Aims. Skeletal muscle insulin resistance and oxidative stress are characteristic metabolic disturbances in people with type 2 diabetes. Studies in insulin resistant rodents show an improvement in skeletal muscle insulin sensitivity and oxidative stress following antioxidant supplementation. We therefore investigated the potential ameliorative effects of antioxidant ascorbic acid (AA) supplementation on skeletal muscle insulin sensitivity and oxidative stress in people with type 2 diabetes.
Methods. Thirteen people with type 2 diabetes (12M, 1F; aged 57.9 ± 2.5y [mean ± SE]) and stable glucose control (HbA1c 7.6 ± 0.2%) commenced a randomized cross-over study involving four months of AA (2 × 500 mg/day) supplementation and 4 months of placebo supplementation. A one month wash-out separated the treatments. Insulin sensitivity was assessed using a hyperinsulinaemic, euglycaemic clamp coupled with infusion of 6,6-D2 glucose. Muscle biopsies were measured for AA concentration and oxidative stress markers that included basal measures (2',7'-dichlorofluorescin [DCFH] oxidation, ratio of reduced-to-oxidized glutathione [GSH/GSSG] and F2-Isoprostanes) and insulin-stimulated measures (DCFH oxidation). Antioxidant concentrations, citrate synthase activity and protein abundances of sodium-dependent vitamin C transporter 2 (SVCT2), total Akt and phosphorylated Akt (ser473) were also measured.
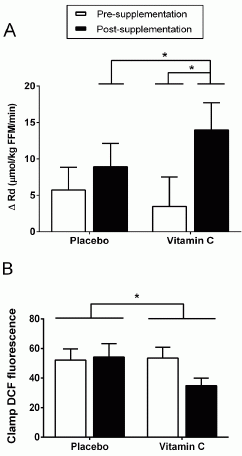
Results. In compliant participants (n=7 [6M, 1F], aged 59.4 ± 3.5y), AA supplementation significantly increased insulin-mediated glucose disposal (delta rate of glucose disappearance; ΔRd) (p=0.009; Fig.1), peripheral insulin-sensitivity index (p=0.046), skeletal muscle AA concentration (p=0.017) and muscle SVCT2 protein expression (p=0.008); but significantly decreased skeletal muscle DCFH oxidation during hyperinsulinaemia (p=0.007; Fig.1) when compared with placebo. Total superoxide dismutase activity was also lower following AA supplementation when compared with placebo (p=0.006). Basal oxidative stress markers, citrate synthase activity, endogenous glucose production, HbA1C and muscle Akt expression were not significantly altered by AA supplementation.

Conclusions. In summary, oral AA supplementation (2 × 500mg over 4 months) increases skeletal muscle vitamin C concentration, ameliorates skeletal muscle oxidative stress during hyperinsulinaemia, and improves insulin-mediated glucose disposal in people with type 2 diabetes. Future studies should investigate the effectiveness of this adjuvant supplementation regimen on glycaemic control in larger cohorts and during physiological postprandial periods. Findings implicate AA supplementation as a potentially inexpensive, convenient, and effective adjunct therapy in the treatment of insulin resistance in people with type 2 diabetes.