Recently the skeletal muscle relaxant dantrolene sodium (DaNa) has been investigated for its ability to modulate SR calcium release in cardiac muscle, with the potential of it being used as a treatment of heart failure. How this modulation occurs is still not well understood. The use of dantrolene analogues may provide some useful insight into the structure activity relationship (SAR) as well as provide future lines of investigation for potential therapeutics of heart failure. Here we investigated the effect of DaNa, azumolene, and two synthesised DaNa analogues on Ca2+ loading and release in skinned cardiomyocytes and on calsequestrin (CSQ2) Ca2+ binding properties.
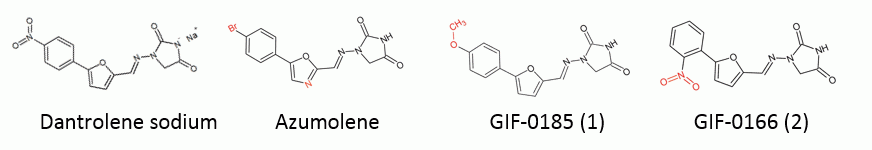
DaNa and azumolene were bought commercially (Sigma) while analogues GIF-0185 (1) and GIF-0166 (2) were synthesised using the procedure described by Hosoya et al., (2003). The structure of dantrolene and the analogues can be seen in the figure below with the differences highlighted in red and will be referred to as analogue 1 and 2. Fresh sheep hearts were obtained from Hardwick’s abattoirs Kyneton, Victoria and the right ventricle was enzymatically digested to produce single cardiac cells. The cells were then allowed to adhere to a 96-well plate (black, clear bottom), by incubating overnight (5% CO2, 37°C). The cells were subsequently skinned (sarcolemma made leaky) for 10 to 45 min in a sodium-based solution containing 0.1% w/v saponin, 8mM ATP and 1mM EGTATotal. After skinning the cells were washed in a solution containing 49.9mM HDTATotal + 0.1mM EGTATotal and resuspended in the same solution containing the Ca2+-sensitive fluorescent dye Fluo4 (5μM). Ca2+ dependence on SR loading and release was investigated using Ca2+ loading solutions (pCa = −log10[Ca2+] 6.54, 6.26, 6.00 and 5.83 0.1mM EGTATotal, 8mM ATP, 1mM Mg2+ for 3.0 min). SR Ca2+ release was induced by the addition of caffeine (24mM final concentration). The fluorescent signal from Fluo4 (Ex 494, Em 516 nm) was continually monitored using the Flexstation 3 (read rate 1.15s for up to 5 min). This was compared to the maximal Ca2+ signal recorded after destroying all the membranous compartments by exposure to 1% v/v Triton X-100 for up to 5 minutes. The effect of dantrolene sodium and the analogues (1μM) on SR Ca2+ loading and release were investigated. Purified recombinant CSQ2 was used to study the effect of all compounds on calcium binding. A plasmid containing the human calsequestrin 2 gene (GeneCopoeia) was inserted into single step KRX competent cells (Promega) and expressed as per the protocol provided (Promega). CSQ2 was purified using a GST affinity column (Sigma) and GST tag cleaved using TEV protease (Sigma). CSQ2 native fluorescence (Ex 280nm, Em 340nm), turbidity (Abs 350nm) and free Ca2+ in solution (measure with Rhod5N; Ex 551nm, Em 576nm) were monitored in the presence of increasing micromolar and millimolar concentrations of Ca2+ in the absence and presence of the drugs (10μM).

DaNa enhanced both SR Ca2+ loading and subsequent release in the presence of lower pCa solutions (pCa 5.83 and 6.26) compared with control, and increased both CSQ2 polymerization and Ca2+ binding. Azumolene slightly increased SR loading and release but had no effect on CSQ2 Ca2+ binding properties. Analogues 1 and 2 had different effects; with analogue 1 enhancing SR Ca2+ release when present in the release solution only, but reduced CSQ2 polymerization; while analogue 2 consistently enhanced SR Ca2+ loading and subsequent release for all pCa used. Analogue 2 also significantly enhanced CSQ2 polymerization at both micromolar and millimolar Ca2+ concentrations. It appears that substitution in the para position of the benzene ring had more effect on the gating properties of the RyR2, while the nitro group was associated with changes in CSQ2 polymerization and Ca2+ binding. The specificity of the analogues may allow SR Ca2+ capacity or RyR2 leak to be targeted independently in heart failure.
Hosoya T, Aoyama H, Ikemoto T, Kihara Y, Hiramatsu T, Endo M, Suzuki M. (2003) Bioorg Med Chem 11, 663-673.