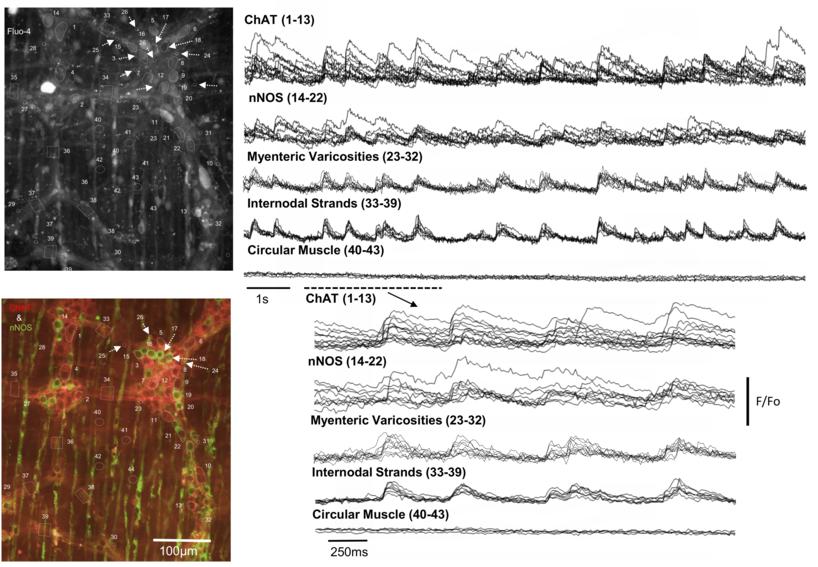
How the enteric nervous system generates propagating neurogenic contractions along the gastrointestinal (GI) tract has remained mysterious. In this study, we used mouse colon to determine intrinsic neurogenic mechanisms underlying cyclical, propagating contractions along the full length of colon, that are known as colonic migrating motor complexes (CMMCs). Whilst it is clear that CMMCs require the enteric nervous system (ENS), it is not clear how the activation of the different classes of myenteric neurons is coordinated over large regions colon to generate an orderly contraction of smooth muscle. In this study, we used the latest high resolution, EMCCD camera (Evolve Delta; Photometrics) to visualize the temporal activation properties of different morphological classes of myenteric neurons in multiple myenteric ganglia along the colon. To do this, the entire colon was removed from mice, euthanized in accordance with the animal welfare committee of Flinders University and mounted serosal side uppermost in an oxygenated Krebs solution at 35°C. To characterize the temporal firing properties of myenteric neurons during these rapid oscillations in the muscle, the calcium indicator, Fluo-4 was loaded into the myenteric plexus. Calcium imaging of intact whole colon revealed that during each CMMC, multiple neighboring myenteric ganglia and internodal strands generated time-locked calcium transients at a remarkably rhythmic frequency of ∼2Hz. The figure shows simultaneous neuronal imaging from myenteric ganglia revealed that multiple ChAT and nNOS+ve neuronal cell bodies, varicosities and internodal strands generated time-locked calcium transients during the colonic migrating motor complex. Left hand images show the raw camera image (top) and the same region stained for nNOS (green) and ChAT (red).

Each CMMC was associated with a repetitive discharge (at ∼2Hz) of synchronized calcium transients in large populations of cholinergic and nitrergic neuronal cell bodies across multiple rows of neighboring myenteric ganglia (including internodal strands) (N=15). The frequency of the repetitive calcium transients in myenteric ganglia (2.0 ± 0.1Hz) and internodal strands (2.2 ± 0.1Hz) was the same as the action potentials evoked in the neighboring smooth muscle layers. All synchronized calcium transients across multiple ganglia were abolished by hexamethonium (N=6). This is the first demonstration of a rhythmic neuronal firing pattern in mammalian enteric nervous system. Moreover, this firing pattern is responsible for repetitive time-locked action potentials in the smooth muscle. Of particular interest was the discovery that large CGRP-positive Dogiel type 2 neurons generated repetitive hexamethonium-sensitive calcium transients at the same time as small cholinergic and nitrergic neurons during each CMMC.