388.
Dinning PG, Wiklendt L, Gibbins I, Patton V, Bampton P, Lubowski DZ, Cook IJ, Arkwright JW. (2013). Low-resolution colonic manometry leads to a gross misinterpretation of the frequency and polarity of propagating sequences: Initial results from fibre-optic high-resolution manometry studies. Neurogastroenterol Motil 25(10), e640–9.
Dinning PG, Wiklendt L, Maslen L, Gibbins I, Patton V, Arkwright JW, Lubowski DZ, O'Grady G, Bampton PA, Brookes SJ, Costa M. (2014). Quantification of in-vivo colonic motor patterns in healthy humans before and after a meal revealed by high-resolution fibre-optic manometry. Neurogastroenterol Motil 26(10), 1443–1457.
Dinning PG, Arkwright JW, Gregersen H, O'grady G, Scott SM. (2010). Technical advances in monitoring human motility patterns. Neurogastroenterol Motil 22(4), 366–380.
Du P, O'grady G, Egbuji JU, Lammers WJ, Budgett D, Nielsen O, Windsor JA, Pullan AJ, Cheng LK. (2009). High-resolution mapping of in vivo gastrointestinal slow wave activity using flexible printed circuit board electrodes: methodology and validation. Annals Biomed Eng 37(4), 839–846.
Du P, Hameed A, Angeli TR, Lahr C, Abell TL, Cheng LK, O'Grady G. (2015). The impact of surgical excisions on human gastric slow wave conduction, defined by high-resolution electrical mapping and in silico modeling. Neurogastroenterol Motil 27(10), 1409–1422.
Kahrilas PJ, Bredenoord AJ, Fox M, Gyawali CP, Roman S, Smout AJ, Pandolfino JE. (2015). The Chicago Classification of esophageal motility disorders, v3.0. Neurogastroenterol Motil 27(2), 160–174.
Koch KL. (2014) Gastric dysrhythmias: a potential objective measure of nausea. Exp Brain Res 232(8), 2553–2561.
Lammers W. (2013). Arrhythmias in the gut. Neurogastroenterol Motil 25, 353-357.
Lammers WJ. (2015a). Inhomogeneities in the propagation of the slow wave in the stomach. Neurogastroenterol Motil 27(10), 1349–1353.
Lammers WJEP. (2015b). Normal and abnormal electrical propagation in the small intestine. Acta physiol 213(2), 349–359.
Lin ZY, McCallum RW, Schirmer BD, Chen JD. (1998). Effects of pacing parameters on entrainment of gastric slow waves in patients with gastroparesis. Am J Physiol, 274, G186–91.
O'Grady G, Berry R, Paskaranandavadivel N, Angeli TR, Beban G, Du P, Cheng K. (2016). Ectopic gastric pacemaking after sleeve gastrectomy and feasibility of gastric pacing therapy. Neurogastroenterol Motil 28, S83-4.
O'Grady G, Du P, Egbuji JU, Lammers WJ, Wahab A, Pullan AJ, Cheng LK, Windsor JA. (2009). A novel laparoscopic device for measuring gastrointestinal slow-wave activity. Surg Endoscopy 23(12), 2842–2848.
O'Grady G, Wang TH, Du P, Angeli T, Lammers WJ, Cheng LK. (2014). Recent progress in gastric arrhythmia: pathophysiology, clinical significance and future horizons. Clin Exp Pharmacol Physiol 41(10), 854–862.
O'Grady G, Angeli TR, Du P, Lahr C, Lammers WJ, Windsor JA, Abell TL, Farrugia G, Pullan AJ, Cheng LK. (2012). Abnormal initiation and conduction of slow-wave activity in gastroparesis, defined by high-resolution electrical mapping. Gastroenterology 143(3), 589–98.
O'Grady G, Du P, Cheng LK, Egbuji JU, Lammers WJ, Windsor JA, Pullan AJ. (2010a). Origin and propagation of human gastric slow-wave activity defined by high-resolution mapping. Am J Physiol Gastrointest Liver Physiol 299(3), G585–92.
O'Grady G, Du P, Paskaranandavadivel N, Angeli TR, Lammers WJ, Asirvatham SJ, Windsor JA, Farrugia G, Pullan AJ, Cheng LK. (2012). Rapid high-amplitude circumferential slow wave propagation during normal gastric pacemaking and dysrhythmias. Neurogastroenterol Motil 24(7), e299–312.
Owyang C & Hasler WL. (2002). Physiology and pathophysiology of the interstitial cells of Cajal: from bench to bedside. VI. Pathogenesis and therapeutic approaches to human gastric dysrhythmias. Am J Physiol Gastrointest Liver Physiol 283(1), G8–15.
Patton V, Wiklendt L, Arkwright JW, Lubowski DZ, Dinning PG. (2013). The effect of sacral nerve stimulation on distal colonic motility in patients with faecal incontinence. Br J Surg 100(7), 959–968.
Szurszewski, J.H., (1998). A 100-year perspective on gastrointestinal motility. Am J Physiol Gastrointest Liver Physiol 274, G447-53.
Tse G, Lai ET, Yeo JM, Tse V, Wong SH. (2016). Mechanisms of electrical activation and conduction in the gastrointestinal system: Lessons from cardiac electrophysiology. Frontiers Physiol 7(77), 182.
Vather R, O'Grady G, Arkwright JW, Rowbotham DS, Cheng LK, Dinning PG, Bissett IP. (2016). Restoration of normal colonic motor patterns and meal responses after distal colorectal resection. Br J Surg 103(4), 451–461.
Yassi R, O'Grady G, Paskaranandavadivel N, Du P, Angeli TR, Pullan AJ, Cheng LK, Erickson JC. (2012). The gastrointestinal electrical mapping suite (GEMS): software for analyzing and visualizing high-resolution (multi-electrode) recordings in spatiotemporal detail. BMC Gastroenterol 12(1), 60.